Pre AP Chemistry -2.1B Mixtures and Separation Techniques- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.1B Mixtures and Separation Techniques- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.1B Mixtures and Separation Techniques- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
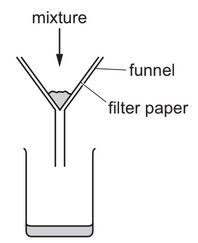
A mixture containing an aqueous salt, sand and hot water is stirred.
The mixture is then poured into the apparatus shown.
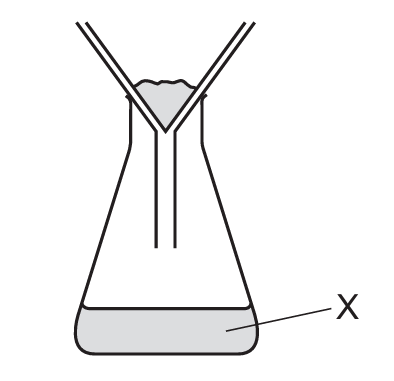
What is X?
A) a filtrate only
B) a residue only
C) a solute only
D) a solvent only
▶️ Answer/Explanation
Ans: A
The described process is filtration. When the mixture is poured into the filtration apparatus, the sand (insoluble solid) will be trapped by the filter paper as the residue, while the aqueous salt solution (filtrate) passes through. X refers to what comes through the filter paper, which is the filtrate – the liquid containing the dissolved salt. It’s not just the solvent (water) because it contains the dissolved salt, and it’s not the solute only because that would imply pure salt without water. The residue (sand) remains in the filter paper.
Sample M contains calcium carbonate and sodium nitrate.
The result of adding water to M, stirring and filtering is shown.
No chemical reaction occurs.
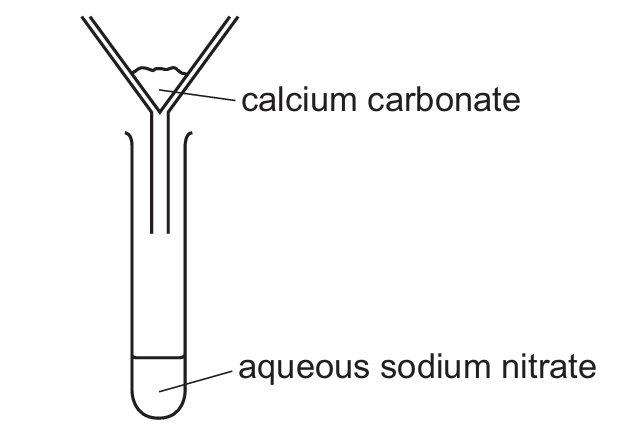
Which terms describe M, calcium carbonate and aqueous sodium nitrate?
| sample M | calcium carbonate | aqueous sodium nitrate | |
|---|---|---|---|
| A | compound | filtrate | residue |
| B | compound | residue | filtrate |
| C | mixture | filtrate | residue |
| D | mixture | residue | filtrate |
▶️ Answer/Explanation
Ans: D
Let’s analyze each component:
1. Sample M: Since it contains two different substances (calcium carbonate and sodium nitrate) physically mixed together, it’s a mixture, not a compound.
2. Calcium carbonate: Insoluble in water, so it remains as the solid residue after filtration.
3. Aqueous sodium nitrate: Soluble in water, so it passes through the filter paper to become the filtrate.
This matches option D where:
– M is a mixture
– Calcium carbonate is the residue
– Aqueous sodium nitrate is the filtrate
The key points are that no chemical reaction occurred (just physical separation) and calcium carbonate is insoluble while sodium nitrate is soluble.
Four different organic compounds are separated by a fractionating column.
The table shows the boiling points of the compounds.
