2.2G Intermolecular Forces and Molecular Polarity- Pre AP Chemistry Study Notes - New Syllabus.
2.2G Intermolecular Forces and Molecular Polarity- Pre AP Chemistry Study Notes
2.2G Intermolecular Forces and Molecular Polarity- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.2.G.1 Create and/or evaluate a claim about the strength and type(s) of intermolecular forces present in a sample based on molecular polarity.
Key Concepts:
- 2.2.G Molecular geometry determines if a molecule has a permanent dipole and therefore the type(s) of intermolecular forces present in that molecule.
2.2.G.1 — Molecular Polarity and Intermolecular Forces
The strength and type(s) of intermolecular forces (IMF) present in a sample can be determined by analyzing molecular polarity. Molecular polarity itself is determined by a molecule’s electron distribution and molecular geometry.
Accordingly, molecular geometry determines whether a molecule has a permanent dipole, which directly controls the intermolecular forces that can operate between particles.
Electrostatic Origin of Intermolecular Forces
All intermolecular forces arise from electrostatic attractions between regions of partial positive and partial negative charge.
- Uneven electron distribution → polarity
- Polarity → stronger intermolecular attractions
The greater the polarity, the stronger the intermolecular forces.
Molecular Geometry and Permanent Dipoles
A molecule has a permanent dipole if:
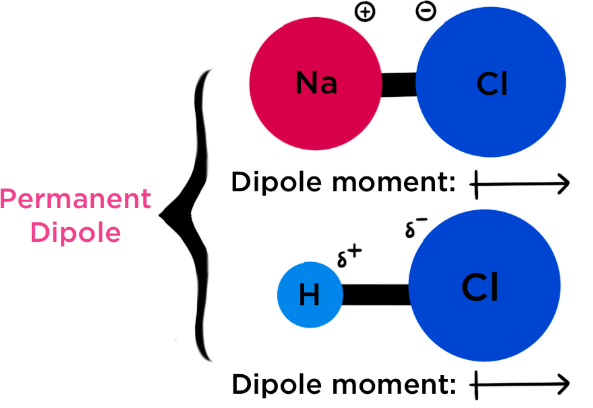
- It contains polar bonds, and
- The molecular geometry is asymmetric
Symmetrical molecules do not have a permanent dipole, even if they contain polar bonds.
Polarity and Types of Intermolecular Forces
Nonpolar Molecules
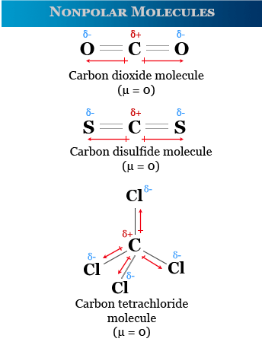
- No permanent dipole
- Even electron distribution
- Only London dispersion forces present
London dispersion forces are weak and result from temporary fluctuations in electron density.
Polar Molecules
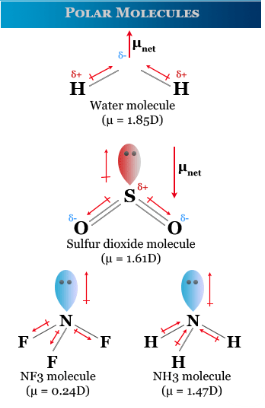
- Permanent dipole present
- Asymmetric electron distribution
- Experience multiple intermolecular forces
Polar molecules experience:
- London dispersion forces
- Dipole–dipole attractions
Hydrogen Bonding and Polarity
Hydrogen bonding is a particularly strong form of dipole–dipole attraction.
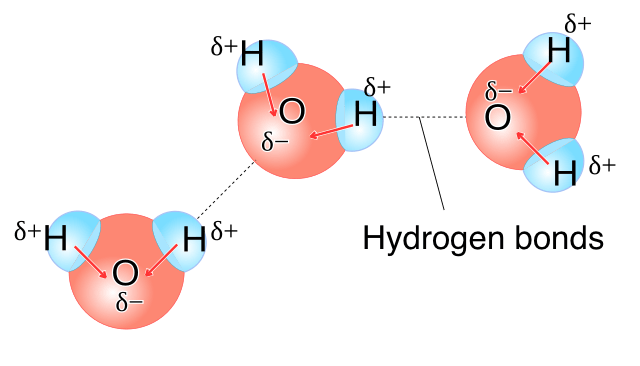
It occurs when:
- Hydrogen is bonded to N, O, or F
- The molecule is polar
- A lone pair is available on a neighboring N, O, or F atom
Molecules capable of hydrogen bonding have stronger intermolecular forces than similar molecules without it.
Comparing IMF Based on Molecular Polarity
| Molecular Property | Type(s) of IMF Present | Relative Strength |
|---|---|---|
| Nonpolar molecule | London dispersion only | Weakest |
| Polar molecule | London dispersion + dipole–dipole | Moderate |
| Polar molecule with H–N/O/F | Dispersion + dipole–dipole + hydrogen bonding | Strongest (IMF) |
Creating and Evaluating Claims
A valid claim about intermolecular forces must:
- Identify whether the molecule is polar or nonpolar
- Link polarity to molecular geometry
- Specify the type(s) of intermolecular forces present
- Compare relative IMF strength when relevant
Claims that ignore geometry or electron distribution are incomplete.
Example
Carbon tetrachloride, CCl₄, contains polar C–Cl bonds. Determine the type(s) of intermolecular forces present and justify your claim.
▶️ Answer / Explanation
Carbon tetrachloride experiences only London dispersion forces.
Although the C–Cl bonds are polar, the tetrahedral geometry is symmetrical, so the bond dipoles cancel and the molecule is nonpolar.
Example
Compare the intermolecular forces present in methanol (CH₃OH) and propane (C₃H₈). Use molecular polarity and geometry to support your claim.
▶️ Answer / Explanation
Methanol has stronger intermolecular forces than propane.
Methanol is polar and contains an O–H bond, allowing for hydrogen bonding, dipole–dipole attractions, and London dispersion forces.
Propane is nonpolar due to its symmetrical geometry and experiences only London dispersion forces.
