2.2D Lewis Diagrams and Structural Isomerism- Pre AP Chemistry Study Notes - New Syllabus.
2.2D Lewis Diagrams and Structural Isomerism- Pre AP Chemistry Study Notes
2.2D Lewis Diagrams and Structural Isomerism- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.2.D.1 Create and/or evaluate Lewis diagrams for molecular compounds and/or polyatomic ions.
2.2.D.2 Determine if given molecules are structural isomers.
Key Concepts:
- 2.2.D A Lewis diagram is a simplified representation of a molecule.
a. Lewis diagrams show the bonding patterns between atoms in a molecule.
b. Molecules with the same number and type of atoms but different bonding patterns are structural isomers, which have different properties from one another.
2.2.D.1 — Lewis Diagrams for Molecular Compounds and Polyatomic Ions
Lewis diagrams (Lewis structures) are particle-level models that show how valence electrons are arranged in molecular compounds and polyatomic ions. They are used to create and evaluate models of bonding by representing shared and unshared electrons.
Purpose of Lewis Diagrams
Lewis diagrams are used to:
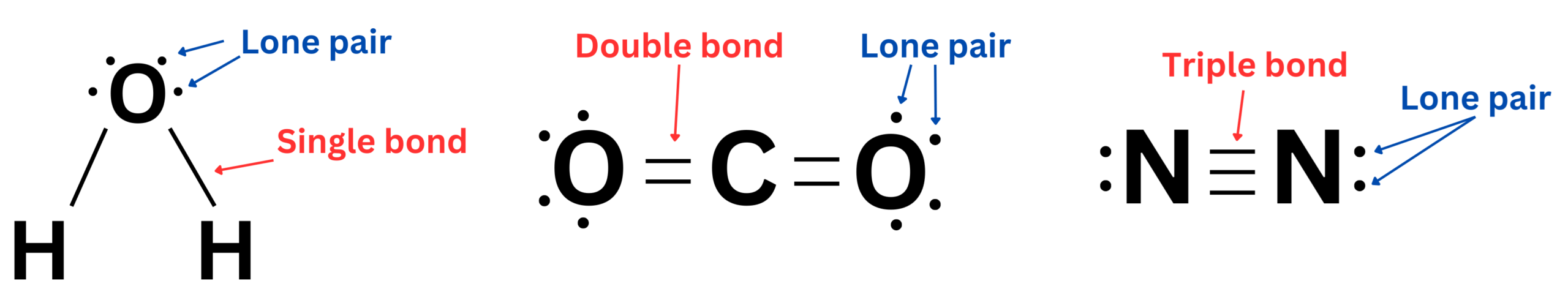
- Show how atoms are bonded in a molecule or ion
- Represent valence electrons as dots or shared pairs
- Determine whether atoms achieve stable electron arrangements
These diagrams model intramolecular forces (covalent bonds), not intermolecular forces.
Valence Electrons and the Octet Rule
The octet rule states that atoms tend to bond in ways that give them eight electrons in their outer shell (exceptions apply).
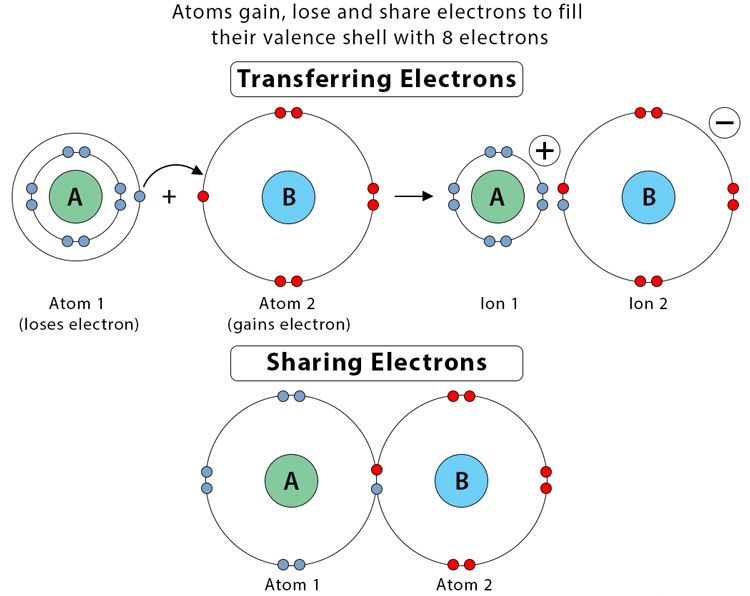
- Hydrogen is stable with 2 electrons
- Main-group elements generally follow the octet rule
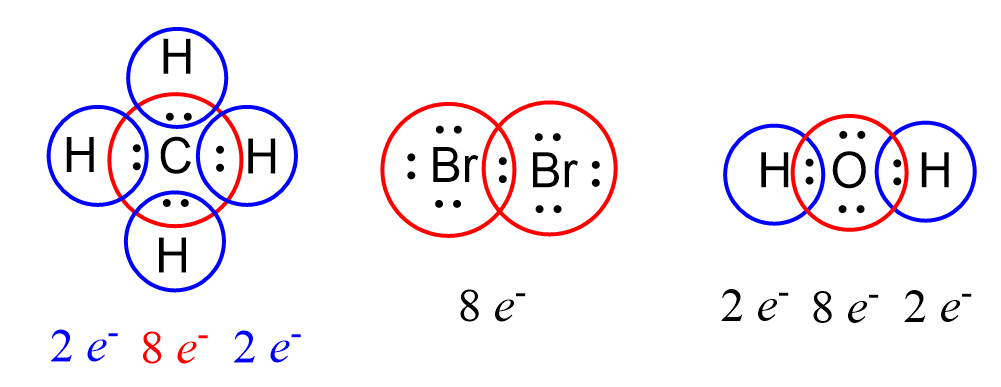
Lewis diagrams track valence electrons to check octet fulfillment.
