Pre AP Chemistry -1.1B Measurement Precision and Experimental Data- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -1.1B Measurement Precision and Experimental Data- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -1.1B Measurement Precision and Experimental Data- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
A student mixes \(25 \mathrm{~cm}^3\) samples of dilute hydrochloric acid with different volumes of aqueous sodium hydroxide.
In each case, the student measures the change in temperature to test if the reaction is exothermic.
Which piece of apparatus is not needed?
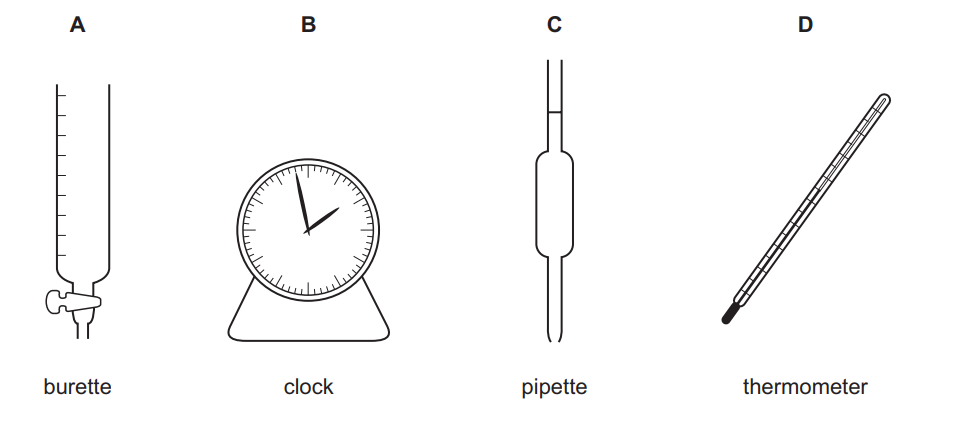
▶️ Answer/Explanation
Ans: B
To measure the temperature change in an exothermic reaction, the following apparatus are needed:
- Burette/Pipette: To accurately measure volumes of acid and alkali.
- Thermometer: To record the temperature change.
A clock is unnecessary because the reaction is instantaneous, and temperature change does not require time measurement. Thus, the correct answer is B (clock).
A thermocouple is used to measure temperature.
Which is an advantage of using a thermocouple instead of a liquid-in-glass thermometer?
A. It can measure temperature in the range –10 °C to 110 °C.
B. It can measure temperature that changes rapidly.
C. It has a linear scale.
D. It is more sensitive.
▶️ Answer/Explanation
Step 1: Understand how a thermocouple works
A thermocouple produces a voltage when there is a temperature difference between two junctions. It responds very quickly to temperature changes.
Step 2: Compare with liquid-in-glass thermometer
A liquid-in-glass thermometer relies on expansion of liquid, which takes time to respond to temperature changes.
Step 3: Evaluate options
Option A is incorrect because both instruments can measure within that range.
Option C is not a special advantage of thermocouples.
Option D is not necessarily true for all thermocouples.
The key advantage is fast response to rapidly changing temperatures.
Ans: B (It can measure temperature that changes rapidly.)
To mark a temperature scale on a thermometer, standard temperatures known as fixed points are needed.
Which of these is a fixed point on the Celsius scale?
A. Room temperature
B. The temperature inside a freezer
C. The temperature of pure melting ice
D. The temperature of pure warm water
▶️ Answer/Explanation
Step 1: Understand fixed points
Fixed points are standard, reproducible temperatures used to calibrate a thermometer.
Step 2: Fixed points on the Celsius scale
The Celsius scale is defined using:
- 0°C → Temperature of pure melting ice
- 100°C → Temperature of pure boiling water (at standard atmospheric pressure)
Step 3: Evaluate options
Room temperature and freezer temperature are not fixed because they vary.
Pure warm water is not a defined standard temperature.
The temperature of pure melting ice is a fixed point (0°C).
Ans: C (The temperature of pure melting ice)
A student measures the volumes of three liquids using three different measuring cylinders.
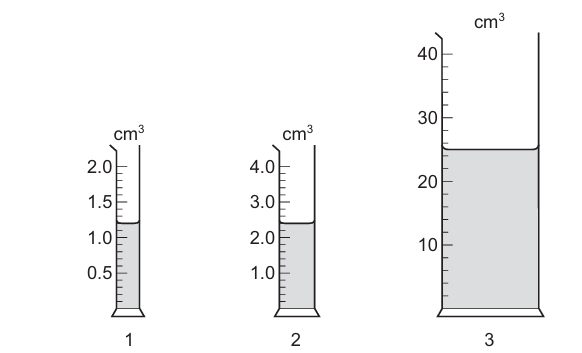
The table shows the volumes recorded by the student.
Which readings are correctly recorded?
