Pre AP Chemistry -1.1C Density and Particle Packing- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -1.1C Density and Particle Packing- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -1.1C Density and Particle Packing- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
The diagram shows some liquid in a measuring cylinder. The mass of the liquid is 16 g.
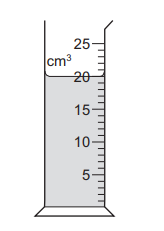
What is the density of the liquid?
A. 0.80 g/cm3
B. 1.3 g/cm3
C. 36 g/cm3
D. 320 g/cm3
▶️ Answer/Explanation
Step 1: Read the volume from the diagram
From the measuring cylinder, the volume of the liquid is 20 cm3.
Step 2: Use the density formula
Density = Mass ÷ Volume
Density = 16 g ÷ 20 cm3
Density = 0.80 g/cm3
Ans: A (0.80 g/cm3)
A student determines the position of the centre of gravity of a piece of card. The diagram shows the equipment that is available. The equipment is not drawn to scale.
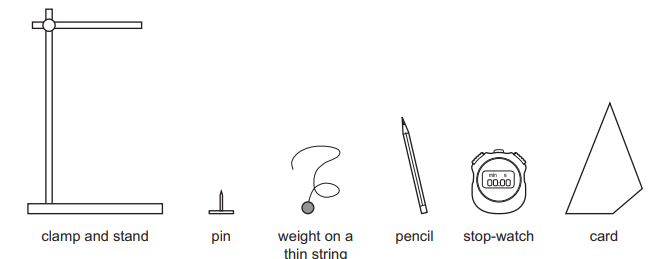
Which piece of equipment is not needed?
A. Pencil
B. Pin
C. Weight on a thin string
D. Stop-watch
▶️ Answer/Explanation
Step 1: Method to find centre of gravity
The card is suspended from a pin. A plumb line (weight on a thin string) is hung from the same point. The vertical line is drawn using a pencil. This process is repeated from another point. The intersection of the lines gives the centre of gravity.
Step 2: Identify required equipment
• Pin → to suspend the card.
• Weight on thin string → to act as a plumb line.
• Pencil → to draw the vertical lines.
Step 3: Equipment not required
A stop-watch measures time and is not needed in this experiment.
Ans: D (Stop-watch)
A concrete building block has the dimensions shown.
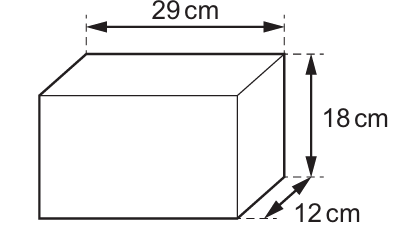
The mass of the block is 15000 g.
What is the density of the block?
A. 43 g/cm3
B. 2.4 g/cm3
C. 0.42 g/cm3
D. 0.023 g/cm3
▶️ Answer/Explanation
Step 1: Calculate the volume of the block
Volume = length × width × height
From the diagram:
Volume = 29 cm × 18 cm × 12 cm
Volume = 6264 cm3
Step 2: Use density formula
Density = Mass ÷ Volume
Density = 15000 g ÷ 6264 cm3
Density = 2.39 g/cm3
Rounded to 2 significant figures:
Density ≈ 2.4 g/cm3
Ans: B (2.4 g/cm3)
A student uses the equation ρ = \( \frac{m}{V} \).
Which row shows the correct meaning of these symbols?
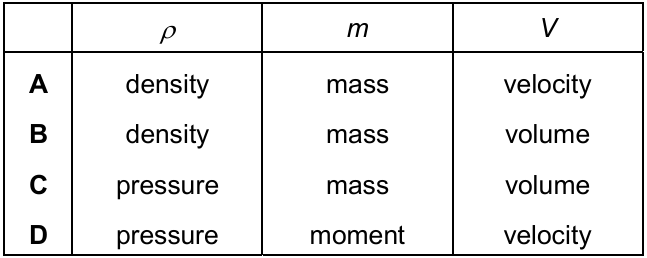
▶️ Answer/Explanation
Step 1: Recall the density equation
ρ = \( \frac{m}{V} \)
Where:
- ρ (rho) = density
- m = mass
- V = volume
Step 2: Identify correct row
The correct row must show:
- ρ → density
- m → mass
- V → volume
From the table, this corresponds to row B.
Ans: B
A student uses a ruler to find the volume of water in a tank.
