4.2B Activity Series and Redox Reactions- Pre AP Chemistry Study Notes - New Syllabus.
4.2B Activity Series and Redox Reactions- Pre AP Chemistry Study Notes
4.2B Activity Series and Redox Reactions- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.2.B.1 Predict whether a redox reaction will occur between two reactants using an activity series.
4.2.B.2 Create and/or evaluate an activity series from experimental measurements.
Key Concepts:
- 4.2.B An activity series lists elements in order of decreasing ease of oxidation and can be used to determine whether a redox reaction will occur between two species.
4.2.B.1 — Predicting Redox Reactions Using an Activity Series
An activity series is a ranked list of elements ordered by their ease of oxidation. It is used to predict whether a redox reaction will occur between two reacting species, particularly in single-displacement reactions.
Because oxidation involves loss of electrons, elements higher in the activity series are more likely to be oxidized and to cause other species to be reduced.
What the Activity Series Represents
The activity series ranks elements based on:
- Their tendency to lose electrons
- Their reactivity as reducing agents
Elements at the top of the series:
- Are easily oxidized
- Are strong reducing agents
Elements lower in the series:
- Resist oxidation
- Are weaker reducing agents
Typical Metal Activity Series (Top → Bottom)
Most active → lithium, potassium, calcium, sodium, magnesium, aluminum, zinc, iron, copper, silver, gold ← least active
Only the relative position matters for predictions—not the exact order.
How the Activity Series Predicts Redox Reactions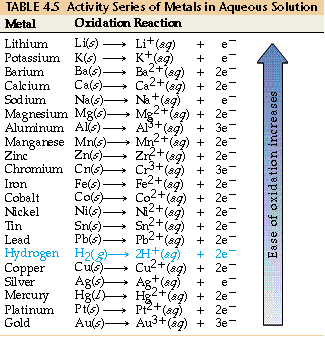
A redox reaction will occur if:
- A more active element is placed in contact with ions of a less active element
The more active element:
- Loses electrons (is oxidized)
- Forces the other species to gain electrons (is reduced)
If the solid metal is below the ion in the activity series, no reaction occurs.
Single-Displacement Redox Reactions
Most activity-series predictions involve reactions of the form:
\( \mathrm{A(s) + B^{n+}(aq) \rightarrow A^{n+}(aq) + B(s)} \)
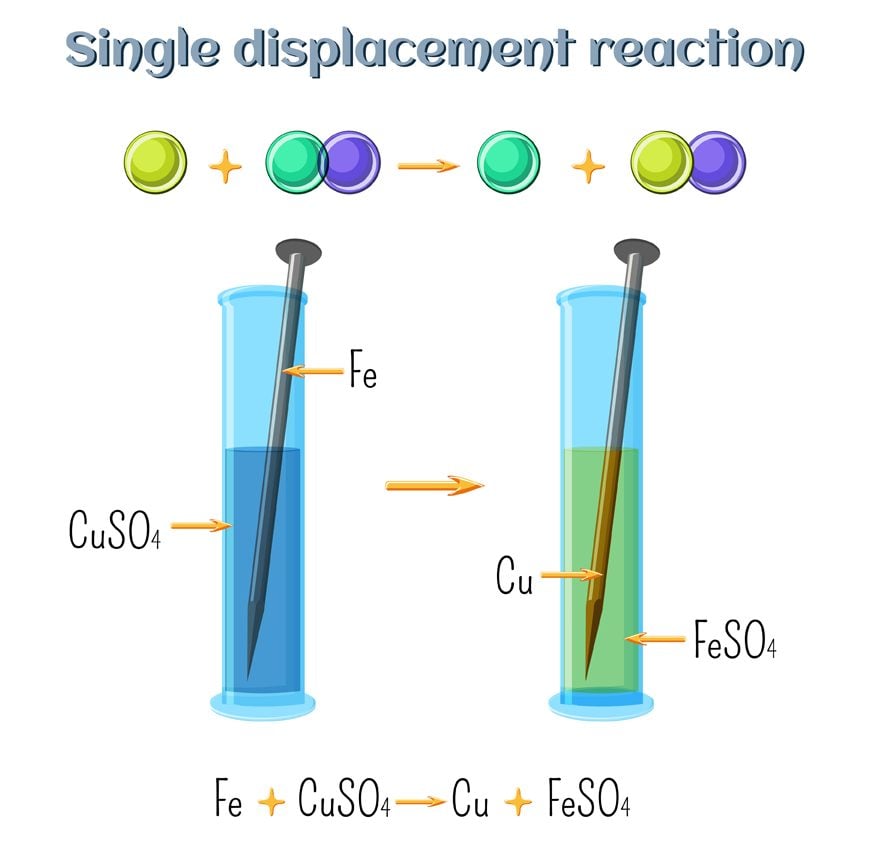
The reaction occurs only if element \( \mathrm{A} \) is higher than \( \mathrm{B} \) in the activity series.