A. 1, 2 and 3
B. 1 and 2 only
C. 1 and 3 only
D. 1 only
▶️ Answer/Explanation
Step 1: Check reading precision
A recorded measurement must match the smallest scale division of the measuring cylinder used.
Reading 1: Correctly recorded to the nearest scale division → Correct.
Reading 2: Not recorded to the correct precision (either too many or too few significant figures) → Incorrect.
Reading 3: Correctly matches the scale precision of the measuring cylinder → Correct.
Conclusion:
Readings 1 and 3 are correctly recorded.
Ans: C
A measuring cylinder contains water.
The diagrams show the measuring cylinder before and after some of the water is poured into a beaker.
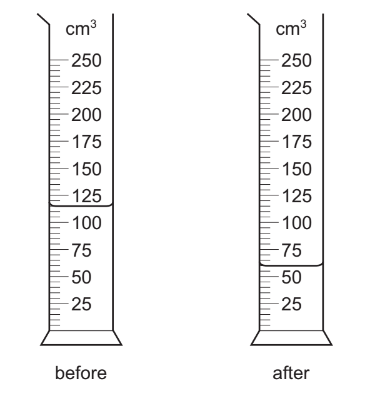
How much water has been poured into the beaker?
A. 51 cm3
B. 52 cm3
C. 55 cm3
D. 63 cm3
▶️ Answer/Explanation
Step 1: Read the initial volume
From the first diagram, the water level is 115 cm3.
Step 2: Read the final volume
From the second diagram, the water level is 60 cm3.
Step 3: Calculate the volume poured out
Volume poured = Initial volume − Final volume
Volume poured = 115 − 60= 55 cm3
Ans: C (55 cm3)
A student uses a measuring cylinder to measure the volume of some water. The diagram shows part of the measuring cylinder. The top and bottom of the meniscus are labelled.
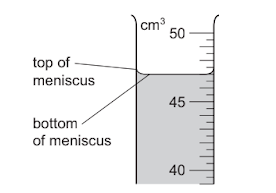
What is the volume of the water?
A. 47.0 cm3
B. 47.5 cm3
C. 49.0 cm3
D. 49.5 cm3
▶️ Answer/Explanation
Step 1: Correct meniscus reading
For water, the meniscus is concave. The correct reading must be taken at the bottom of the meniscus, not the top.
Step 2: Read the scale
From the diagram, the bottom of the meniscus aligns exactly with the 47.0 cm3 mark.
Although the top of the meniscus is closer to 49.0 cm3, this is not the correct reading point.
Ans: A (47.0 cm3)
A student is asked to find the volume of a small irregularly-shaped piece of rock.
He has the following apparatus available.
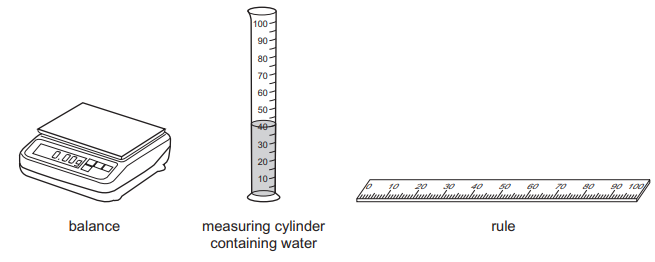
Which apparatus must the student use to find the volume of the small piece of rock?
A. Balance and rule
B. Rule only
C. Balance and measuring cylinder
D. Measuring cylinder only
▶️ Answer/Explanation
Step 1: Identify the shape
The rock is irregularly shaped, so its dimensions cannot be measured accurately using a rule.
Step 2: Suitable method
The correct method is the water displacement method, where the volume of water displaced equals the volume of the rock.
Step 3: Required apparatus
To measure displacement, only a measuring cylinder is required.
A balance is used to measure mass, not volume.
Ans: D (Measuring cylinder only)
The diagrams show the readings on a measuring cylinder before and after a small metal cube is added.
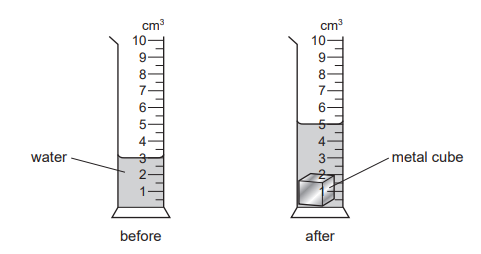
How many more identical cubes can be added to the cylinder, without causing the water to overflow? Do not include the cube already in the cylinder.
A. 1
B. 2
C. 3
D. 4
▶️ Answer/Explanation
Step 1: Volume of one cube
Initial water level = 3 cm3
After adding one cube = 5 cm3
Increase = 5 − 3 = 2 cm3
So, volume of one cube = 2 cm3.
