4.3C Acid–Base Reactions- Pre AP Chemistry Study Notes - New Syllabus.
4.3C Acid–Base Reactions- Pre AP Chemistry Study Notes
4.3C Acid–Base Reactions- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.3.C.1 Predict the products of a reaction between a strong acid and a strong base.
Key Concepts:
- 4.3.C Acid–base reactions involve the transfer of a hydrogen ion from the acid to the base. Strong acid–base reactions produce water and an aqueous ionic compound.
4.3.C.1 — Predicting Products of Strong Acid–Strong Base Reactions
A reaction between a strong acid and a strong base is called a neutralization reaction. These reactions involve the transfer of a hydrogen ion from the acid to the base and consistently produce water and an aqueous ionic compound (a salt).
Key Concept: Acid–Base Neutralization
In aqueous solution: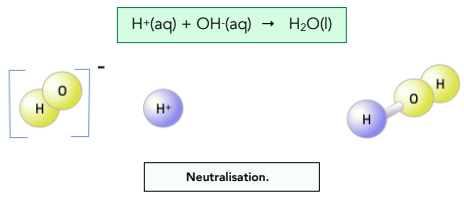
- Strong acids completely produce \( \mathrm{H^+} \)
- Strong bases completely produce \( \mathrm{OH^-} \)
The defining reaction is the combination of these ions:
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
This reaction always forms liquid water.
General Products of Strong Acid–Strong Base Reactions
When a strong acid reacts with a strong base, the products are:
![]()
- Water (\( \mathrm{H_2O(l)} \))
- A soluble ionic compound (salt) in aqueous solution
The salt forms from the spectator ions that do not participate directly in the proton transfer.
Identifying the Salt Formed
To predict the salt:
- Identify the anion from the acid
- Identify the cation from the base
- Combine them to form an ionic compound
The salt remains dissolved because strong acid–strong base reactions typically produce soluble salts.
Molecular, Ionic, and Net Ionic Models
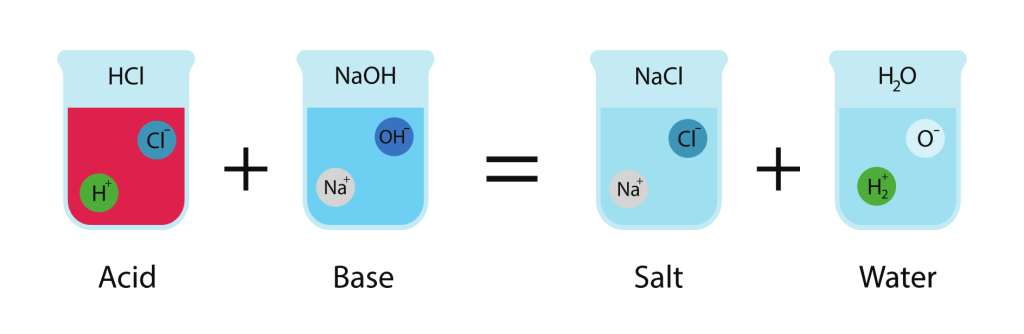
Molecular equation:
\( \mathrm{HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)} \)
Complete ionic equation:
\( \mathrm{H^+(aq) + Cl^-(aq) + Na^+(aq) + OH^-(aq) \rightarrow Na^+(aq) + Cl^-(aq) + H_2O(l)} \)
Net ionic equation:
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
This net ionic equation is the same for all strong acid–strong base reactions.
Particle-Level Interpretation
At the particle level:
- \( \mathrm{H^+} \) ions collide with \( \mathrm{OH^-} \) ions
- A water molecule forms
- Spectator ions remain dispersed in solution
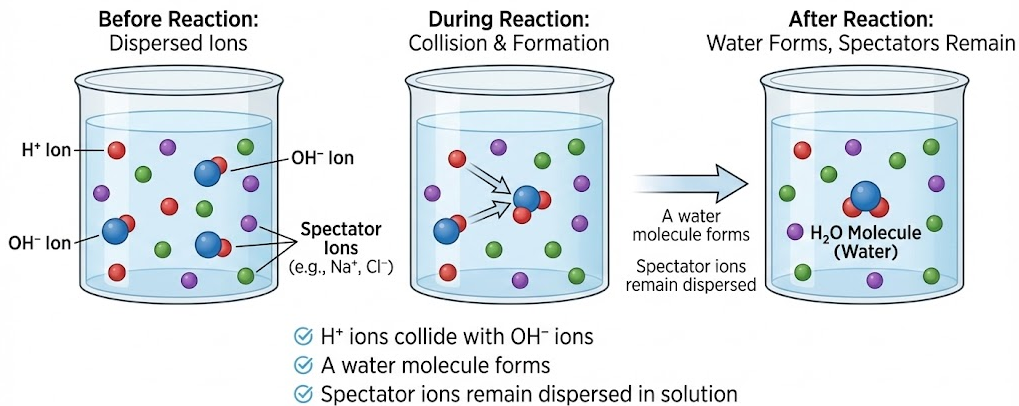
A correct particulate model must show water molecules forming and unchanged spectator ions.
Evaluating Claims About Reaction Products
A correct claim must state that:
- A hydrogen ion is transferred from acid to base
- Water is produced
- An aqueous ionic compound (salt) forms
Claims that predict gas formation or precipitates are incorrect for strong acid–strong base reactions.
Example
Predict the products of the reaction between nitric acid and potassium hydroxide.
▶️ Answer / Explanation
Nitric acid provides \( \mathrm{NO_3^-} \) and potassium hydroxide provides \( \mathrm{K^+} \).
Products:
\( \mathrm{KNO_3(aq) + H_2O(l)} \)
This is a strong acid–strong base neutralization reaction.
Example
A student predicts that the reaction between hydrochloric acid and sodium hydroxide produces sodium chloride gas and water. Evaluate this claim.
▶️ Answer / Explanation
The claim is incorrect.
Sodium chloride is an ionic compound that remains dissolved in water, not a gas.
The correct products are aqueous sodium chloride and liquid water, formed by transfer of a hydrogen ion from the acid to the base.
