4.4B Bond Energies and Enthalpy Changes- Pre AP Chemistry Study Notes - New Syllabus.
4.4B Bond Energies and Enthalpy Changes- Pre AP Chemistry Study Notes
4.4B Bond Energies and Enthalpy Changes- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.4.B.1 Create and/or evaluate a claim about the energy transferred as a result of a chemical reaction based on bond energies.
Key Concepts:
- 4.4.B The relative strength of bonds in reactants and products determines the energy change in a reaction. Bond energy tables and Lewis diagrams provide a way to estimate these changes quantitatively for a wide variety of chemical reactions.
4.4.B.1 — Energy Changes in Reactions Using Bond Energies
The energy transferred during a chemical reaction depends on the relative strengths of bonds in the reactants and products. By comparing the energy required to break bonds with the energy released when new bonds form, we can determine whether a reaction is endothermic or exothermic.
Bond energy tables and Lewis diagrams provide a quantitative way to estimate these energy changes.
Key Principles of Bond Energies
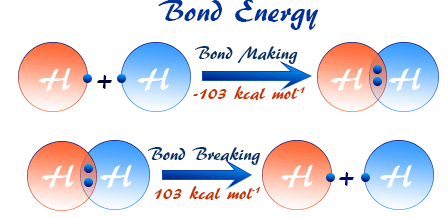
- Bond energy is the energy required to break a bond
- Stronger bonds have higher bond energies
- Breaking bonds is always endothermic
- Forming bonds is always exothermic
The overall energy change depends on which process dominates.
Using Lewis Diagrams in Bond Energy Analysis
Lewis diagrams help identify:
![]()
- Which bonds are present in the reactants
- Which bonds are present in the products
- Which bonds must be broken and formed
Accurate bond-energy calculations require correct Lewis structures.
Calculating Energy Change Using Bond Energies
The estimated energy change of a reaction is calculated using:
\( \mathrm{\Delta E = \sum E_{broken} – \sum E_{formed}} \)
- \( \mathrm{\sum E_{broken}} \) = total energy absorbed breaking bonds
- \( \mathrm{\sum E_{formed}} \) = total energy released forming bonds
This calculation estimates the reaction’s overall energy transfer.
Interpreting the Result
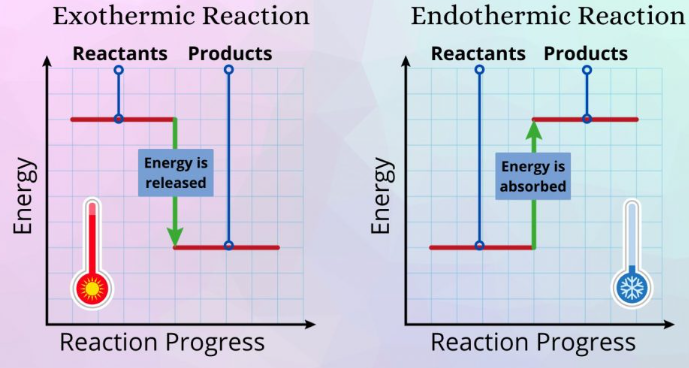
- \( \mathrm{\Delta E < 0} \): more energy released → exothermic
- \( \mathrm{\Delta E > 0} \): more energy absorbed → endothermic
The sign of \( \mathrm{\Delta E} \) directly reflects energy transfer.
Bond Strength Comparison Logic
A reaction tends to be exothermic when:
- Strong bonds form in the products
- Weaker bonds are broken in the reactants
A reaction tends to be endothermic when:
- Strong bonds must be broken
- Weaker bonds are formed
Evaluating Claims Using Bond Energy Data
A valid claim must:
- Identify bonds broken and bonds formed
- Reference relative bond strengths or bond energy values
- Correctly predict the direction of energy transfer
Claims without bond-level justification are incomplete.
Example
Consider a reaction in which weak single bonds in the reactants are replaced by stronger double bonds in the products. Create a claim about the energy change of the reaction.
▶️ Answer / Explanation
The reaction is exothermic.
Energy absorbed breaking the weaker bonds is less than the energy released when stronger bonds form in the products. Therefore, energy is released overall.
Example
Using bond energy data, a student calculates that \( \mathrm{1800\ kJ} \) are required to break bonds in the reactants and \( \mathrm{2100\ kJ} \) are released forming bonds in the products. Evaluate the energy change of the reaction.
▶️ Answer / Explanation
The energy change is:
\( \mathrm{\Delta E = 1800 – 2100 = -300\ kJ} \)
The negative value shows that the reaction releases energy and is exothermic. The stronger bonds formed in the products release more energy than is absorbed breaking reactant bonds.