Step 2: Maximum capacity of cylinder
From the scale, the cylinder can hold up to 10 cm3.
Step 3: Remaining space
Current level (with one cube) = 5 cm3
Remaining space = 10 − 5 = 5 cm3
Step 4: Number of additional cubes
Each cube displaces 2 cm3.
5 ÷ 2 = 2 cubes (since 3 cubes would cause overflow).
Ans: B (2)
A student wishes to measure accurately the volume of approximately 40 cm3 of water. She has two measuring cylinders: a larger one that can hold 100 cm3, and a smaller one that can hold 50 cm3. The water forms a meniscus where it touches the glass.
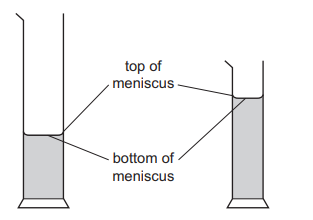
Which cylinder should the student use and which water level should she use to ensure an accurate result?
Cylinder Water level
A. Larger cylinder – bottom of meniscus
B. Larger cylinder – top of meniscus
C. Smaller cylinder – bottom of meniscus
D. Smaller cylinder – top of meniscus
▶️ Answer/Explanation
Step 1: Choose the most suitable cylinder
To obtain a more accurate reading, the measuring cylinder with the smaller maximum capacity should be used. This provides smaller scale divisions and therefore greater precision.
Since the required volume is about 40 cm3, the 50 cm3 cylinder is more accurate than the 100 cm3 cylinder.
Step 2: Correct meniscus reading
For water, the meniscus curves downward (concave). The correct reading is taken at the bottom of the meniscus.
Conclusion:
Smaller cylinder + bottom of meniscus
Ans: C
A geologist compares the volumes of three rocks, X, Y and Z. Three measuring cylinders contain different volumes of water. He places each rock into one of the measuring cylinders.
The diagrams show the measuring cylinders before and after the rocks are put in.
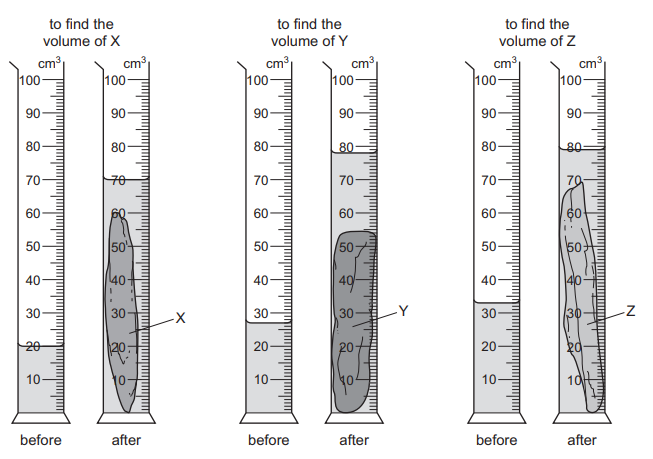
Which row shows the volumes of X, Y and Z in order, from largest to smallest?
▶️ Answer/Explanation
Step 1: Determine volume displacement for each rock
The volume of each rock equals the rise in water level in its measuring cylinder.
Rock X: Final level − Initial level = Volume of X
Rock Y: Final level − Initial level = Volume of Y
Rock Z: Final level − Initial level = Volume of Z
Step 2: Compare the increases
From the diagrams, rock Y causes the greatest increase in water level, rock X causes the next largest increase, and rock Z causes the smallest increase.
Step 3: Arrange from largest to smallest
Y > X > Z
This order corresponds to row B.
Ans: B
Diagram 1 shows a measuring cylinder containing water.
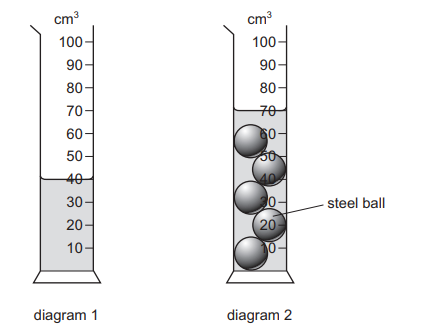
Five identical steel balls are lowered into the measuring cylinder. Diagram 2 shows the new water level.
What is the volume of each steel ball?
A. 6 cm3
B. 14 cm3
C. 30 cm3
D. 70 cm3
▶️ Answer/Explanation
Step 1: Determine the initial water level
From Diagram 1, the initial volume of water is 40 cm3.
Step 2: Determine the final water level
From Diagram 2, the water level rises to 70 cm3.
Step 3: Calculate total volume displaced
Increase in volume = 70 − 40 = 30 cm3
This 30 cm3 is the total volume of the 5 steel balls.
Step 4: Volume of one ball
30 ÷ 5 = 6 cm3
Ans: A (6 cm3)