The diagram shows the position in the fractionating column where they are separated.
| compound | boiling point/°C |
|---|---|
| Q | 69 |
| R | 196 |
| S | 90 |
| T | 125 |
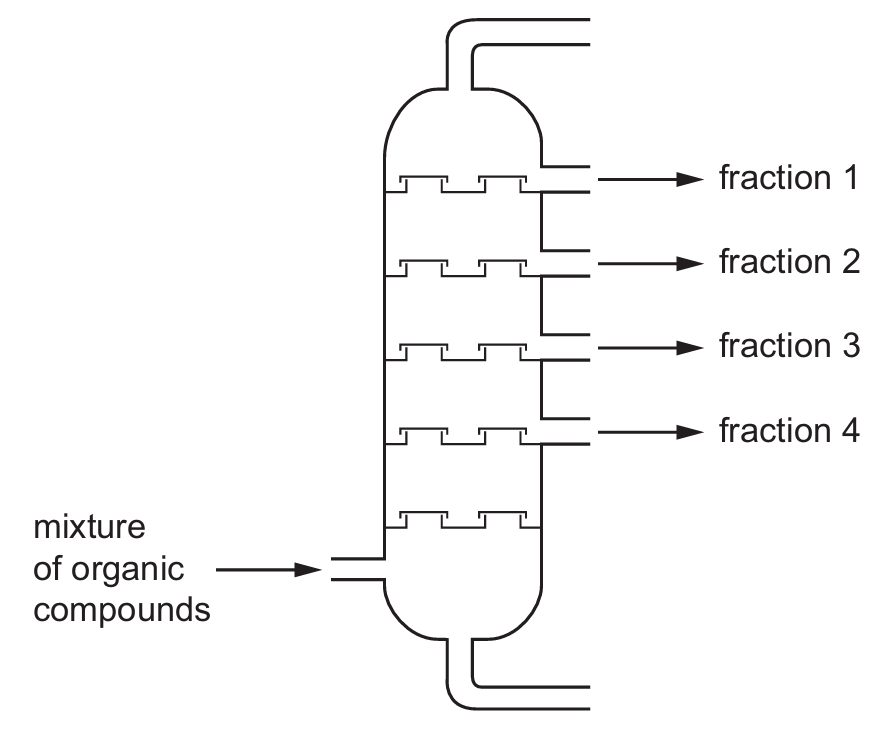
Which row identifies the compound in each fraction?
| fraction 1 | fraction 2 | fraction 3 | fraction 4 | |
|---|---|---|---|---|
| A | Q | S | T | R |
| B | Q | T | S | R |
| C | R | T | S | Q |
| D | R | S | T | Q |
▶️ Answer/Explanation
Ans: A
In fractional distillation, compounds with the lowest boiling points come off first (at the top of the column), while those with higher boiling points come off later (at the bottom).
Ordering the compounds by boiling point:
1. Q (69°C) – lowest boiling point, comes first
2. S (90°C)
3. T (125°C)
4. R (196°C) – highest boiling point, comes last
This matches option A where the fractions are ordered Q → S → T → R.
The diagram shows fraction 1 at the top (lowest boiling point) through to fraction 4 at the bottom (highest boiling point).
Sea water contains dissolved sodium chloride. Which method is used to obtain pure water from sea water?
A) Chromatography
B) Distillation
C) Evaporation
D) Filtration
▶️ Answer/Explanation
Ans: B
To obtain pure water from seawater:
1. Distillation is used because it involves boiling the water and condensing the vapor, leaving behind dissolved salts like sodium chloride.
2. Chromatography (A) separates mixtures based on solubility, not purification.
3. Evaporation (C) leaves behind solids, but does not collect pure water.
4. Filtration (D) removes insoluble impurities, not dissolved salts.
Thus, distillation (B) is the correct method for obtaining pure water from seawater.
The fractional distillation of petroleum is shown.
