4.4A Energy Changes in Chemical Reactions- Pre AP Chemistry Study Notes - New Syllabus.
4.4A Energy Changes in Chemical Reactions- Pre AP Chemistry Study Notes
4.4A Energy Changes in Chemical Reactions- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.4.A.1 Create and/or evaluate a claim about whether a reaction is endothermic or exothermic from experimental observations.
4.4.A.2 Explain the relationship between the measured change in temperature of a solution and the energy transferred by a chemical reaction.
4.4.A.3 Calculate energy changes in chemical reactions from calorimetry data.
Key Concepts:
- 4.4.A A temperature change during a reaction is the result of energy transfer during the process of breaking and forming bonds.
a. Bond breaking is always an endothermic process and bond formation is always an exothermic process.
b. Calorimetry can be used to quantify energy changes in a reaction.
4.4.A.1 — Identifying Endothermic and Exothermic Reactions from Experimental Observations
A reaction can be classified as endothermic or exothermic by analyzing experimental observations, especially temperature changes in the surroundings. These temperature changes result from energy transfer during bond breaking and bond formation.
Understanding this standard requires connecting macroscopic observations to particle-level energy changes.
Core Definitions
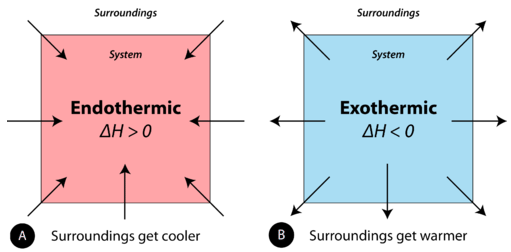
- Endothermic reaction: absorbs energy from the surroundings
- Exothermic reaction: releases energy to the surroundings
Energy is transferred because chemical bonds are broken and formed during a reaction.
Bond Energy and Energy Transfer
At the particle level:
![]()
- Bond breaking always requires energy → endothermic process
- Bond formation always releases energy → exothermic process
The overall energy change of a reaction depends on the relative amounts of energy absorbed and released.
Linking Temperature Change to Reaction Type
Experimental observations often involve monitoring temperature.
Observed temperature increase:
- Energy is released to surroundings
- Bond formation releases more energy than bond breaking absorbs
- The reaction is exothermic
Observed temperature decrease:
- Energy is absorbed from surroundings
- Bond breaking absorbs more energy than bond formation releases
- The reaction is endothermic
Energy Flow Between System and Surroundings
In chemical reactions:
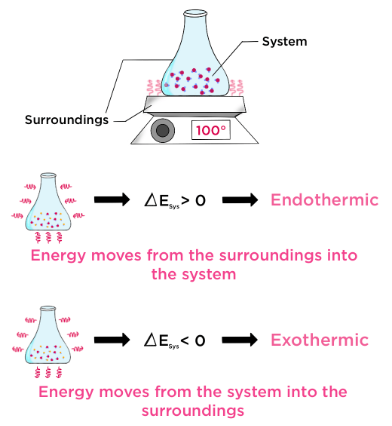
- System = the reacting chemicals
- Surroundings = everything else (solution, container, air)
Temperature changes are measured in the surroundings, not directly in the system.
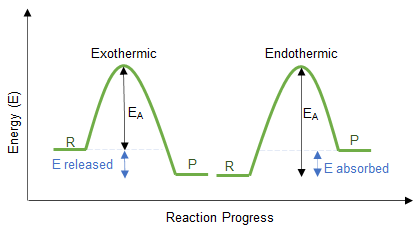
Energy Profile (Potential Energy) Diagrams
Energy profile diagrams model energy changes during reactions:
 F
F
- Endothermic: products have higher energy than reactants
- Exothermic: products have lower energy than reactants
These diagrams visually reinforce the temperature observations.
Evaluating Claims Using Experimental Data
A strong claim about reaction type must:
- Reference measured temperature change
- Identify direction of energy transfer
- Explain bond breaking and bond formation
Claims stating only “temperature changed” without explanation are incomplete.
Common Experimental Setups
- Chemical reactions in insulated cups (calorimetry)
- Dissolving salts in water
- Combustion reactions
All rely on temperature as evidence of energy transfer.
Example
During a reaction, the temperature of the surrounding solution increases from \( \mathrm{22.0^\circ C} \) to \( \mathrm{28.5^\circ C} \). Create a claim identifying whether the reaction is endothermic or exothermic.
▶️ Answer / Explanation
The reaction is exothermic.
The increase in temperature shows that energy was released from the system to the surroundings. This means more energy was released during bond formation than absorbed during bond breaking.
