3.2A Chemical Transformations and Conservation of Atoms- Pre AP Chemistry Study Notes - New Syllabus.
3.2A Chemical Transformations and Conservation of Atoms- Pre AP Chemistry Study Notes
3.2A Chemical Transformations and Conservation of Atoms- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
3.2.A.1 Create and/or evaluate models of chemical transformations.
Key Concepts:
- 3.2.A All chemical transformations involve the rearrangement of atoms to form new combinations.
a. Since the atoms are not created or destroyed, the total numbers of each atom must remain constant.
b. Chemical transformations can be modeled by balanced chemical equations and particulate representations.
3.2.A.1 — Models of Chemical Transformations
A chemical transformation (chemical reaction) is a process in which atoms are rearranged to form new substances. Although substances change, the atoms themselves are not created or destroyed.
Because of this, all chemical transformations must obey the law of conservation of matter, and they can be represented using balanced chemical equations and particulate (particle-level) models.
What Happens During a Chemical Transformation
At the particle level:
![]()
- Existing bonds between atoms are broken
- New bonds are formed
- Atoms rearrange into new combinations
The total number of each type of atom remains constant before and after the reaction.
Conservation of Atoms
In every chemical transformation:
- Atoms are not created
- Atoms are not destroyed
- Atoms are rearranged into new substances
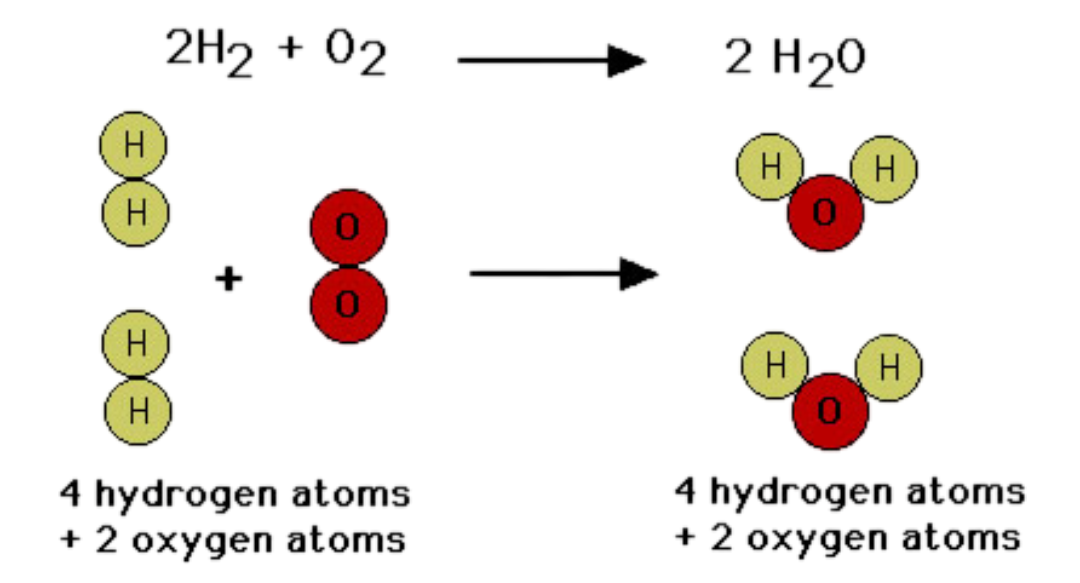
This principle explains why chemical equations must be balanced.
Balanced Chemical Equations as Models
A balanced chemical equation is a symbolic model that shows:
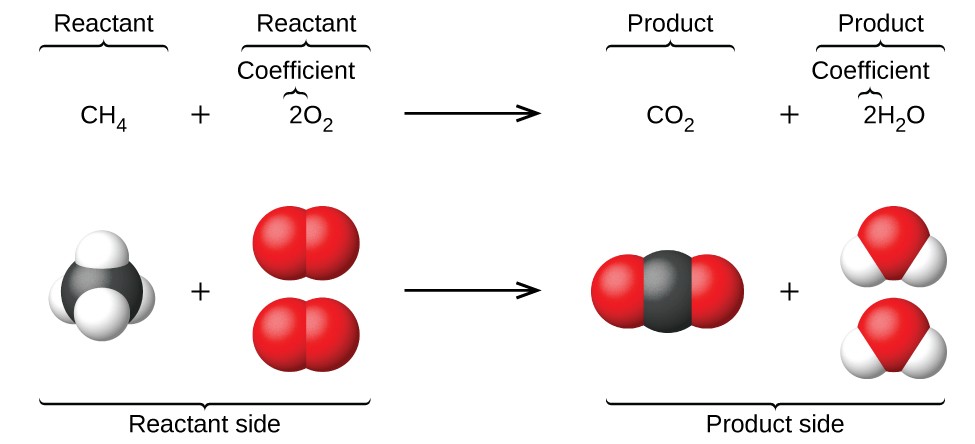
- The reactants (starting substances)
- The products (substances formed)
- The conservation of each type of atom
For example:
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
This equation shows:
- 4 hydrogen atoms on both sides
- 2 oxygen atoms on both sides
Coefficients balance equations by changing the number of particles, not the identity of substances.