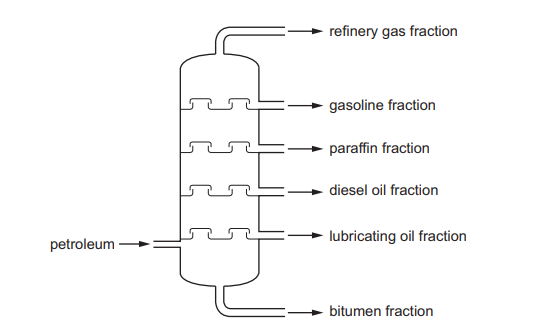
Which fraction is the least volatile?
A) bitumen
B) diesel oil
C) gasoline
D) refinery gas
▶️ Answer/Explanation
Ans: A
In fractional distillation, petroleum is separated based on boiling points. The least volatile fraction has the highest boiling point and is collected at the bottom of the fractionating column. Bitumen is the heaviest and least volatile fraction, used for road surfacing. In contrast, refinery gas (D) is the most volatile, followed by gasoline (C) and diesel oil (B).
Copper(II) sulfate crystals are blue. They are made by adding an excess of copper(II) oxide to sulfuric acid. The mixture is heated and stirred. The mixture is then filtered and the filtrate is allowed to evaporate, leaving blue crystals. Why is filtration necessary?
A) to remove soluble impurities
B) to remove sulfuric acid
C) to remove the blue crystals
D) to remove unreacted copper(II) oxide
▶️ Answer/Explanation
Ans: D
Filtration is necessary to remove the unreacted copper(II) oxide (D). Here’s why:
1. Copper(II) oxide is insoluble in water, while copper(II) sulfate is soluble.
2. After the reaction, the excess solid CuO remains and must be separated from the CuSO₄ solution.
3. Filtration retains the solid CuO while allowing the CuSO₄ solution to pass through.
4. The filtrate (containing dissolved CuSO₄) is then evaporated to obtain pure blue crystals.
Thus, the correct answer is D.
Molten iron from the blast furnace contains impurities.
The process of turning the impure iron into steel involves blowing oxygen into the molten iron and adding calcium oxide.
What are the reasons for blowing in oxygen and adding calcium oxide?

▶️ Answer/Explanation
Ans: A
In the steelmaking process:
1. Oxygen is blown into the molten iron to oxidize impurities like carbon, silicon, and phosphorus, converting them into oxides (e.g., CO₂, SiO₂, P₂O₅).
2. Calcium oxide (CaO) is added as a flux to react with acidic oxides (e.g., SiO₂, P₂O₅) and form slag, which is removed to purify the iron.
Thus, the correct answer is A—oxygen oxidizes impurities, and calcium oxide removes acidic oxides.
Petroleum is separated into fractions by fractional distillation.
Separation occurs in a fractionating column.
Some properties of three of these fractions are shown.

Which statement is correct?
- Fraction 1 has a higher boiling point range than fraction 2.
- Fraction 2 is removed from a higher point in the fractionating column than fraction 1.
- Molecules in fraction 3 have shorter chains than those in fraction 2.
- None of the fractions are liquid at room temperature.
▶️ Answer/Explanation
Ans: C
1. Option A is incorrect because Fraction 1 (LPG) has a lower boiling point range than Fraction 2 (gasoline).
2. Option B is incorrect since Fraction 2 (gasoline) is collected lower in the column than Fraction 1 (LPG).
3. Option C is correct—Fraction 3 (kerosene) has longer hydrocarbon chains than Fraction 2 (gasoline).
4. Option D is incorrect because some fractions (e.g., gasoline) are liquid at room temperature.
Copper(II) sulfate is prepared by adding excess copper(II) oxide to warm dilute sulfuric acid.
Which purification methods are used to obtain pure solid copper(II) sulfate from the reaction mixture?
- crystallisation
- filtration
- chromatography
- distillation
A) 1 and 4
B) 1 and 2
C) 2 and 3
D) 3 and 4
▶️ Answer/Explanation
Ans: B
To obtain pure copper(II) sulfate from the reaction mixture:
- Filtration (2) is used first to remove the excess insoluble copper(II) oxide.
