4.2C Representations of Redox Reactions- Pre AP Chemistry Study Notes - New Syllabus.
4.2C Representations of Redox Reactions- Pre AP Chemistry Study Notes
4.2C Representations of Redox Reactions- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.2.C.1 Create and/or evaluate models of redox reactions.
Key Concepts:
- 4.2.C Redox reactions can be modeled by molecular equations, net ionic equations, and particulate representations.
4.2.C.1 — Modeling Oxidation–Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons between reacting species. Because electron transfer is not always obvious from chemical formulas, redox reactions are best understood using multiple complementary models:
- Molecular equations
- Net ionic equations
- Particulate (particle-level) representations
Each model highlights a different aspect of the same reaction and must be consistent with the others.
Why Redox Reactions Require Multiple Models
Redox reactions involve changes at the electron level, which cannot always be seen directly in symbolic equations. Using multiple models helps:
- Track electron transfer
- Identify oxidized and reduced species
- Connect particle behavior to observable changes
Molecular Equation Model
The molecular equation shows reactants and products as complete substances, including physical states.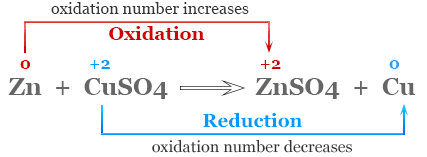
Example:
\( \mathrm{Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s)} \)
This model shows:
- A solid metal reacting with an aqueous ionic compound
- Formation of a new solid metal
- Overall substance-level changes
However, it does not directly show electron movement.
Net Ionic Equation Model
The net ionic equation removes spectator ions and focuses only on the species undergoing oxidation and reduction.
Example:
\( \mathrm{Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)} \)
This model clearly shows:
- Zinc losing electrons (oxidation)
- Copper ions gaining electrons (reduction)
- Direct electron transfer between species
Identifying Oxidation and Reduction in Models
Using oxidation numbers:
- Zinc: \( \mathrm{0 \rightarrow +2} \) → oxidized
- Copper: \( \mathrm{+2 \rightarrow 0} \) → reduced
These changes must be supported by both the molecular and net ionic models.
Particulate (Particle-Level) Models
Particulate models illustrate redox reactions by showing individual particles such as atoms, ions, and electrons.
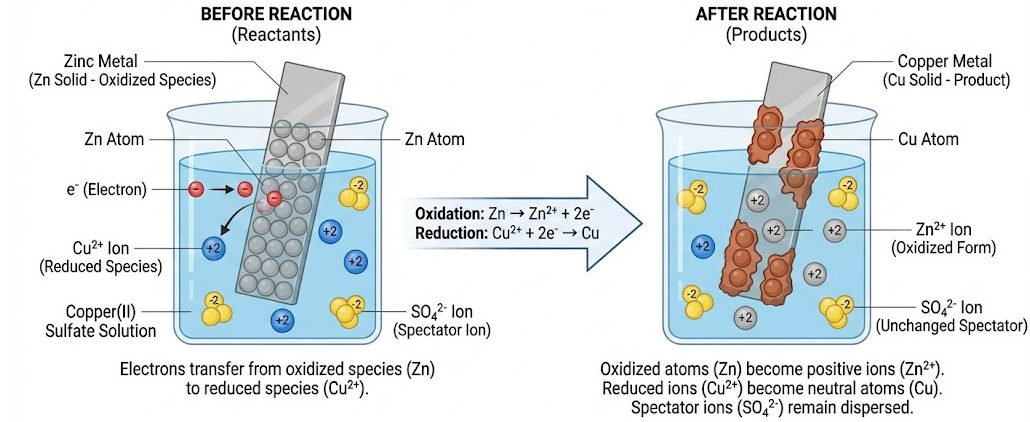
A correct particulate redox model must show:
- Electrons transferring from the oxidized species to the reduced species
- Oxidized atoms becoming positive ions
- Reduced ions becoming neutral atoms
Spectator ions must remain unchanged and dispersed in solution.
Consistency Across Models
All redox models must agree on:
- Which species is oxidized
- Which species is reduced
- Conservation of electrons
If one model contradicts another, the representation is incorrect.
Evaluating Redox Models
To evaluate a redox model, check that:
- Oxidation numbers change appropriately
- Electron loss equals electron gain
- Net ionic equations exclude spectator ions
- Particle diagrams match symbolic equations
Models that only show substances without electron reasoning are incomplete.
Example
Write the net ionic equation for the redox reaction between solid magnesium and aqueous iron(III) chloride, and identify which species is oxidized.
▶️ Answer / Explanation
Molecular reaction:
\( \mathrm{Mg(s) + FeCl_3(aq) \rightarrow MgCl_2(aq) + Fe(s)} \)
Net ionic equation:
\( \mathrm{Mg(s) + Fe^{3+}(aq) \rightarrow Mg^{2+}(aq) + Fe(s)} \)
Magnesium changes from \( \mathrm{0} \) to \( \mathrm{+2} \), so magnesium is oxidized.
Example
A particulate diagram shows electrons leaving aluminum atoms but does not show electrons being gained by any species. Evaluate this model.
▶️ Answer / Explanation
The model is incorrect.
In a redox reaction, electrons must be conserved. If aluminum loses electrons, another species must gain them. The particulate model must include a reduced species receiving the electrons.