Electron-Transfer Interpretation
At the particle level:
- The more active metal donates electrons
- The less active metal ion accepts electrons
- Electrons are conserved
The activity series predicts whether this electron transfer is energetically favorable.
Evaluating Claims Using the Activity Series
A valid claim must:
- Reference the relative positions of elements in the activity series
- Identify which species would be oxidized and reduced
- Correctly predict whether a reaction occurs
Claims that ignore element position in the series are unsupported.
Example
Predict whether a redox reaction will occur when solid zinc is placed in a solution of copper(II) nitrate.
▶️ Answer / Explanation
Zinc is higher than copper in the activity series.
Therefore, zinc will be oxidized and copper(II) ions will be reduced. A redox reaction occurs.
\( \mathrm{Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)} \)
Example
A student predicts that silver metal will react with iron(II) nitrate. Evaluate this claim using the activity series.
▶️ Answer / Explanation
The claim is incorrect.
Silver is lower than iron in the activity series, so silver is less easily oxidized than iron.
Because silver cannot oxidize iron(II) ions, no redox reaction will occur.
4.2.B.2 — Creating and Evaluating an Activity Series from Experimental Data
An activity series can be constructed by analyzing experimental measurements and observations that show how readily different elements undergo oxidation. Experimental data from redox and single-displacement reactions provide evidence for ranking elements by their relative reactivity.
A valid activity series must be based on observable electron transfer, not assumptions or memorized lists.
Experimental Basis of the Activity Series
An activity series ranks elements according to their ease of oxidation:
- Elements that oxidize easily appear higher
- Elements that resist oxidation appear lower
This ranking is determined experimentally by observing which reactions occur and which do not.
Typical Experiments Used to Generate an Activity Series
1. Single-Displacement Reactions
A solid metal is placed into a solution containing ions of another metal.
\( \mathrm{M(s) + X^{n+}(aq) \rightarrow M^{n+}(aq) + X(s)} \)
If the reaction occurs, the solid metal is more active than the ion.
2. Observable Experimental Evidence
Evidence that a reaction has occurred includes:
- Formation of a solid metal deposit
- Color change in solution
- Temperature change
- Loss of metal mass
These observations indicate electron transfer.
Creating an Activity Series from Experimental Data
To construct an activity series:
- Test each metal against solutions of other metal ions
- Record whether a reaction occurs
- Identify which metal is oxidized in each reaction
- Rank metals from most easily oxidized to least
Metals that displace many others are placed higher in the series.
Interpreting Experimental Results
Experimental conclusions follow this logic:
- If metal A displaces metal B → A is more active than B
- If no reaction occurs → A is less active than B
Each successful displacement adds evidence for relative placement.
Evaluating the Reliability of an Activity Series
When evaluating a student-generated activity series, check whether:
- Conclusions are based on observed reactions
- Electron transfer is clearly identified
- Conflicting data are resolved logically
- All tested reactions are consistent with the final ranking
A single experiment is not sufficient—patterns matter.
Particle-Level Explanation
At the particle level:
- More active metals lose electrons more easily
- Less active metal ions gain electrons more readily
- Electron transfer confirms relative activity
The activity series is a macroscopic summary of microscopic behavior.
Evaluating Claims Based on Experimental Data
A valid claim must:
- Reference specific experimental observations
- Identify oxidation and reduction
- Justify element ranking logically
Claims not tied to data are unsupported.
Example
A student places zinc metal into copper(II) nitrate solution and observes a reddish-brown solid forming on the zinc. No reaction occurs when copper metal is placed into zinc nitrate solution. Use this evidence to compare zinc and copper.
▶️ Answer / Explanation
Zinc displaces copper from solution, showing that zinc is oxidized and copper(II) ions are reduced.
Because copper does not displace zinc, zinc is more active than copper. Zinc should be placed above copper in the activity series.
Example
Experimental data show that:
- Magnesium displaces iron from solution
- Iron displaces copper from solution
- Copper does not displace magnesium
Construct a partial activity series using this data and justify your ranking.
▶️ Answer / Explanation
Magnesium must be the most active because it displaces both iron and copper.
Iron is less active than magnesium but more active than copper, since it displaces copper but not magnesium.
The activity series is:
\( \mathrm{Mg > Fe > Cu} \)