Steps to Draw Lewis Diagrams (Molecular Compounds)
- Count total valence electrons for all atoms
- Identify the central atom (least electronegative, never H)
- Connect atoms with single bonds (2 electrons each)
- Distribute remaining electrons to satisfy octets
- Form double or triple bonds if needed
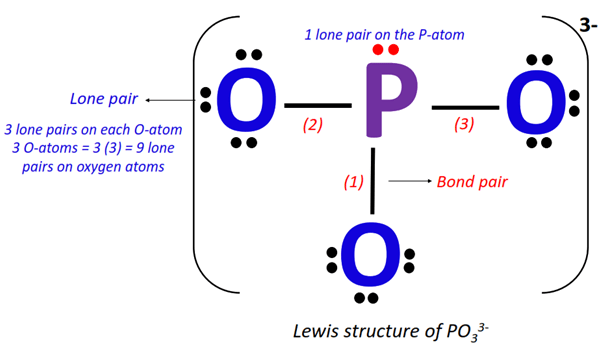
Lewis Diagrams for Polyatomic Ions
Polyatomic ions are charged groups of covalently bonded atoms. Their Lewis diagrams are drawn similarly to molecules, but with two key differences:
- Total electrons are adjusted for the ion’s charge
- The structure is enclosed in brackets with the charge shown
Negative charge → add electrons Positive charge → remove electrons
Evaluating Lewis Diagrams
A correct Lewis diagram must:
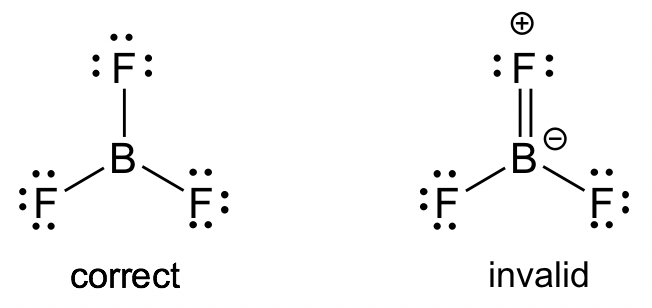
- Use the correct number of total valence electrons
- Show appropriate bonding between atoms
- Satisfy octet (or duet) requirements where applicable
- Correctly represent ion charge (for polyatomic ions)
Diagrams that miscount electrons or ignore charge are incorrect.
Common Errors to Avoid
- Placing hydrogen as a central atom
- Exceeding hydrogen’s duet rule
- Forgetting to adjust electrons for ionic charge
- Ignoring the need for multiple bonds
Example
Create a Lewis diagram for carbon dioxide, CO₂, and evaluate whether the octet rule is satisfied.
▶️ Answer / Explanation
Carbon contributes 4 valence electrons, and each oxygen contributes 6, giving a total of 16 valence electrons.
Carbon is the central atom. Two double bonds form between carbon and oxygen.![]()
Step 1: Count valence electrons: Carbon (4) + Oxygen (6 × 2) = 16 electrons.
Step 2: Place carbon in the center with two oxygen atoms bonded to it.
Step 3: Place single bonds first → 4 electrons used, 12 left.
Step 4: Distribute remaining electrons to complete octets: each oxygen gets 6 more electrons (12 total used).
Step 5: Carbon has only 4 electrons, so convert lone pairs into double bonds with each oxygen.
