Pre AP Chemistry -2.1A Pure Substances- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.1A Pure Substances- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.1A Pure Substances- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which statement about the boxes P, Q and R is correct?

A) Box P contains two compounds, and box R contains two elements.
B) Box P contains two elements, and box Q contains a mixture.
C) Box P contains two elements, and box Q contains one compound.
D) Box Q contains two compounds, and box R contains a mixture.
▶️ Answer/Explanation
Ans: C
Without the diagram, we can analyze the options based on typical exam patterns:
Option C is correct because it’s common for exam questions to show two elements (like separate atoms of different types) in one box (P), and a compound (molecules made of bonded atoms) in another box (Q).
The other options are less likely because:
A: If P contains two compounds, R would typically show a mixture, not elements.
B: If Q contains a mixture, it would show separate compounds/elements, not just one type.
D: Two compounds in Q would typically be shown as distinct molecules, which would make R a mixture of these, but this is less common in basic questions.
A diagram representing a mixture of four particles is shown.

Which statement describes the mixture of particles?
A) It is a mixture of two different compounds.
B) It is a mixture of two different elements.
C) It is a mixture of four different compounds.
D) It is a mixture of four different elements.
▶️ Answer/Explanation
Ans: A
While the diagram isn’t shown, we can deduce from the answer choices and the correct answer (A) that:
The mixture must contain two types of compounds (molecules made of different elements bonded together).
For example, it might show H₂O molecules and CO₂ molecules mixed together – two different compounds, each made of multiple atoms.
Option B would describe a mixture like O₂ and H₂ (two elements), which isn’t the case here.
Four substances in a mixture are listed.
• calcium nitrate
• iron(II) sulfate
• oxygen
• water
Which statement describes the mixture?
A) It contains 6 elements.
B) It contains 3 compounds and 1 element.
C) It contains 2 compounds and 2 elements.
D) It contains 4 compounds.
▶️ Answer/Explanation
Ans: B
1. Calcium nitrate (Ca(NO₃)₂), iron(II) sulfate (FeSO₄), and water (H₂O) are compounds (multiple elements chemically bonded).
2. Oxygen (O₂) is a diatomic element (pure substance).
3. Thus, the mixture contains 3 compounds and 1 element.
4. Option B is correct as it matches this description.
Which diagram represents a mixture of compounds?
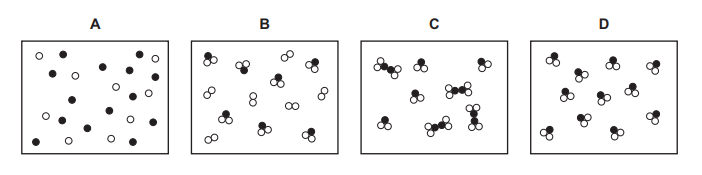
A) Diagram A
B) Diagram B
C) Diagram C
D) Diagram D
▶️ Answer/Explanation
Ans: C
A mixture of compounds consists of two or more different compounds (molecules made of different elements) mixed together. In Diagram C:
1. There are two distinct types of molecules (e.g., diatomic and triatomic).
2. Both are compounds (since they contain different atoms bonded together).
3. They are not chemically reacting, just coexisting as a mixture.
Thus, Diagram C correctly represents a mixture of compounds.
Matter exists as elements, compounds and mixtures. Which row identifies an element, a compound and a mixture?

▶️ Answer/Explanation
Ans: C
1. Calcium (Ca) is an element because it consists of only one type of atom and cannot be broken down into simpler substances.
2. Sodium chloride (NaCl) is a compound formed by the chemical combination of sodium (Na) and chlorine (Cl) in a fixed ratio.
3. Brass is a mixture (specifically an alloy) of copper and zinc, where the components are physically combined but not chemically bonded.
Thus, row C correctly identifies an element (Ca), a compound (NaCl), and a mixture (Brass).
Which substances can be separated by filtration?
A) Insoluble liquid and water
B) Insoluble solid and water
C) Solution of soluble liquid in water
D) Solution of soluble solid in water
▶️ Answer/Explanation
Ans: B
Filtration is a separation technique used to separate an insoluble solid from a liquid. Here’s why:
1. Insoluble solids (e.g., sand in water) can be trapped by filter paper, while the liquid passes through.
2. Soluble substances (liquids or solids) dissolve completely and pass through the filter, making filtration ineffective.