Example
A student observes that a reaction mixture becomes cold to the touch. However, several new bonds are formed in the products. Evaluate whether the reaction can still be endothermic.
▶️ Answer / Explanation
Yes, the reaction can still be endothermic.
Although bond formation releases energy, bond breaking always requires energy. If more energy is absorbed breaking bonds than is released forming new bonds, the overall reaction absorbs energy and is endothermic, causing the surroundings to cool.
4.4.A.2 — Temperature Change and Energy Transfer in Chemical Reactions
The measured change in temperature of a solution during a chemical reaction is direct evidence of energy transfer between the reacting chemicals (the system) and the surrounding solution (the surroundings). This relationship is explained using calorimetry and particle-level reasoning.
System and Surroundings
- System → the chemical reaction occurring
- Surroundings → the solution, container, and thermometer
Temperature is measured in the surroundings, not directly in the reacting bonds. The temperature change reflects how much energy is transferred between them.
Why Temperature Changes
During a reaction:
- Energy is absorbed to break bonds (endothermic step)
- Energy is released when new bonds form (exothermic step)
The balance between these two processes determines whether energy flows into or out of the solution.
Interpreting Temperature Change
Temperature increase (\( \mathrm{\Delta T > 0} \)):
- Energy released from the system
- Energy flows into the solution
- Reaction is exothermic
Temperature decrease (\( \mathrm{\Delta T < 0} \)):
- Energy absorbed by the system
- Energy flows out of the solution
- Reaction is endothermic
Quantifying Energy Transfer (Calorimetry)
The energy transferred to or from the solution is calculated using:![]()
\( \mathrm{q = mc\Delta T} \)
- \( \mathrm{q} \) = energy transferred (\( \mathrm{J} \))
- \( \mathrm{m} \) = mass of solution (\( \mathrm{g} \))
- \( \mathrm{c} \) = specific heat capacity (\( \mathrm{J\,g^{-1}\,^\circ C^{-1}} \))
- \( \mathrm{\Delta T} \) = temperature change (\( \mathrm{^\circ C} \))
This equation links a measured temperature change to the amount of energy transferred.
Relationship Between Solution Energy and Reaction Energy
Energy conservation requires that:
![]()
\( \mathrm{q_{reaction} = -q_{solution}} \)
- If the solution gains energy, the reaction loses energy
- If the solution loses energy, the reaction gains energy
This inverse relationship explains why solution temperature reveals reaction energetics.
Particle-Level Explanation
At the particle level:
- Higher temperature → particles move faster
- Lower temperature → particles move slower
Energy released from bond formation increases particle kinetic energy, raising temperature. Energy absorbed for bond breaking decreases particle kinetic energy, lowering temperature.
Evaluating Experimental Claims
A valid explanation must:
- Reference the observed temperature change
- Describe the direction of energy transfer
- Link macroscopic data to bond-level processes
Stating only that “temperature changed” is insufficient.
Example
During a reaction, the temperature of a solution decreases by \( \mathrm{4.0^\circ C} \). Explain what this indicates about energy transfer.
▶️ Answer / Explanation
The decrease in temperature shows that energy was absorbed from the solution by the reacting chemicals.
This means more energy was required to break bonds than was released during bond formation, so the reaction is endothermic.
Example
A reaction causes a solution’s temperature to rise. However, the calculated value of \( \mathrm{q_{reaction}} \) is negative. Explain why this is consistent.
▶️ Answer / Explanation
The solution gaining energy results in a temperature increase, so \( \mathrm{q_{solution}} \) is positive.
Because energy is conserved, \( \mathrm{q_{reaction} = -q_{solution}} \), making \( \mathrm{q_{reaction}} \) negative.
This confirms the reaction released energy and is exothermic.
4.4.A.3 — Calculating Energy Changes Using Calorimetry
Calorimetry is used to quantify the energy change in a chemical reaction by measuring the temperature change of the surroundings, usually a solution. The measured temperature change is then used to calculate the amount of energy transferred during the reaction.
![]()
This standard requires connecting experimental data, mathematical calculations, and energy conservation.
Key Principle: Energy Conservation
In calorimetry, energy is conserved:
\( \mathrm{q_{reaction} = -q_{solution}} \)
- If the solution gains energy, the reaction loses energy
- If the solution loses energy, the reaction gains energy
This relationship allows us to calculate the energy change of the reaction without measuring it directly.
