3.1B Ideal Gas Law and Gas Particles- Pre AP Chemistry Study Notes - New Syllabus.
3.1B Ideal Gas Law and Gas Particles- Pre AP Chemistry Study Notes
3.1B Ideal Gas Law and Gas Particles- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
3.1.B.1 Explain the relationships between macroscopic properties of gas samples.
3.1.B.2 Perform calculations using the ideal gas law.
3.1.B.3 Create and/or evaluate models based on the ideal gas law.
Key Concepts:
- 3.1.B The ideal gas law describes the mathematical relationship between pressure, volume, number of gas particles, and temperature.
a. Two samples of gas with the same pressure, volume, and temperature have the same number of particles.
b. The mass of the particles can be computed from atomic masses.
c. Because macroscopic samples of a gas contain many particles, moles are useful units for counting particles.
3.1.B.1 — Relationships Between Macroscopic Properties of Gas Samples
The behavior of gases can be described using macroscopic properties such as pressure, volume, temperature, and the number of particles. These properties are mathematically related by the ideal gas law, which connects observable measurements to particle-level behavior.
Because a macroscopic gas sample contains an enormous number of particles, moles are used as a practical unit to count particles and relate them to mass.
Macroscopic Properties of Gases
![]()
- Pressure (P) — force exerted by gas particles colliding with container walls
- Volume (V) — space occupied by the gas
- Temperature (T) — measure of average kinetic energy of particles (in kelvin)
- Amount (n) — number of gas particles, measured in moles
Changing any one of these properties affects the others.
The Ideal Gas Law
The relationship among the macroscopic properties of a gas is given by:![]()
\( \mathrm{PV = nRT} \)
- \( \mathrm{P} \) = pressure
- \( \mathrm{V} \) = volume
- \( \mathrm{n} \) = number of moles of gas
- \( \mathrm{R} \) = ideal gas constant
- \( \mathrm{T} \) = temperature in kelvin
This equation shows that no gas property acts independently.
Same Pressure, Volume, and Temperature → Same Number of Particles
From the ideal gas law, if two gas samples have the same pressure, volume, and temperature, then:
\( \mathrm{n = \dfrac{PV}{RT}} \)
Because \( \mathrm{P} \), \( \mathrm{V} \), and \( \mathrm{T} \) are the same, the number of moles \( \mathrm{n} \) must also be the same.
This means both samples contain the same number of gas particles, regardless of the identity of the gas.
Role of Moles in Counting Gas Particles
Gas samples contain on the order of \( \mathrm{10^{23}} \) particles. Counting individual particles is impractical, so chemists use moles.
One mole of any gas contains:
\( \mathrm{6.022 \times 10^{23}\ particles} \)
Thus, the amount of gas in the ideal gas law is expressed in moles.
Relating Number of Particles to Mass
The mass of a gas sample depends on:
- The number of particles (moles)
- The mass of each particle (molar mass)
Once the number of moles is known, the mass can be calculated using:
\( \mathrm{m = nM} \)
Different gases with the same \( \mathrm{n} \), \( \mathrm{P} \), \( \mathrm{V} \), and \( \mathrm{T} \) can have different masses because their particles have different molar masses.
Connecting the Ideas
The ideal gas law links all macroscopic properties:
- Pressure depends on particle collisions
- Temperature reflects particle kinetic energy
- Volume determines collision frequency
- Moles represent how many particles are present
Changing the number of particles changes pressure or volume, unless temperature also changes.
Creating and Evaluating Claims
A correct explanation must:
- Use the ideal gas law to connect properties
- Explain why equal \( \mathrm{P} \), \( \mathrm{V} \), and \( \mathrm{T} \) imply equal moles
- Distinguish between number of particles and mass
Example
Two gas samples are held at the same pressure, volume, and temperature. One is oxygen gas and the other is nitrogen gas. Compare the number of particles in each sample.
▶️ Answer / Explanation
The two samples contain the same number of particles.
Because the pressure, volume, and temperature are the same, the ideal gas law requires the number of moles to be equal for both gases.
Example
Two gas samples have the same volume and temperature. Sample A has a higher pressure than Sample B. Explain, using the ideal gas law, what this indicates about the number of particles in Sample A compared to Sample B.
▶️ Answer / Explanation
Sample A contains more gas particles than Sample B.
At constant volume and temperature, an increase in pressure corresponds to an increase in the number of moles. More particles result in more frequent collisions with the container walls, producing higher pressure.