3. Immiscible liquids (e.g., oil and water) are separated by decantation, not filtration.
Thus, only option B (insoluble solid and water) is correct.
Impurities change the melting and boiling points of substances.
Sodium chloride is added to a sample of pure water.
How does the addition of sodium chloride affect the melting point and the boiling point of the water?
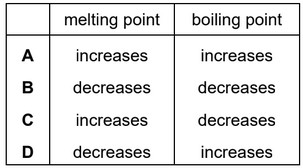
▶️ Answer/Explanation
Ans: D
When sodium chloride is dissolved in water, two key effects occur:
- Melting point depression: The dissolved ions disrupt water’s hydrogen bonding, making it harder to freeze. Thus, the melting point decreases.
- Boiling point elevation: The ions increase intermolecular forces, requiring more energy for water to vaporize. Thus, the boiling point increases.
This matches option D in the given table, where the melting point is lowered and the boiling point is raised.
A student put exactly 25.00 \(cm^3\) of dilute hydrochloric acid into a conical flask.
The student added 2.5 g of solid sodium carbonate and measured the change in temperature of
the mixture.
Which apparatus does the student need to use?
A) balance, measuring cylinder, thermometer
B) balance, pipette, stopwatch
C) balance, pipette, thermometer
D) burette, pipette, thermometer
▶️ Answer/Explanation
Ans: C
The student needs:
1. A balance to accurately measure the 2.5 g of sodium carbonate.
2. A pipette to measure exactly 25.00 \(cm^3\) of hydrochloric acid (a measuring cylinder is less precise).
3. A thermometer to record the temperature change during the reaction.
A stopwatch (option B) is unnecessary since temperature change is the measured variable, not time.
Rock salt is a mixture of sand and sodium chloride. Sodium chloride is soluble in water but not in hexane. Sand is insoluble in both water and hexane. What is required to separate the sand from the sodium chloride?
- filter paper
- fractionating column
- hexane
- water
A) 1 and 3
B) 1 and 4
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: B
To separate sand from sodium chloride:
- Water is needed to dissolve sodium chloride (soluble), while sand remains undissolved (insoluble).
- Filter paper is required to separate the insoluble sand from the sodium chloride solution via filtration.
- Hexane is not useful since sodium chloride is insoluble in it, and sand is also unaffected.
- A fractionating column is unnecessary as no distillation is involved.
Thus, the correct tools are filter paper (1) and water (4).
2.00 g of powdered calcium carbonate is added to 50.0 \(cm^{3}\) of hydrochloric acid. Which apparatus is used to measure the calcium carbonate and the hydrochloric acid?
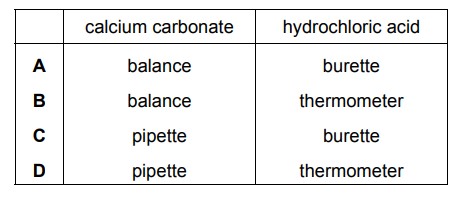
▶️ Answer/Explanation
Ans: A
To measure the quantities accurately:
1. A balance is used to measure the mass of calcium carbonate (2.00 g) since mass is best measured using a balance.
2. A burette is used to measure the volume of hydrochloric acid (50.0 \(cm^{3}\)) because it provides precise volume measurements for liquids.
The other options (B, C, D) involve incorrect apparatus for the given measurements.
A student is asked to measure the time taken for 0.4 g of magnesium carbonate to react completely with 25.0 cm3 of dilute hydrochloric acid.
Which pieces of apparatus does the student need?
- balance, stop-clock, pipette
- balance, stop-clock, thermometer
- balance, pipette, thermometer
- stop-clock, pipette, thermometer
▶️ Answer/Explanation
Ans: A
To measure the time taken for the reaction:
1. A balance is needed to measure the mass of magnesium carbonate (0.4 g).
2. A stop-clock is required to record the reaction time.
3. A pipette is used to accurately measure the volume of hydrochloric acid (25.0 cm3).
A thermometer is unnecessary since temperature is not being monitored. Thus, option A is correct.
Some information about solid silver chloride and solid sodium chloride is shown.
- Silver chloride and sodium chloride do not dissolve in kerosene.
- Silver chloride is insoluble in water but sodium chloride is soluble in water.
