4.2A Oxidation Numbers and Electron Transfer- Pre AP Chemistry Study Notes - New Syllabus.
4.2A Oxidation Numbers and Electron Transfer- Pre AP Chemistry Study Notes
4.2A Oxidation Numbers and Electron Transfer- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.2.A.1 Identify a reaction as an oxidation–reduction reaction based on the change in oxidation numbers of reacting substances.
4.2.A.2 Create and/or evaluate a claim about which reacting species is oxidized or reduced in an oxidation–reduction reaction.
Key Concepts:
- 4.2.A Electrons are transferred between reactants in oxidation–reduction (redox) reactions.
a. Substances lose electrons in the process of oxidation and gain electrons in the process of reduction.
b. Oxidation numbers are useful for determining if electrons are transferred in a chemical reaction.
c. Electrons are conserved in redox reactions.
4.2.A.1 — Identifying Oxidation–Reduction (Redox) Reactions
An oxidation–reduction (redox) reaction is a chemical reaction in which electrons are transferred between reacting substances. Redox reactions can be identified by examining changes in oxidation numbers of atoms in the reaction.
Because electrons are neither created nor destroyed, electrons are conserved in all redox reactions.
Key Definitions
![]()
![]()
- Oxidation: loss of electrons
- Reduction: gain of electrons
A helpful mnemonic is:
OIL RIG — Oxidation Is Loss, Reduction Is Gain
Oxidation and reduction always occur together—one cannot happen without the other.
Oxidation Numbers
Oxidation numbers are assigned values that represent the hypothetical charge an atom would have if electrons were completely transferred. They are used to track electron movement in reactions.

Basic Oxidation Number Rules (Pre-AP)
- Elements in their elemental form have an oxidation number of \( \mathrm{0} \)
- Group 1 metals have an oxidation number of \( \mathrm{+1} \)
- Group 2 metals have an oxidation number of \( \mathrm{+2} \)
- Oxygen is usually \( \mathrm{-2} \)
- Hydrogen is usually \( \mathrm{+1} \)
- The sum of oxidation numbers equals the total charge of the compound or ion
How Oxidation Numbers Identify Redox Reactions
A reaction is classified as a redox reaction if:
![]()
- At least one atom’s oxidation number increases (oxidation)
- At least one atom’s oxidation number decreases (reduction)
If no oxidation numbers change, the reaction is not a redox reaction.
Oxidation Number Changes and Electrons
Oxidation number changes correspond to electron transfer:
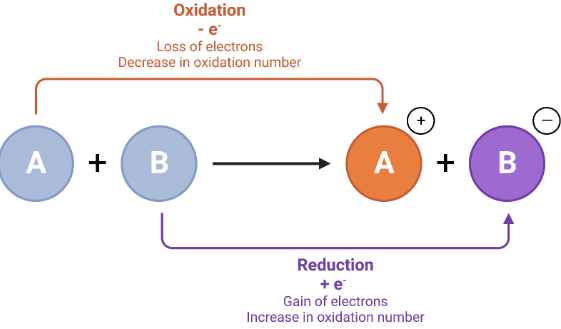
- Increase in oxidation number → loss of electrons
- Decrease in oxidation number → gain of electrons
The number of electrons lost equals the number of electrons gained, demonstrating conservation of electrons.
Electron Conservation in Redox Reactions
In every redox reaction:
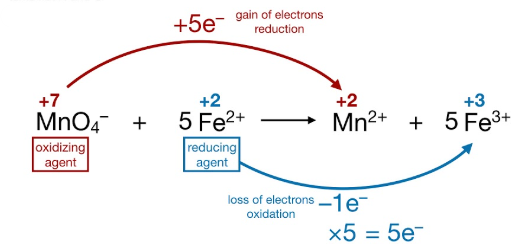
- Total electrons lost = total electrons gained
- Oxidation and reduction occur simultaneously
This principle applies to simple reactions and complex reactions alike.
Evaluating Whether a Reaction Is Redox
To evaluate a reaction:
- Assign oxidation numbers to each element
- Compare oxidation numbers before and after the reaction
- Determine whether oxidation and reduction have occurred
Identifying electron transfer—not reaction type—is the key criterion.