- Crystallisation (1) is then performed to obtain pure solid copper(II) sulfate from the filtered solution.
Chromatography (3) and distillation (4) are not suitable for this purification process. Thus, the correct methods are 1 and 2.
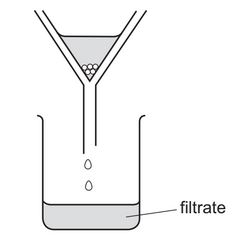
A mixture is separated using the apparatus shown.
What is the mixture?
A) aqueous copper(II) sulfate and aqueous sodium chloride
B) aqueous copper(II) sulfate and copper
C) copper and sulfur
D) ethanol and ethanoic acid
▶️ Answer/Explanation
Ans: B
The apparatus shown is a filtration setup, which separates an insoluble solid from a liquid. Among the options:
- A: Both are aqueous (soluble), so filtration cannot separate them.
- B: Copper (insoluble solid) can be separated from aqueous copper(II) sulfate (liquid) via filtration.
- C: Both copper and sulfur are solids; filtration is ineffective.
- D: Both are miscible liquids; filtration does not apply.
Thus, the correct mixture is B) aqueous copper(II) sulfate and copper.
The apparatus used to separate a mixture of sand, methanol, and ethanol is shown.
▶️ Answer/Explanation
Ans: D
The given apparatus is a fractional distillation setup, which is used to separate miscible liquids with different boiling points.
1. Sand (solid) is removed first by filtration.
2. Methanol (BP: 64.7°C) and ethanol (BP: 78.4°C) are separated based on their boiling points.
3. The fractionating column improves separation efficiency by allowing repeated condensation and vaporization.
Thus, the correct method for separating this mixture involves fractional distillation.
A student separates sugar from pieces of broken glass by dissolving the sugar in water and filtering off the broken glass.
What is the filtrate?
- broken glass only
- broken glass and sugar solution
- pure water
- sugar solution
▶️ Answer/Explanation
Ans: D
In filtration:
1. The filtrate is the liquid that passes through the filter paper.
2. Sugar dissolves in water, forming a solution that passes through.
3. Broken glass (insoluble) remains as the residue on the filter paper.
Therefore, the filtrate is the sugar solution (Option D).
Copper(II) chloride crystals are made by adding solid copper(II) carbonate to dilute hydrochloric acid until no more dissolves. Which process is used to obtain pure copper(II) chloride crystals from the mixture?
A) distillation of the mixture
B) evaporation of the mixture
C) filtration followed by drying of the residue
D) filtration followed by evaporation of the filtrate
▶️ Answer/Explanation
Ans: D
1. Reaction: Copper(II) carbonate reacts with HCl to form soluble copper(II) chloride, water, and CO2 gas.
2. Filtration: Removes unreacted solid copper(II) carbonate, leaving copper(II) chloride in solution (filtrate).
3. Evaporation: The filtrate is heated to evaporate water, leaving pure copper(II) chloride crystals.
Distillation (A) is unnecessary, and evaporation alone (B) would leave impurities. Drying the residue (C) would not yield the desired product.
Which statement about aqueous sodium hydroxide is correct?
- When it is added to a solution containing sulfate ions, a white precipitate is formed.
- When it is added to a solution of copper(II) ions, a blue precipitate is formed which dissolves in excess to give deep blue solution.
- When it is added to a solution of iron(II) ions, a green precipitate is formed which does not dissolve in excess.
- When it is added to ammonium chloride, a gas is produced which turns blue litmus red.
▶️ Answer/Explanation
Ans: C
Option A: Sodium hydroxide does not form a precipitate with sulfate ions (Ba²⁺ is needed for a white precipitate).
Option B: While Cu(OH)₂ (blue precipitate) forms, it does not dissolve in excess NaOH (only in ammonia).
Option C: Correct. Fe(OH)₂ forms a green precipitate and remains insoluble in excess NaOH.
Option D: NH₃ gas is produced (turns red litmus blue, not blue litmus red).
