4.5A Reaction Rates and Factors Affecting it- Pre AP Chemistry Study Notes - New Syllabus.
4.5A Reaction Rates and Factors Affecting it- Pre AP Chemistry Study Notes
4.5A Reaction Rates and Factors Affecting it- Pre AP Chemistry Study Notes – New Syllabus.
LEARNING OBJECTIVE
4.5.A.1 Construct and/or evaluate particulate representations that illustrate how changes in concentration, temperature, or surface area of reactants alter the rate of a chemical reaction.
4.5.A.2 Explain how experimental changes in the rate of reaction are related to changes in the concentration, temperature, or surface area of the reactants.
Key Concepts:
- 4.5.A The rate of a chemical reaction can be measured by determining how quickly reactants are transformed into products.
a. The reaction rate is related to the frequency of collisions between reactant species and the proportion of effective collisions.
b. The frequency of collisions increases with the concentration of gases or dissolved species and with the surface area of a solid.
c. The proportion of effective collisions increases directly as temperature increases.
4.5.A.1 — Reaction Rate and Particle-Level Models
The rate of a chemical reaction describes how quickly reactants are converted into products. Reaction rate can be understood and explained by using particulate (particle-level) representations that focus on collisions between reactant particles.
Changes in concentration, temperature, and surface area affect reaction rate by changing the frequency of collisions and the proportion of effective collisions.
Collision Theory: The Foundation of Reaction Rate
According to collision theory:
- Reactant particles must collide to react
- Not all collisions result in a reaction
- Only effective collisions lead to product formation

Therefore, the reaction rate depends on:
- The frequency of collisions
- The proportion of effective collisions
Effective vs Ineffective Collisions
For a collision to be effective, particles must:
- Collide with sufficient energy
- Collide with the correct orientation
Collisions lacking enough energy or proper orientation are ineffective and do not produce products.
Modeling the Effect of Concentration
Increasing concentration increases the number of reactant particles in a given volume.
![]()
Particle-level interpretation:
- More particles in the same space
- More frequent collisions
- Higher reaction rate
In a particulate model, higher concentration is shown by more particles packed into the same volume.
Modeling the Effect of Temperature
Increasing temperature increases the kinetic energy of particles.
![]()
Particle-level interpretation:
- Particles move faster
- Collisions occur more often
- A larger fraction of collisions have enough energy to react
Temperature primarily affects the proportion of effective collisions, not just collision frequency.
Modeling the Effect of Surface Area
Surface area applies mainly to reactions involving solids.
![]()
Particle-level interpretation:
- Smaller pieces expose more surface particles
- More particles are available to collide
- Collision frequency increases
In particulate models:
- A powdered solid reacts faster than a single large chunk
- The total number of particles stays the same, but exposure changes
Measuring Reaction Rate
Reaction rate can be measured by:
![]()
- Change in concentration of reactants over time
- Change in concentration of products over time
- Volume of gas produced per unit time
- Mass of solid consumed or produced over time
All measurements reflect how quickly particles are transformed.
What Correct Particulate Models Must Show
A scientifically accurate particulate representation must:
- Clearly show relative particle numbers (concentration)
- Indicate particle speed with motion lines or spacing (temperature)
- Show exposed solid particles (surface area)
Models that only label conditions without showing particle behavior are incomplete.
Evaluating Claims About Reaction Rate
A valid claim must:
- Identify the variable changed
- Explain how collisions are affected
- Link particle behavior to reaction rate
Claims based only on memorized rules are insufficient without particle reasoning.
Example
Two diagrams show the same reaction mixture. One diagram contains twice as many reactant particles in the same volume. Compare the reaction rates.
▶️ Answer / Explanation
The mixture with more particles has the faster reaction rate.
Higher concentration results in more frequent collisions between reactant particles, increasing the rate of reaction.
Example
A student claims that increasing temperature always increases reaction rate because particles collide more often. Evaluate and improve this claim using a particulate explanation.
▶️ Answer / Explanation
The claim is incomplete.
Increasing temperature does increase collision frequency, but more importantly it increases the proportion of collisions that have enough energy to overcome the energy barrier.
A correct particulate model must show faster-moving particles and a greater number of effective collisions.
4.5.A.2 — Explaining Experimental Changes in Reaction Rate
Experimental observations of reaction rate can be explained by examining how changes in concentration, temperature, or surface area affect particle collisions. Using collision theory, macroscopic data from experiments can be directly linked to particle-level behavior.