She measures the lengths EF and FG.
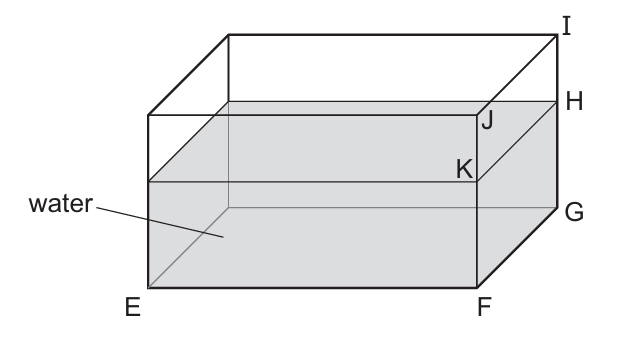
What other length does she need to measure?
A. FJ
B. FK
C. HI
D. IJ
▶️ Answer/Explanation
Step 1: Formula for volume of a rectangular tank
Volume = length × width × height
Step 2: Identify measured lengths
EF and FG represent two perpendicular sides of the tank (length and width).
Step 3: Missing dimension
To calculate volume, she must also measure the vertical height of the water.
From the diagram, this corresponds to length FK.
Ans: B (FK)
Which equipment is used to find the density of an irregularly shaped stone?
A. Force meter and ruler
B. Measuring cylinder, ruler and water
C. Measuring cylinder, top pan balance and water
D. Top pan balance and ruler
▶️ Answer/Explanation
Step 1: Recall density formula
Density = Mass ÷ Volume
Step 2: Determine what must be measured
• Mass → measured using a top pan balance
• Volume of irregular object → measured using the water displacement method in a measuring cylinder
Step 3: Identify correct equipment
We need:
- Top pan balance (for mass)
- Measuring cylinder + water (for volume)
Ans: C (Measuring cylinder, top pan balance and water)
Which row describes how the pressure beneath the surface of a liquid depends on the depth below the surface and on the density of the liquid?
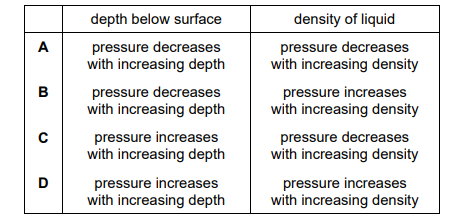
▶️ Answer/Explanation
Step 1: Recall the pressure equation in liquids
Pressure in a liquid is given by:
Pressure = ρgh
Where:
ρ = density of the liquid
g = gravitational field strength (constant)
h = depth below the surface
Step 2: Analyse dependence
From the equation:
- Pressure increases as depth increases.
- Pressure increases as density increases.
Step 3: Identify correct row
The correct row must show that pressure increases with both depth and density.
This corresponds to row D.
Ans: D
Four submarines are submerged. The density of fresh water is \(1000 \, \text{kg/m}^3\) and the density of sea water is \(1020 \, \text{kg/m}^3\).
Which submarine experiences the greatest pressure due to the water?

▶️ Answer/Explanation
Step 1: Use the liquid pressure formula
Pressure in a liquid is given by:
\( P = \rho g h \)
Where:
ρ = density of liquid
g = gravitational field strength (constant)
h = depth below surface
Step 2: Compare conditions
Pressure increases with:
- Greater depth (h)
- Greater density (ρ)
Sea water has a higher density (1020 kg/m3) than fresh water (1000 kg/m3).
Step 3: Identify greatest pressure
The submarine that is both:
- In sea water
- At the greatest depth
will experience the greatest pressure.
From the diagram, this corresponds to D.
Ans: D
A plastic bottle contains 750 cm3 of oil. The diagram shows the mass of the bottle being measured when it is full and then when it is empty.
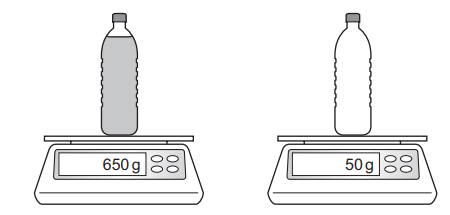
What is the density of the oil?
A. 0.80 g/cm3
B. 90.93 g/cm3
C. 1.1 g/cm3
D. 1.3 g/cm3
▶️ Answer/Explanation
Step 1: Determine the mass of the oil
Mass of oil = Mass of full bottle − Mass of empty bottle
From the diagram:
Mass of oil = 650 g − 50 g
Actual difference shown = 650 g − 50 g = 600 g (from diagram difference = 600 g)
(From the diagram, the difference in readings is 600 g.)