Thus, only option C is entirely accurate.
Which process is used to obtain lime from limestone?
- Cracking
- Fractional distillation
- Neutralization
- Thermal decomposition
▶️ Answer/Explanation
Ans: D
To obtain lime (calcium oxide, CaO) from limestone (calcium carbonate, CaCO3):
1. Thermal decomposition is used, where limestone is heated strongly (~900°C) to break it down into lime and carbon dioxide.
2. The chemical equation is: \[ \text{CaCO}_3 \xrightarrow{\text{heat}} \text{CaO} + \text{CO}_2 \]
3. Cracking (A) breaks large hydrocarbons into smaller ones (irrelevant here).
4. Fractional distillation (B) separates liquid mixtures (not applicable).
5. Neutralization (C) involves acids and bases reacting (not used here).
Thus, thermal decomposition (D) is the correct process.
Which method can be used to separate a mixture of salt and water to obtain both parts of the mixture?
A) crystallisation
B) distillation
C) evaporation
D) filtration
▶️ Answer/Explanation
Ans: B
To separate salt and water while recovering both components, distillation is the appropriate method. Here’s why:
- Distillation involves boiling the mixture and condensing the vapor, allowing pure water to be collected while salt remains behind.
- Evaporation (C) and crystallisation (A) only recover the salt, losing the water.
- Filtration (D) is unsuitable as salt is dissolved in water and cannot be filtered out.
Thus, distillation is the only method that separates and retains both salt and water.
The diagram shows how muddy water can be purified.
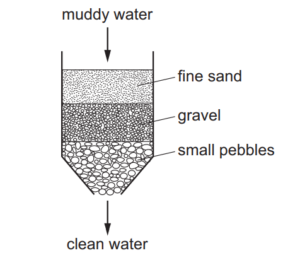
Which process for purifying the muddy water is shown?
A) crystallisation
B) distillation
C) filtration
D) solvent extraction
▶️ Answer/Explanation
Ans: C
The diagram shows filtration (C) because:
1. Muddy water contains insoluble solid particles suspended in water.
2. The filter paper traps the solid mud particles while allowing clean water to pass through.
3. Crystallization (A) and distillation (B) are used for soluble solids or liquid mixtures, not suspensions.
4. Solvent extraction (D) involves separating compounds based on solubility differences, which doesn’t apply here.
Therefore, filtration is the correct method for separating insoluble solids from liquids.
Which method separates a mixture of sugar and glass?
A) dissolve, filter and evaporate
B) distillation and filter
C) fractionally distillation
D) use chromatography
▶️ Answer/Explanation
Ans: A
To separate sugar (soluble) from glass (insoluble):
1. Dissolve the mixture in water—sugar dissolves, while glass remains undissolved.
2. Filter to remove insoluble glass particles, leaving a sugar solution.
3. Evaporate the water to recover solid sugar.
This method (A) is effective because it exploits the solubility difference between sugar and glass.
Which method of purification would produce water most suitable for drinking?
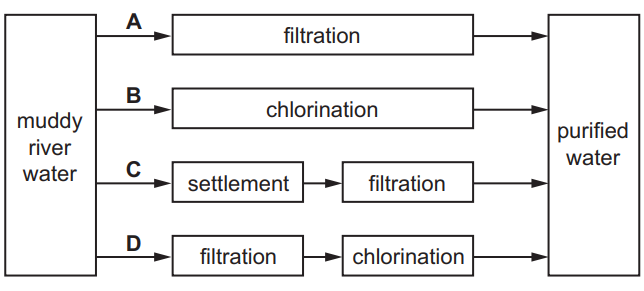
▶️ Answer/Explanation
Ans: D
1. Option A (Filtration) removes insoluble impurities but not dissolved salts or microbes.
2. Option B (Chlorination) kills bacteria but does not remove solid impurities or dissolved chemicals.
3. Option C (Distillation) produces pure water but removes essential minerals, making it less ideal for drinking.
4. Option D (Filtration + Chlorination) combines removing suspended particles (filtration) and disinfecting (chlorination), producing safe and potable water.