Example
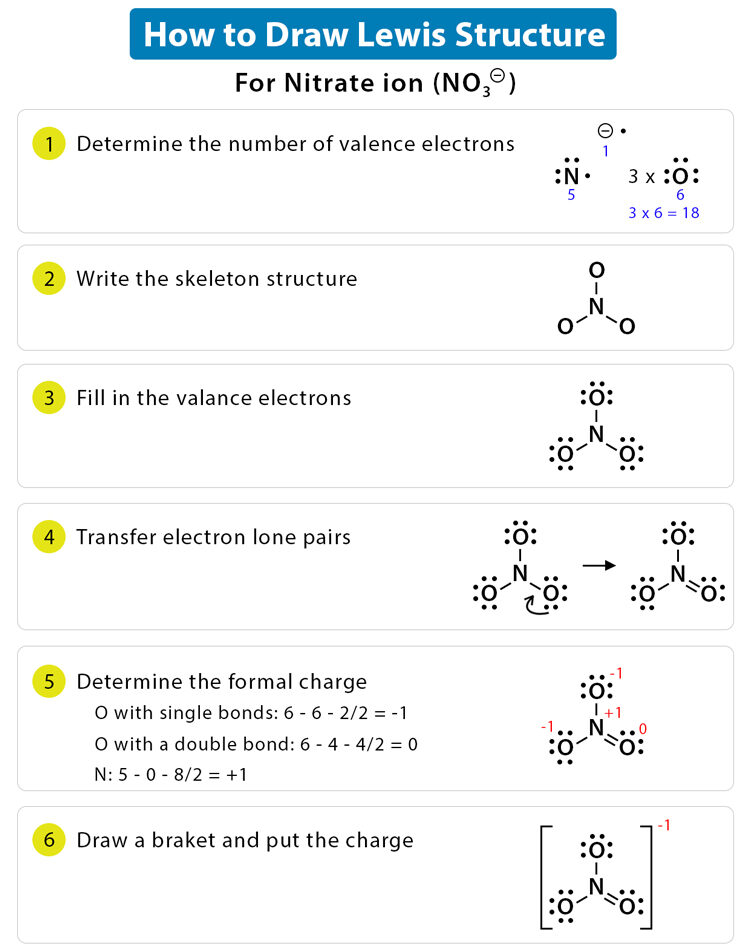
Create and evaluate a Lewis diagram for the nitrate ion, NO₃⁻. Explain how the charge is represented and how bonding is arranged.
▶️ Answer / Explanation
Nitrogen contributes 5 valence electrons, and three oxygens contribute 18, plus one extra electron for the −1 charge, totaling 24 electrons.
Nitrogen is the central atom, bonded to three oxygens. One double bond and two single bonds are formed to satisfy octets.
The structure is enclosed in brackets with a −1 charge shown. The diagram correctly represents a polyatomic ion.![]()
Step 1: Count valence electrons: N (5) + O (6×3) + 1 (extra for charge) = 24 electrons.
Step 2: Place N in the center, bond to 3 O atoms with single bonds (6 electrons used).
Step 3: 18 electrons left → distribute to O atoms to complete octets.
Step 4: N has only 6 electrons → make one double bond with an O.
Step 5: Place brackets with a –1 charge.
2.2.D.2 — Structural Isomers
Structural isomers are compounds that have the same molecular formula but differ in the arrangement (connectivity) of their atoms. To determine whether given molecules are structural isomers, both the formula and the bonding arrangement must be evaluated.
Key Definition
Structural isomers:
![]()
- Have the same molecular formula
- Have different connectivity between atoms
- Are different substances with different properties
Changing how atoms are bonded creates a new compound, even if the formula stays the same.
What Structural Isomers Are NOT
- They are not the same molecule drawn differently
- They are not molecules with different molecular formulas
- They are not Lewis structures showing resonance
Resonance structures have the same connectivity; structural isomers do not.
Steps to Determine Structural Isomers
![]()
- Compare molecular formulas
→ If formulas differ, they are not structural isomers - Compare atom connectivity
→ Check which atoms are bonded to each other - Look for different carbon skeletons or functional group placement
If the atoms are connected differently, the molecules are structural isomers.
Common Ways Connectivity Can Differ
- Straight-chain vs branched structures
- Functional groups attached to different carbons
- Different bonding patterns between the same atoms
Particle-Level Interpretation
Structural isomers differ because electrons are shared between different pairs of atoms. This changes molecular shape, polarity, boiling point, and reactivity.
Evaluating Structural Isomer Claims
A correct claim must state that:
- The molecules have the same molecular formula
- The atoms are connected differently
- The compounds are distinct substances
Claims based only on visual appearance without checking connectivity are incomplete.
Example
Two molecules both have the formula C₄H₁₀. One has a straight chain of four carbons, and the other has a branched carbon structure. Determine whether they are structural isomers.
▶️ Answer / Explanation
Yes, the molecules are structural isomers.

They have the same molecular formula but different carbon connectivity (straight-chain versus branched), making them different substances.
Example
Two compounds have the molecular formula C₂H₆O. One is an alcohol and the other is an ether. Determine if they are structural isomers and justify your answer.
▶️ Answer / Explanation
Yes, the compounds are structural isomers.
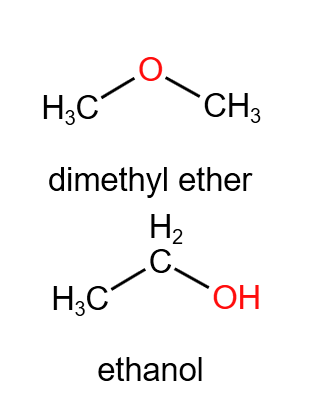
Although both have the same molecular formula, the atoms are connected differently: one has an O–H bond (alcohol) while the other has an O bonded between two carbons (ether).
The different connectivity results in different functional groups and properties.