Calorimetry Equation
The energy change of the solution is calculated using:
\( \mathrm{q = mc\Delta T} \)
- \( \mathrm{q} \) = energy transferred (\( \mathrm{J} \))
- \( \mathrm{m} \) = mass of solution (\( \mathrm{g} \))
- \( \mathrm{c} \) = specific heat capacity (\( \mathrm{J\,g^{-1}\,^\circ C^{-1}} \))
- \( \mathrm{\Delta T = T_{final} – T_{initial}} \)
For aqueous solutions, the specific heat capacity is often approximated as:
\( \mathrm{c = 4.18\ J\,g^{-1}\,^\circ C^{-1}} \)
Step-by-Step Strategy for Calorimetry Calculations
- Determine the temperature change \( \mathrm{\Delta T} \)
- Calculate the energy change of the solution using \( \mathrm{q = mc\Delta T} \)
- Use energy conservation to find \( \mathrm{q_{reaction}} \)
- Assign the correct sign to indicate endothermic or exothermic
Sign Conventions
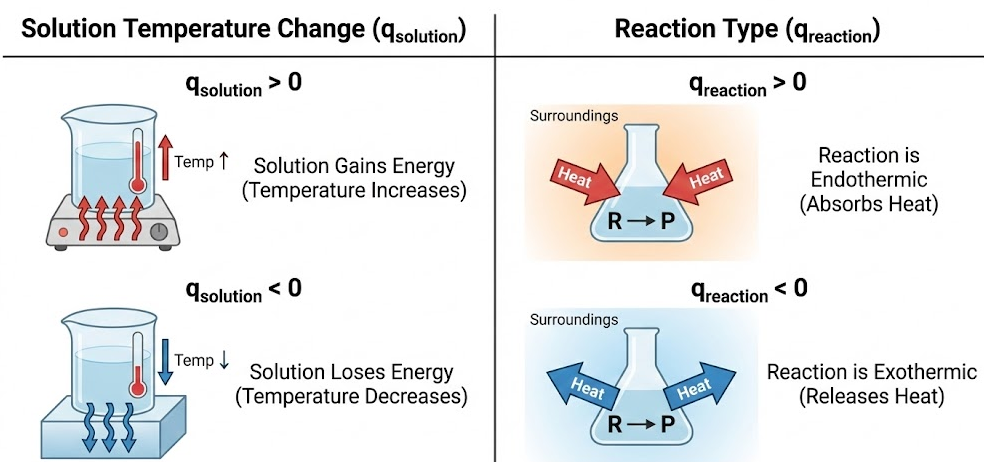
- \( \mathrm{q_{solution} > 0} \): solution gains energy (temperature increases)
- \( \mathrm{q_{solution} < 0} \): solution loses energy (temperature decreases)
- \( \mathrm{q_{reaction} > 0} \): reaction is endothermic
- \( \mathrm{q_{reaction} < 0} \): reaction is exothermic
Correct sign usage is essential for full credit.
Determining the Mass of the Solution
In most Pre-AP problems:
- The mass of the solution is approximated using total volume
- \( \mathrm{1\ mL \approx 1\ g} \) for aqueous solutions
For example, \( \mathrm{100\ mL} \) of solution is treated as \( \mathrm{100\ g} \).
Evaluating Calorimetry Results
A correct calorimetry calculation must:
- Use correct units throughout
- Include the correct sign for \( \mathrm{q} \)
- Explain the physical meaning of the result
Numerical answers without interpretation are incomplete.
Example
A reaction causes \( \mathrm{100\ g} \) of solution to increase in temperature from \( \mathrm{20.0^\circ C} \) to \( \mathrm{25.0^\circ C} \). Calculate the energy transferred by the reaction.
▶️ Answer / Explanation
Step 1: Calculate temperature change:
\( \mathrm{\Delta T = 25.0 – 20.0 = 5.0^\circ C} \)
Step 2: Calculate energy change of solution:
\( \mathrm{q_{solution} = mc\Delta T} \)
\( \mathrm{q_{solution} = 100 \times 4.18 \times 5.0 = 2090\ J} \)
Step 3: Determine energy change of reaction:
\( \mathrm{q_{reaction} = -2090\ J} \)
The reaction is exothermic.
Example
Two solutions are mixed, producing a total mass of \( \mathrm{150\ g} \). The temperature decreases from \( \mathrm{24.0^\circ C} \) to \( \mathrm{20.5^\circ C} \). Calculate \( \mathrm{q_{reaction}} \) and classify the reaction.
▶️ Answer / Explanation
Step 1: Calculate temperature change:
\( \mathrm{\Delta T = 20.5 – 24.0 = -3.5^\circ C} \)
Step 2: Calculate energy change of solution:
\( \mathrm{q_{solution} = 150 \times 4.18 \times (-3.5)} \)
\( \mathrm{q_{solution} = -2195\ J} \)
Step 3: Apply energy conservation:
\( \mathrm{q_{reaction} = +2195\ J} \)
The reaction is endothermic.