Particulate Models of Chemical Transformations
Particulate representations visually model reactions at the atomic level. They show:
- Individual atoms or molecules before the reaction
- How atoms rearrange during the reaction
- New groupings of atoms after the reaction
A correct particulate model must display the same number of each atom before and after the reaction.
Connecting Equation Models and Particle Models
Both representations describe the same transformation:
- Chemical equations show numerical relationships
- Particle models show physical rearrangement
A balanced equation should always match its particulate model.
Evaluating Models of Chemical Transformations
A scientifically valid model must:
- Conserve the number of each type of atom
- Show rearrangement rather than disappearance of atoms
- Correctly match reactants to products
Models that change the total number of atoms violate conservation of matter.
Why Models Are Important
Models help chemists:
- Predict amounts of substances formed or consumed
- Visualize invisible atomic processes
- Check whether reasoning follows physical laws
Example
A reaction is represented by the equation:
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
Explain how this equation models conservation of atoms.
▶️ Answer / Explanation
The equation shows two magnesium atoms and two oxygen atoms on both sides of the reaction.
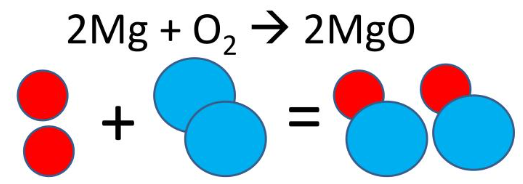
The atoms are rearranged to form magnesium oxide, but no atoms are created or destroyed, demonstrating conservation of matter.
Example
A particulate diagram shows three molecules of hydrogen reacting with one molecule of nitrogen to form two molecules of ammonia.
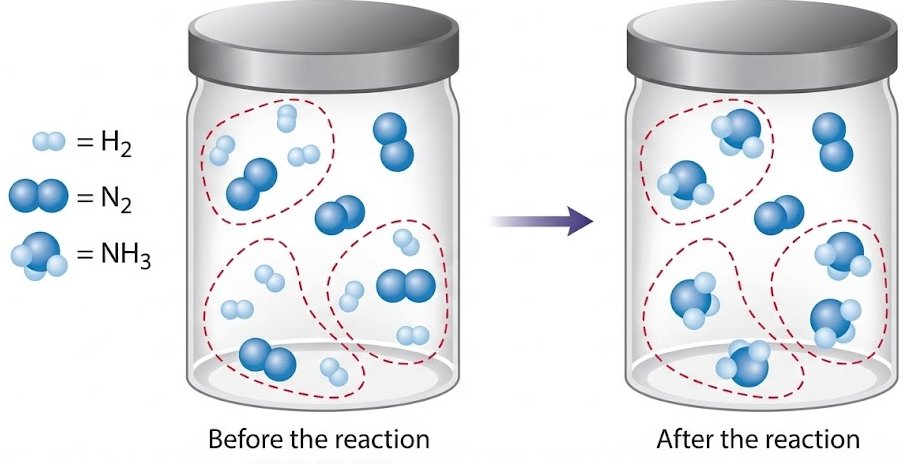
Create the balanced chemical equation and explain how the particulate model supports it.
▶️ Answer / Explanation
The balanced chemical equation is:
\( \mathrm{N_2 + 3H_2 \rightarrow 2NH_3} \)
The particulate model shows two nitrogen atoms and six hydrogen atoms before and after the reaction. The atoms are rearranged to form ammonia molecules, confirming conservation of atoms.