Thus, D is the most suitable method for drinking water purification.
Which statement about methane is not correct?
- It is a liquid produced by distilling petroleum.
- It is produced as vegetation decomposes.
- It is produced by animals, such as cows.
- It is used as a fuel.
▶️ Answer/Explanation
Ans: A
Methane (CH4) is a gas, not a liquid. The incorrect statement is:
- A – Methane is not obtained from petroleum distillation (that produces liquid hydrocarbons like gasoline).
The other statements are correct:
- B – Methane forms when organic matter decomposes anaerobically (e.g., in swamps).
- C – Cows produce methane during digestion (enteric fermentation).
- D – Methane is a primary component of natural gas, used as fuel.
A mixture is separated using the apparatus shown.
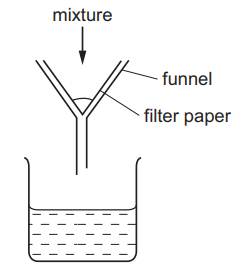
What is the mixture?
- aqueous copper chloride and copper
- aqueous copper chloride and sodium chloride
- ethane and methane
- ethanol and water
▶️ Answer/Explanation
Ans: A
The apparatus shown is a filtration setup, which separates an insoluble solid from a liquid. Analyzing the options:
- A: Copper (insoluble solid) can be separated from aqueous copper chloride (liquid) via filtration.
- B: Both are aqueous (soluble), so filtration cannot separate them.
- C: Both ethane and methane are gases; filtration is irrelevant.
- D: Both are miscible liquids; filtration does not apply.
Thus, the correct mixture is A) aqueous copper chloride and copper.
The table shows some fractions that are obtained from petroleum by fractional distillation, together with some of their uses.

Which row correctly identifies fractions 1, 2 and 3?
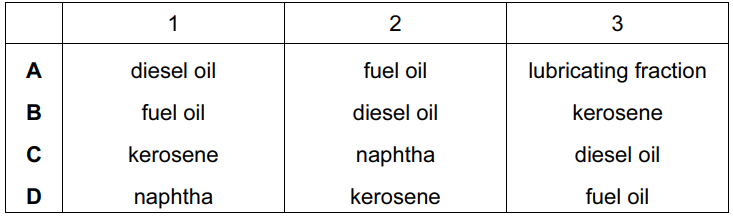
▶️ Answer/Explanation
Ans: B
In fractional distillation of petroleum:
1. Fraction 1 (used in jet fuel) corresponds to kerosene (intermediate boiling point).
2. Fraction 2 (used in road surfacing) is bitumen (highest boiling point).
3. Fraction 3 (used in bottled gas) is refinery gas (lowest boiling point).
Thus, the correct identification is B (kerosene, bitumen, refinery gas).
Petroleum is a mixture of hydrocarbons which can be separated into fractions using fractional distillation.
Which fraction is used as fuel in jet engines?
- bitumen
- gasoline
- kerosene
- naphtha
▶️ Answer/Explanation
Ans: C
In fractional distillation of petroleum:
1. Kerosene (Fraction C) is used as jet fuel due to its high energy output and stability at high altitudes.
2. Bitumen (A) is too viscous, used for roads.
3. Gasoline (B) fuels cars, not jets.
4. Naphtha (D) is a lighter fraction used in chemicals, not aviation.
Thus, the correct answer is C) kerosene.
A solid mixture contains an ionic salt, X, and a covalent organic compound, Y. Two students suggest methods of separating the mixture as shown.