3.1.B.2 — Calculations Using the Ideal Gas Law
The ideal gas law is used to calculate unknown gas properties by relating pressure, volume, temperature, and the amount of gas. This equation connects measurable macroscopic quantities to the number of gas particles present.
Ideal gas law calculations are valid when gases behave ideally, which is a good approximation under typical laboratory conditions.
The Ideal Gas Law Equation
The mathematical relationship is:
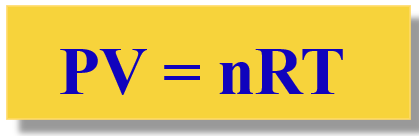
- \( \mathrm{P} \) = pressure
- \( \mathrm{V} \) = volume
- \( \mathrm{n} \) = amount of gas (moles)
- \( \mathrm{R} \) = ideal gas constant
- \( \mathrm{T} \) = temperature in kelvin
Every ideal gas law problem uses this same equation, with the variables rearranged as needed.
Units and Constants
To perform correct calculations, units must be consistent. The most commonly used value of the gas constant is:
\( \mathrm{R = 0.08206\ L\,atm\,mol^{-1}\,K^{-1}} \)
When using this value:
- Pressure must be in atmospheres (atm)
- Volume must be in liters (L)
- Temperature must be in kelvin (K)
Temperature must always be converted from °C to K using:
\( \mathrm{T(K) = T(^\circ C) + 273} \)
General Problem-Solving Strategy
- Write down the given values and the unknown
- Convert all quantities to the correct units
- Substitute values into \( \mathrm{PV = nRT} \)
- Rearrange the equation to solve for the unknown
- Check units and reasonableness of the answer
Rearranging the Ideal Gas Law
Common rearranged forms include:
\( \mathrm{n = \dfrac{PV}{RT}} \)
\( \mathrm{V = \dfrac{nRT}{P}} \)
\( \mathrm{P = \dfrac{nRT}{V}} \)
Link to Particle-Level Meaning
- Increasing \( \mathrm{n} \) increases pressure if volume and temperature stay constant
- Increasing \( \mathrm{T} \) increases pressure due to faster particle motion
- Increasing \( \mathrm{V} \) lowers pressure by reducing collision frequency
Every calculation reflects changes in particle behavior.
Example
Calculate the volume of 1.50 mol of an ideal gas at a pressure of 1.20 atm and a temperature of 27 °C.
▶️ Answer / Explanation
Step 1: Convert temperature to kelvin.
\( \mathrm{T = 27 + 273 = 300\ K} \)
Step 2: Rearrange the ideal gas law to solve for volume.
\( \mathrm{V = \dfrac{nRT}{P}} \)
Step 3: Substitute values.
\( \mathrm{V = \dfrac{1.50 \times 0.08206 \times 300}{1.20}} \)
\( \mathrm{V = 30.8\ L} \)
Example
A gas sample occupies 10.0 L at 2.50 atm and 127 °C. Calculate the mass of the gas if its molar mass is 28.0 \( \mathrm{g\,mol^{-1}} \).
▶️ Answer / Explanation
Step 1: Convert temperature to kelvin.
\( \mathrm{T = 127 + 273 = 400\ K} \)
Step 2: Calculate moles using the ideal gas law.
\( \mathrm{n = \dfrac{PV}{RT}} \)
\( \mathrm{n = \dfrac{2.50 \times 10.0}{0.08206 \times 400}} \)
\( \mathrm{n = 0.762\ mol} \)
Step 3: Convert moles to mass.
\( \mathrm{m = nM} \)
\( \mathrm{m = 0.762 \times 28.0 = 21.3\ g} \)
3.1.B.3 — Models Based on the Ideal Gas Law
Models based on the ideal gas law are used to explain and predict how macroscopic gas properties—pressure, volume, temperature, and number of particles— are related to particle-level behavior.
These models connect the mathematical relationship \( \mathrm{PV = nRT} \) to how gas particles move, collide, and occupy space.
What a Gas Model Represents
A valid gas model must include both:
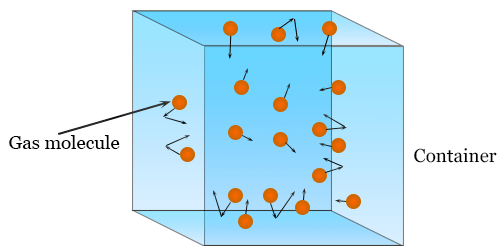
- Macroscopic variables (P, V, T, n)
- Microscopic particle behavior (motion and collisions)
Models may be visual (particle diagrams), graphical, or mathematical.