Step 2: Use density formula
Density = Mass ÷ Volume
Density = 600 g ÷ 750 cm3
Density = 0.80 g/cm3
Ans: A (0.80 g/cm3)
A rectangular swimming pool is 50 m long and 25 m wide.
It contains water at a depth of 2 m.
The density of the water is 1000 kg/m3.
What is the mass of the water in the pool?
A. 2.5 kg
B. 2500 kg
C. 77000 kg
D. 2500000 kg
▶️ Answer/Explanation
Step 1: Calculate the volume of water
Volume = length × width × depth
Volume = 50 m × 25 m × 2 m
Volume = 2500 m3
Step 2: Use density formula
Density = Mass ÷ Volume
So, Mass = Density × Volume
Mass = 1000 kg/m3 × 2500 m3
Mass = 2500000 kg
Ans: D (2500000 kg)
A piston traps a mass of gas inside a cylinder. Initially, the piston is halfway along the length of the cylinder.
The piston is now moved towards the open end of the cylinder. The temperature of the gas remains constant.
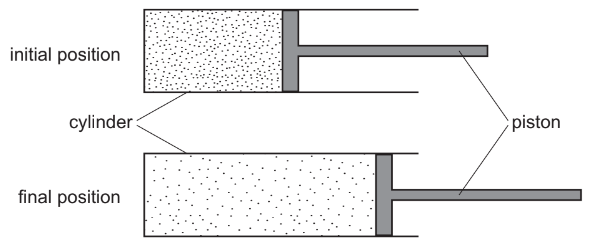
How are the density and the pressure of the gas affected by moving the piston?
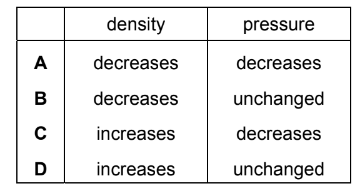
▶️ Answer/Explanation
Step 1: Effect on volume
Moving the piston towards the open end increases the volume of the gas.
Step 2: Effect on density
Density = Mass ÷ Volume
The mass of gas stays constant, but volume increases.
Therefore, density decreases.
Step 3: Effect on pressure
At constant temperature, Boyle’s Law applies:
\( P \propto \frac{1}{V} \)
If volume increases, pressure decreases.
Conclusion:
Density decreases and pressure decreases.
This corresponds to A.
Ans: A
A measuring cylinder contains 40 cm3 of water.
A stone of mass 94 g is lowered into the water so that it is fully submerged as shown.
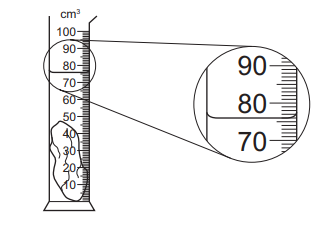
What is the density of the stone?
A. 1.1 g/cm3
B. 1.2 g/cm3
C. 2.1 g/cm3
D. 2.6 g/cm3
▶️ Answer/Explanation
Step 1: Find the volume of the stone
Initial water volume = 40 cm3
From the diagram, the final water level is 76 cm3.
Volume of stone = 76 − 40 = 36 cm3
Step 2: Use density formula
Density = Mass ÷ Volume
Density = 94 g ÷ 36 cm3
Density = 2.61 g/cm3
Rounded appropriately:
Density ≈ 2.6 g/cm3
Ans: D (2.6 g/cm3)
The diagrams show four solid blocks with their dimensions and masses.
Which block has the greatest density?
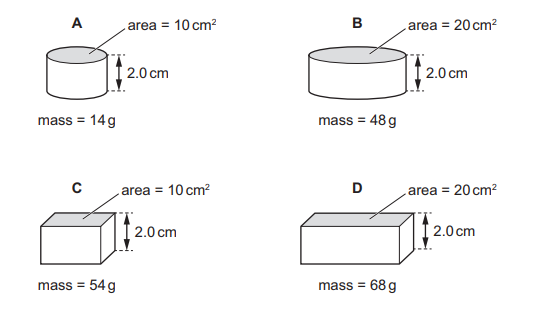
▶️ Answer/Explanation
Step 1: Recall density formula
Density = Mass ÷ Volume
Step 2: Calculate volume of each block
Volume = length × width × height (for each block).