- The boiling point of silver chloride is 1547°C and the boiling point of sodium chloride is 1413°C.
Which processes are used to separate a mixture of solid silver chloride and solid sodium chloride?
- Add kerosene, stir and then filter.
- Add water, stir and then filter.
- Add water, stir and then leave to crystallize.
- Add water, stir and then perform fractional distillation.
▶️ Answer/Explanation
Ans: B
1. Water is added because sodium chloride (NaCl) dissolves in water, while silver chloride (AgCl) remains insoluble.
2. Stirring ensures complete dissolution of NaCl, leaving AgCl as a solid residue.
3. Filtration separates the insoluble AgCl (retained on filter paper) from the NaCl solution (filtrate).
4. Other options (A, C, D) are incorrect because kerosene doesn’t dissolve either compound, crystallization doesn’t separate them, and fractional distillation is unnecessary due to their high boiling points.
A student measures the time taken for 2.0 g of magnesium to dissolve in 50 cm3 of dilute sulfuric acid.
Which apparatus is essential to complete the experiment?
- stop-clock
- measuring cylinder
- thermometer
- balance
A) 1, 2 and 4
B) 1 and 2 only
C) 1 and 4 only
D) 2, 3 and 4
▶️ Answer/Explanation
Ans: A
To conduct the experiment properly:
1. A balance is needed to measure the exact mass of magnesium (2.0 g).
2. A measuring cylinder is required to measure the volume of sulfuric acid (50 cm3).
3. A stop-clock is essential to record the time taken for dissolution.
A thermometer is unnecessary since temperature control is not mentioned in the procedure. Thus, the correct choice is 1, 2, and 4 (Option A).
Which method should be used to separate a mixture of two liquids?
- crystallisation
- electrolysis
- filtration
- fractional distillation
▶️ Answer/Explanation
Ans: D
1. Crystallisation is used to separate a solid dissolved in a liquid (not two liquids).
2. Electrolysis decomposes compounds using electricity (not for separating mixtures).
3. Filtration separates solids from liquids (not liquid-liquid mixtures).
4. Fractional distillation is ideal for separating two liquids with close boiling points, as it uses a fractionating column for repeated condensation and vaporization.
Thus, the correct method is D (fractional distillation).
Information about the solubility of four solids, P, Q, R and S, is given in the table.

A student attempted to separate mixtures of these solids using the following method.
1. Add the mixture to a beaker of water and stir.
2. Filter the mixture.
3. Crystallise one of the solids from the filtrate.
Which of the following mixtures could not be separated by this method?
A) A mixture of P and R
B) A mixture of Q and P
C) A mixture of Q and R
D) A mixture of R and S
▶️ Answer/Explanation
Ans: C
The separation method involves dissolving one solid in water, filtering out the insoluble solid, and crystallizing the dissolved solid from the filtrate. Here’s the analysis:
1. P (soluble) and R (insoluble): P dissolves, R remains as residue; P can be crystallized.
2. Q (insoluble) and P (soluble): Q is filtered out, P is crystallized.
3. Q and R (both insoluble): Neither dissolves, so filtration leaves both solids undivided—no filtrate for crystallization.
4. R (insoluble) and S (soluble): R is filtered, S is crystallized.
Thus, option C (Q and R) cannot be separated by this method.
The melting points and boiling points of pure substances W, X and Y are shown.
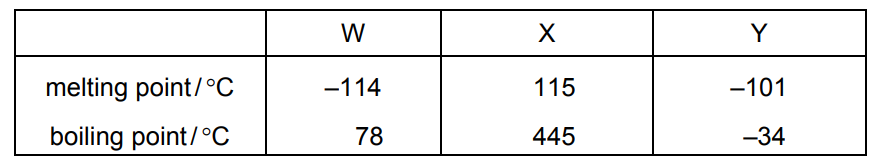
The substances are chlorine, ethanol and sulfur.
Which row identifies W, X and Y?
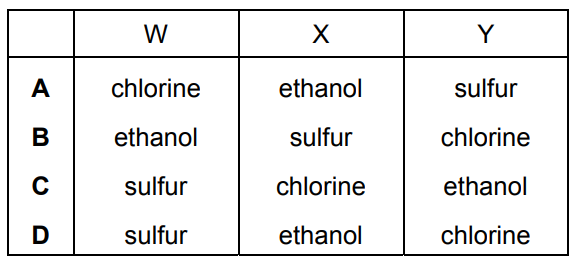
▶️ Answer/Explanation
Ans: B
From the given melting and boiling points:
- X has the highest melting and boiling points, indicating it is a solid at room temperature—this matches sulfur.