Particle-Level Assumptions of the Ideal Gas Model
The ideal gas law is based on several simplifying assumptions:
![]()
- Gas particles are very small compared to the container volume
- Particles move in constant, random motion
- Collisions between particles and container walls are elastic
- Attractive forces between particles are negligible
These assumptions allow a simple relationship between macroscopic properties.
Modeling Pressure
In particle models, pressure is represented by collisions of gas particles with the container walls.
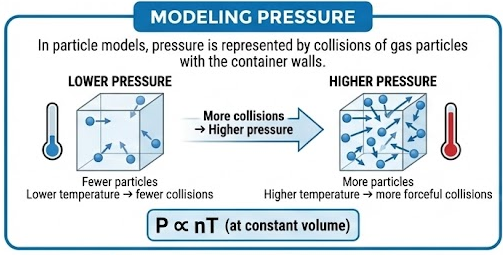
- More particles → more collisions → higher pressure
- Higher temperature → faster particles → more forceful collisions
This explains the relationship:
\( \mathrm{P \propto nT} \) (at constant volume)
Modeling Volume
In models, volume is the space available for particle motion.
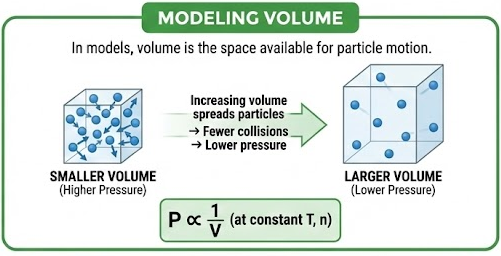
- Increasing volume spreads particles farther apart
- Fewer collisions with container walls → lower pressure
At constant temperature and amount of gas:
\( \mathrm{P \propto \dfrac{1}{V}} \)
Modeling Temperature
Temperature in gas models represents the average kinetic energy of particles.
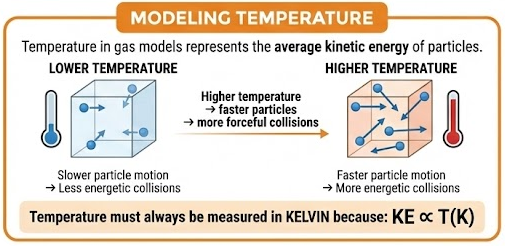
- Higher temperature → faster particle motion
- Faster particles → more energetic collisions
Temperature must always be measured in kelvin because:
\( \mathrm{KE \propto T(K)} \)
Modeling Number of Particles (Moles)
The variable \( \mathrm{n} \) represents how many gas particles are present.
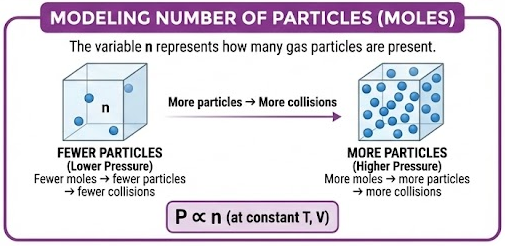
- More moles → more particles
- More particles → more collisions
At constant temperature and volume:
\( \mathrm{P \propto n} \)
Using the Ideal Gas Law as a Mathematical Model
The equation \( \mathrm{PV = nRT} \) serves as a quantitative model linking all four variables.
If three variables are known, the fourth can be predicted.
Evaluating Gas Models
A scientifically accurate gas model must:
- Correctly represent particle motion and spacing
- Show how changes in one variable affect others
- Be consistent with \( \mathrm{PV = nRT} \)
Models that ignore particle behavior or use incorrect relationships are not valid representations of gas behavior.
Comparing Two Gas Models
| Change Made | Correct Model Prediction |
|---|---|
| Increase temperature | Particles move faster; pressure increases |
| Increase volume | Particles farther apart; pressure decreases |
| Add gas particles | More collisions; pressure increases |
Example
A container of gas is heated while the volume remains constant. Describe how a particle model based on the ideal gas law would change.
▶️ Answer / Explanation
Heating the gas increases the temperature, which increases the average kinetic energy of particles.
In the particle model, particles move faster and collide with the container walls more frequently and with greater force, resulting in increased pressure.
Example
Two gas samples are shown in particle diagrams at the same temperature and volume. Sample A shows twice as many particles as Sample B. Use the ideal gas law to evaluate which sample has a higher pressure and explain why.
▶️ Answer / Explanation
Sample A has the higher pressure.
At constant volume and temperature, the ideal gas law shows that pressure is directly proportional to the number of moles. More particles in Sample A result in more frequent collisions with the container walls, producing higher pressure.
