2.3B Properties of Ionic and Covalent Compounds- Pre AP Chemistry Study Notes - New Syllabus.
2.3B Properties of Ionic and Covalent Compounds- Pre AP Chemistry Study Notes
2.3B Properties of Ionic and Covalent Compounds- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
2.3.B.1 Interpret the results of an experiment to determine the type of bonding present in a substance.
Key Concepts:
- 2.3.B Ionic and covalent compounds have different properties based on their bonding.
a. Properties of ionic compounds result from electrostatic attractions of constituent ions.
b. Properties of covalent compounds result from bonds created by the sharing of electrons and intermolecular forces.
2.3.B.1 — Interpreting Experimental Results to Determine Bonding Type
The type of bonding present in a substance can be determined by carefully interpreting experimental results and connecting observable properties to particle-level explanations.
Because ionic and covalent compounds have fundamentally different bonding, they display distinct macroscopic properties that can be used as evidence to support a scientific claim.
Particle-Level Basis of Bonding
Understanding experimental data requires recognizing how particles interact:
![]()
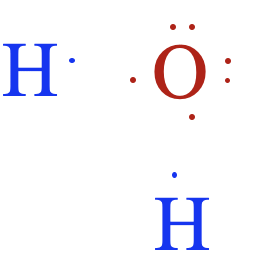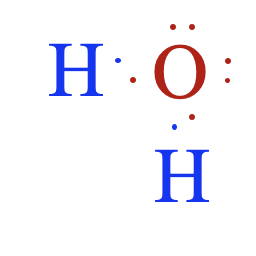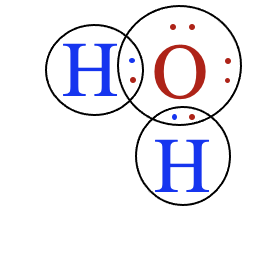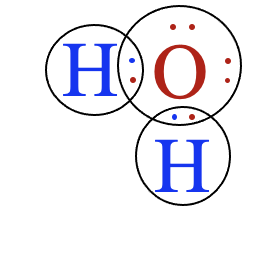
- Ionic compounds consist of positively and negatively charged ions arranged in a crystal lattice. These ions are held together by strong electrostatic attractions.
- Covalent compounds consist of neutral molecules formed by the sharing of electrons. Molecules attract one another only through intermolecular forces.
These particle-level differences directly cause the properties observed in experiments.
Experimental Test 1: Electrical Conductivity
Electrical conductivity tests whether charged particles can move freely.
![]()
- Ionic compounds do not conduct as solids because ions are fixed in place within the lattice.
- Ionic compounds do conduct electricity when molten or dissolved, because ions are free to move and carry charge.
- Covalent compounds do not conduct electricity in any state, because they are made of neutral molecules.
Conductivity is one of the strongest indicators of ionic bonding.
Experimental Test 2: Melting Point
Melting point reflects how strongly particles are held together.
![]()
![]()
- Ionic compounds have very high melting points due to strong electrostatic forces between ions.
- Covalent compounds have low to moderate melting points because intermolecular forces are much weaker than ionic attractions.
A high melting point suggests ionic bonding, while a low melting point suggests covalent bonding.
Experimental Test 3: Solubility in Water
Solubility depends on how particles interact with water molecules.
![]()
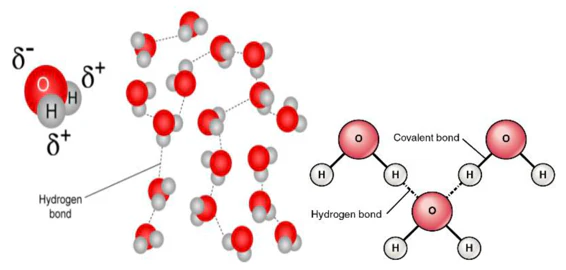
- Many ionic compounds dissolve in water because water stabilizes ions through ion–dipole attractions.
- Polar covalent compounds may dissolve due to dipole–dipole interactions and hydrogen bonding with water.
- Nonpolar covalent compounds typically do not dissolve in water.
Solubility alone is not conclusive, but it supports other experimental evidence.
Experimental Test 4: Physical State and Appearance
The physical state of a substance provides additional clues.
![]()
- Ionic compounds are usually hard, brittle crystalline solids.
- Covalent compounds may be gases, liquids, or soft solids.
Crystalline appearance and brittleness strongly suggest ionic bonding.
Connecting Experimental Evidence to Bonding Type
| Observed Property | Likely Bonding | Particle-Level Reason |
|---|---|---|
| Conducts electricity in solution | Ionic | Mobile ions carry charge |
| Very high melting point | Ionic | Strong electrostatic attractions |
| Low melting point, nonconductive | Covalent | Weak intermolecular forces |
| Gas or liquid at room temperature | Covalent | Discrete neutral molecules |
Creating and Evaluating Claims from Experimental Data
A strong scientific claim must:
- Cite specific experimental observations
- Explain what those observations reveal about particle behavior
- Correctly identify ionic or covalent bonding
Listing observations without linking them to bonding mechanisms is insufficient.
Example
An unknown solid dissolves in water, and the resulting solution conducts electricity. Interpret these experimental results to determine the bonding type.
▶️ Answer / Explanation
The compound most likely contains ionic bonding.
Dissolving in water produces free-moving ions, which allow the solution to conduct electricity. This behavior is characteristic of ionic compounds held together by electrostatic attraction.
Example
A substance has a melting point above 800 °C, forms a brittle crystal, and does not conduct electricity as a solid but does conduct when molten. Use the experimental data to identify the bonding type and justify your reasoning.
▶️ Answer / Explanation
The substance contains ionic bonding.
The high melting point and brittle crystal structure indicate strong electrostatic attractions. Electrical conductivity when molten confirms the presence of mobile ions, which is a defining property of ionic compounds.