- Y has the lowest boiling point, suggesting it is a gas at room temperature—this corresponds to chlorine.
- W has intermediate values, consistent with a liquid at room temperature—this aligns with ethanol.
Thus, the correct identification is W = ethanol, X = sulfur, and Y = chlorine, which corresponds to option B.
A student is given a mixture of barium sulfate, copper(II) sulfate and water.
The table shows information about barium sulfate and copper(II) sulfate.
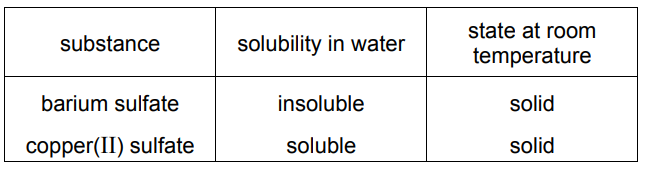
How does the student obtain copper(II) sulfate crystals from the mixture?
- crystallization followed by distillation
- crystallization followed by filtration
- distillation followed by crystallization
- filtration followed by crystallization
▶️ Answer/Explanation
Ans: D
The mixture contains:
- Barium sulfate (BaSO4) – insoluble in water (can be filtered out).
- Copper(II) sulfate (CuSO4) – soluble in water (remains in solution).
Step 1: Filtration – Removes insoluble BaSO4, leaving a CuSO4 solution.
Step 2: Crystallization – Evaporate water from the filtrate to obtain pure CuSO4 crystals.
Thus, the correct sequence is filtration followed by crystallization (Option D).
In which row are the substances correctly classified?

▶️ Answer/Explanation
Ans: C
The correct classification is as follows:
- Sulfur (S) is an element because it consists of only one type of atom.
- Water (H₂O) is a compound since it is formed by the chemical combination of hydrogen (H) and oxygen (O) in a fixed ratio.
- Brass is a mixture (specifically a homogeneous alloy) of copper (Cu) and zinc (Zn), where the composition can vary.
Only Row C correctly identifies sulfur as an element, water as a compound, and brass as a mixture.
Alcohol and water are completely miscible. This means when mixed together they form only one liquid layer.
Which method is used to separate alcohol from water?
- crystallization
- filtration
- fractional distillation
- precipitation
▶️ Answer/Explanation
Ans: C
Since alcohol and water are miscible liquids, they cannot be separated by methods like filtration or crystallization. Fractional distillation is used because:
1. Alcohol (ethanol) has a lower boiling point (~78°C) than water (100°C).
2. When heated, alcohol vaporizes first and is condensed back into a liquid, while water remains behind.
3. This method is effective for liquids with different boiling points, making it ideal for separating alcohol-water mixtures.
A student measures the rate of two reactions.
In one reaction, there is a change in mass of the reactants during the reaction.
In the second reaction, there is a change in temperature during the reaction.
Which piece of apparatus would be essential in both experiments?
- balance
- clock
- pipette
- thermometer
▶️ Answer/Explanation
Ans: B
1. First reaction (change in mass): A balance measures mass loss, and a clock records time to determine the rate.
2. Second reaction (change in temperature): A thermometer tracks temperature change, and a clock measures time.
The only apparatus common to both experiments is the clock, as reaction rates require time measurement in both cases.
Part of the instructions in an experiment reads as follows: Quickly add 50 cm3 of acid.
What is the best piece of apparatus to use?
- a burette
- a conical flask
- a measuring cylinder
- a pipette
▶️ Answer/Explanation
Ans: C
1. Burette (A): Accurate but slow due to controlled drop-by-drop addition—unsuitable for “quick” addition.
2. Conical flask (B): Not a measuring apparatus—cannot measure volume.
3. Measuring cylinder (C): Allows rapid pouring of a fixed volume (50 cm3) with reasonable accuracy.
4. Pipette (D): Precise but time-consuming for large volumes and requires careful handling.
Thus, a measuring cylinder (Option C) best meets the need for both speed and volume measurement.