Example
Determine whether the following reaction is a redox reaction:
\( \mathrm{2Mg(s) + O_2(g) \rightarrow 2MgO(s)} \)
▶️ Answer / Explanation
Magnesium changes from \( \mathrm{0} \) to \( \mathrm{+2} \), so magnesium is oxidized.
Oxygen changes from \( \mathrm{0} \) to \( \mathrm{-2} \), so oxygen is reduced.
Because oxidation and reduction both occur, this is a redox reaction.
Example
Identify the species oxidized and reduced in the following reaction:
\( \mathrm{Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)} \)
▶️ Answer / Explanation
Zinc changes from \( \mathrm{0} \) to \( \mathrm{+2} \), so zinc is oxidized (loses electrons).
Copper changes from \( \mathrm{+2} \) to \( \mathrm{0} \), so copper is reduced (gains electrons).
The electrons lost by zinc are gained by copper, confirming conservation of electrons.
4.2.A.2 — Identifying Oxidized and Reduced Species in Redox Reactions
In an oxidation–reduction (redox) reaction, electrons are transferred between reacting species. A scientifically valid claim must clearly identify which species is oxidized and which species is reduced by analyzing oxidation number changes.
Because electrons are conserved, oxidation and reduction always occur together. One species cannot be oxidized unless another is reduced.
Review of Core Definitions
- Oxidation: loss of electrons (oxidation number increases)
- Reduction: gain of electrons (oxidation number decreases)
Mnemonic reminder:
OIL RIG — Oxidation Is Loss, Reduction Is Gain
Oxidized and Reduced Species
To correctly identify species in a redox reaction:
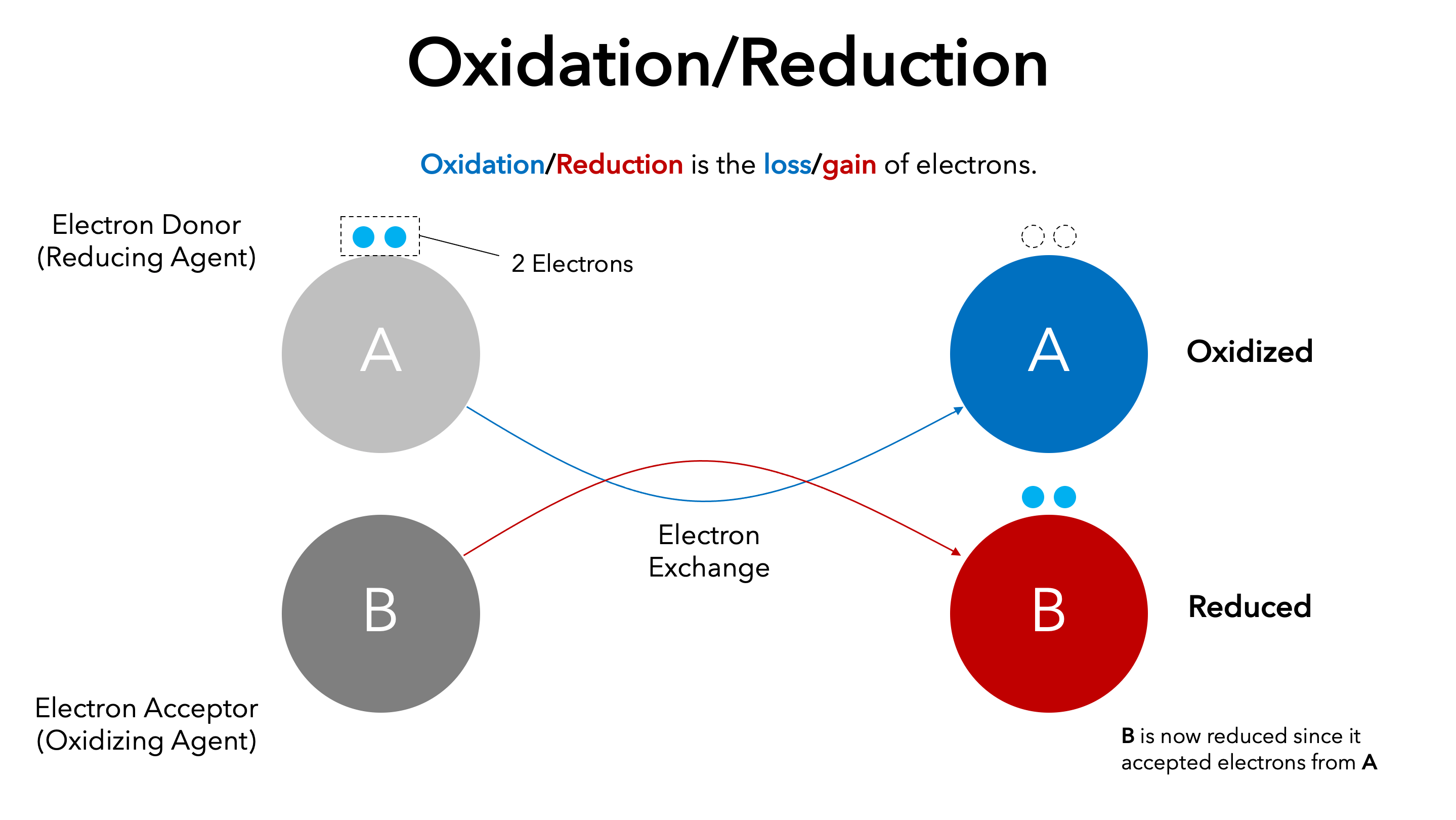
- The oxidized species is the substance that loses electrons
- The reduced species is the substance that gains electrons
This identification is based solely on oxidation number changes, not on reactant order or chemical type.
Oxidizing and Reducing Agents
Redox claims often also identify agents:
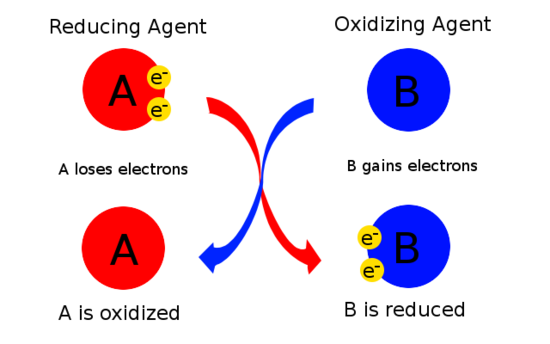
- Reducing agent: causes another species to be reduced by losing electrons; it is itself oxidized