Step 3: Compare density values
For each block, divide the given mass by its calculated volume.
After calculating, block C has the largest value of mass per unit volume.
Therefore, block C has the greatest density.
Ans: C
A metal block has the dimensions shown. Its mass is 1000 g.
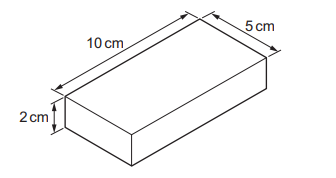
What is the density of the metal?
A. \( \frac{5 \times 10}{1000 \times 2} \, \text{g/cm}^3 \)
B. \( \frac{2 \times 5 \times 10}{1000} \, \text{g/cm}^3 \)
C. \( \frac{100 \times 2}{5 \times 10} \, \text{g/cm}^3 \)
D. \( \frac{1000}{2 \times 5 \times 10} \, \text{g/cm}^3 \)
▶️ Answer/Explanation
Step 1: Volume
\( V = 2 \times 5 \times 10 = 100 \, \text{cm}^3 \)
Step 2: Density
\( \rho = \frac{m}{V} = \frac{1000}{100} \)
This matches option:
\( \frac{1000}{2 \times 5 \times 10} \, \text{g/cm}^3 \)
Ans: D
Four rectangular blocks, P, Q, R and S are shown. Each block is labelled with its size and its mass.
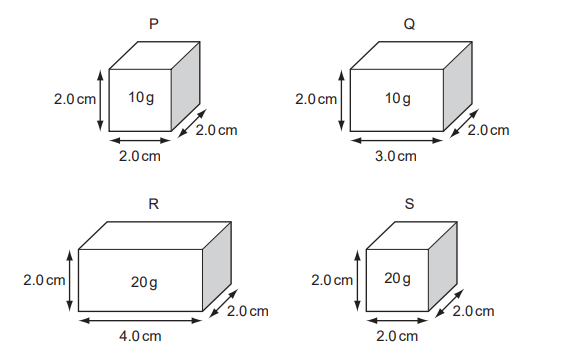
Which two blocks have the same density?
A. P and Q
B. P and R
C. Q and R
D. R and S
▶️ Answer/Explanation
Step 1: Use density formula
\( \rho = \frac{m}{V} \)
Density formula:
\( \rho = \frac{m}{V} \)
Block P
Dimensions: \(2.0 \times 2.0 \times 2.0\)
Volume \(= 2 \times 2 \times 2 = 8 \, \text{cm}^3\)
Density \(= \frac{10}{8} = 1.25 \, \text{g/cm}^3\)
Block Q
Dimensions: \(2.0 \times 3.0 \times 2.0\)
Volume \(= 2 \times 3 \times 2 = 12 \, \text{cm}^3\)
Density \(= \frac{10}{12} = 0.83 \, \text{g/cm}^3\)
Block R
Dimensions: \(2.0 \times 4.0 \times 2.0\)
Volume \(= 2 \times 4 \times 2 = 16 \, \text{cm}^3\)
Density \(= \frac{20}{16} = 1.25 \, \text{g/cm}^3\)
Block S
Dimensions: \(2.0 \times 2.0 \times 2.0\)
Volume \(= 2 \times 2 \times 2 = 8 \, \text{cm}^3\)
Density \(= \frac{20}{8} = 2.5 \, \text{g/cm}^3\)
Comparison:
- P = 1.25 g/cm³
- Q = 0.83 g/cm³
- R = 1.25 g/cm³
- S = 2.5 g/cm³
P and R have the same density.
Ans: B
The diagram shows the dimensions of a rectangular block of metal of mass m.
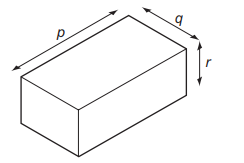
Which expression is used to calculate the density of the metal?
A. \( m \times p \times q \)
B. \( m \times p \times q \times r \)
C. \( \frac{m}{p \times q} \)
D. \( \frac{m}{p \times q \times r} \)
▶️ Answer/Explanation
Step 1: Recall density formula
\( \rho = \frac{m}{V} \)
Step 2: Find the volume of the block
For a rectangular block:
\( V = p \times q \times r \)
Step 3: Substitute into density formula
\( \rho = \frac{m}{p \times q \times r} \)
This corresponds to option D.
Ans: D