This standard focuses on explaining why the rate changes, not merely stating that it changes.
Connecting Experiments to Collision Theory
In laboratory investigations, reaction rate is commonly determined by:
- Time taken for a visible change to occur (gas formation, precipitate, color change)
- Change in concentration of reactants or products over time
- Volume of gas produced or mass change per unit time
Each of these measurements reflects how frequently effective collisions between reactant particles occur.
Effect of Concentration: Explaining Experimental Results
When experimental data show that a reaction occurs faster at higher concentration, the explanation is:
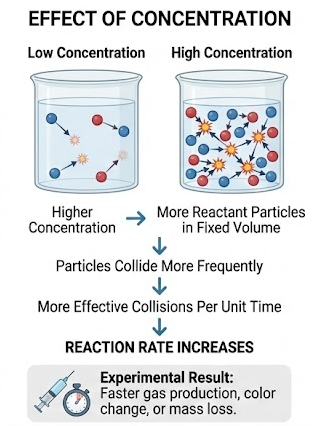
- Higher concentration means more reactant particles in a fixed volume
- Particles collide more frequently
- More collisions per unit time result in more effective collisions
- The reaction rate increases
When concentration is decreased, collision frequency decreases, and the reaction rate slows.
Effect of Surface Area: Explaining Experimental Results
For reactions involving solids, experiments often show faster reaction rates when the solid is crushed or powdered.

This occurs because:
- Breaking a solid into smaller pieces exposes more surface particles
- More particles are available to collide with other reactants
- Collision frequency at the solid surface increases
- The reaction rate increases
The total amount of solid remains the same; only the number of exposed particles changes.
Effect of Temperature: Explaining Experimental Results
Temperature has a strong effect on reaction rate because it changes the kinetic energy of particles.
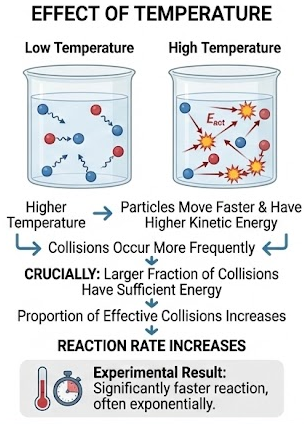
When temperature increases:
- Particles move faster
- Collisions occur more frequently
- A larger fraction of collisions have sufficient energy to react
- The proportion of effective collisions increases
Unlike concentration and surface area, temperature primarily increases the effectiveness of collisions rather than just their frequency.
Distinguishing Collision Frequency and Collision Effectiveness
A complete explanation must clearly distinguish between:
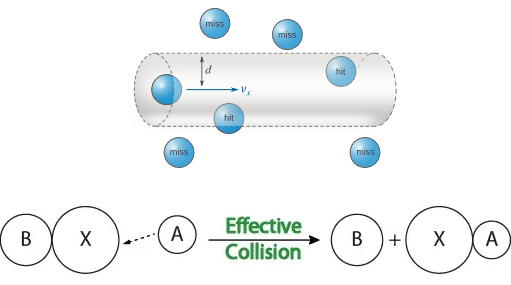
- Collision frequency — how often particles collide (mainly affected by concentration and surface area)
- Collision effectiveness — how many collisions lead to reaction (mainly affected by temperature)
Strong explanations address both concepts explicitly.
Evaluating Experimental Claims About Rate Changes
When evaluating a claim based on experimental evidence, verify that the explanation:
- Clearly identifies which variable was changed
- Correctly links that variable to particle motion or availability
- Explains how collisions are affected
- Uses collision theory rather than memorized rules
Claims that only state “the rate increased” without explanation are incomplete.
Example
An experiment shows that doubling the concentration of a reactant halves the time required for a reaction to complete. Explain this observation using collision theory.
▶️ Answer / Explanation
Doubling the concentration increases the number of reactant particles in the same volume.
This increases the frequency of collisions, resulting in more effective collisions per unit time and a faster reaction rate.
Example
A reaction rate increases when temperature is raised, but the concentration and surface area remain constant. Explain why the rate increases using particle-level reasoning.
▶️ Answer / Explanation
Increasing temperature increases the kinetic energy of particles.
Although collision frequency increases slightly, the main effect is that a greater proportion of collisions have enough energy to overcome the energy barrier.
As a result, more collisions are effective, leading to an increased reaction rate.