- Oxidizing agent: causes another species to be oxidized by gaining electrons; it is itself reduced
Agents are named based on what they cause, not what happens to them.
Using Oxidation Numbers to Support Claims
A claim about oxidation or reduction must be supported by explicit oxidation number changes.
For example:
![]()
Claims that do not reference oxidation numbers are incomplete.
Evaluating Redox Claims
When evaluating a claim, verify that it:
- Correctly identifies oxidation number changes
- Matches oxidation to electron loss and reduction to electron gain
- Maintains conservation of electrons
A correct claim must identify both species involved.
Particle-Level Interpretation
At the particle level:
- Electrons move from the oxidized species to the reduced species
- The oxidized species becomes more positively charged
- The reduced species becomes less positively charged or more negatively charged
Oxidation numbers are bookkeeping tools that track this movement.
Example
Consider the reaction:
\( \mathrm{2Al(s) + 3Cu^{2+}(aq) \rightarrow 2Al^{3+}(aq) + 3Cu(s)} \)
Create a claim identifying which species is oxidized and which is reduced.
▶️ Answer / Explanation
Aluminum is oxidized because its oxidation number increases from \( \mathrm{0} \) to \( \mathrm{+3} \).
Copper(II) ions are reduced because copper’s oxidation number decreases from \( \mathrm{+2} \) to \( \mathrm{0} \).
The electrons lost by aluminum are gained by copper, conserving electrons.
Example
A student claims that chlorine is oxidized in the following reaction:
\( \mathrm{Cl_2(g) + 2Br^-(aq) \rightarrow 2Cl^-(aq) + Br_2(l)} \)
Evaluate this claim and correct it if necessary.
▶️ Answer / Explanation
The claim is incorrect.
Chlorine changes from \( \mathrm{0} \) to \( \mathrm{-1} \), so chlorine gains electrons and is reduced.
Bromide ions change from \( \mathrm{-1} \) to \( \mathrm{0} \), so bromide is oxidized.
This correction follows oxidation number changes and electron transfer.