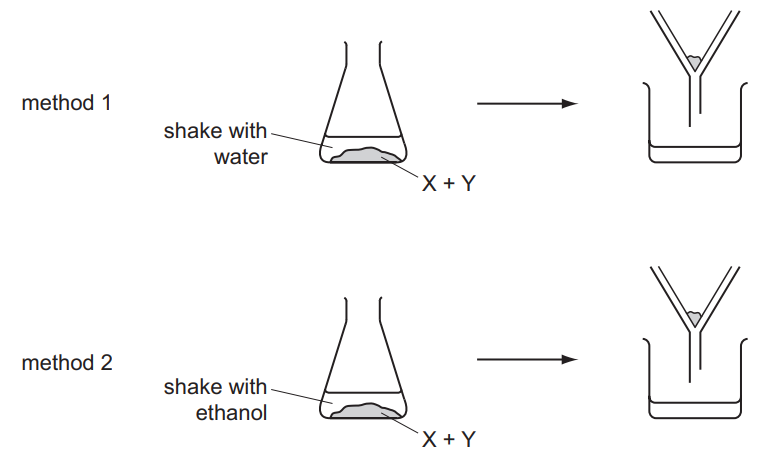
Which methods of separation are likely to work?
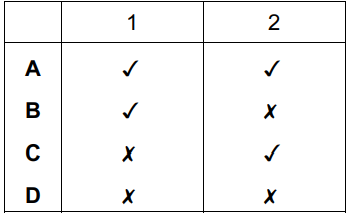
▶️ Answer/Explanation
Ans: A
1. Student 1’s Method: Dissolving in water separates ionic salt X (soluble) from covalent compound Y (insoluble). Filtration isolates Y, and evaporation recovers X. This works.
2. Student 2’s Method: Sublimation requires Y to sublime (transition directly from solid to gas), which is unlikely for most covalent organic solids. Thus, this method fails.
Only Student 1’s method is viable, making option A correct.
Which properties of the different compounds in petroleum enable its separation into fractions?
- 1. boiling point
- 2. chain length
- 3. chemical reactivity
- 4. solubility in water
A) 1 and 2
B) 1 and 3
C) 2 and 4
D) 3 and 4
▶️ Answer/Explanation
Ans: A
Petroleum is separated into fractions via fractional distillation, which relies on differences in:
1. Boiling point: Different hydrocarbons condense at different temperatures in the fractionating column.
2. Chain length: Longer hydrocarbon chains have higher boiling points, allowing separation.
Chemical reactivity (3) and solubility in water (4) are irrelevant to this physical separation process. Thus, the correct properties are 1 and 2 (Option A).
Bitumen is a substance obtained from the fractional distillation of petroleum.
Which row describes its boiling point and the size of its molecules?
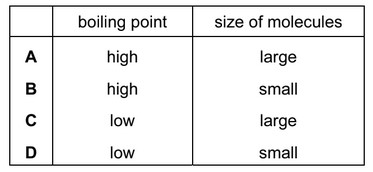
▶️ Answer/Explanation
Ans: A
In fractional distillation of petroleum:
1. Bitumen is obtained at the bottom of the fractionating column, meaning it has the highest boiling point (option 1 in the table).
2. As the heaviest fraction, bitumen consists of very large molecules (option 1 in the table).
3. This matches row A in the given options.
4. The other rows incorrectly describe bitumen’s properties (B: small molecules, C: low boiling point, D: small molecules with high boiling point).
Pieces of copper, iron, magnesium and zinc are added to separate test-tubes containing dilute hydrochloric acid.
Which test-tube contains iron and dilute hydrochloric acid?
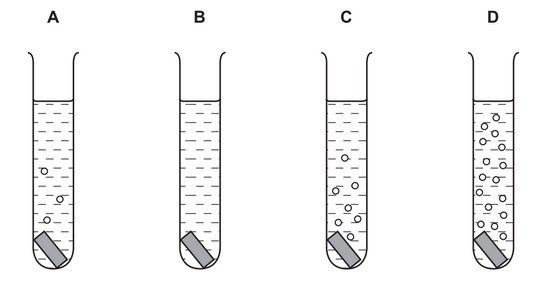
▶️ Answer/Explanation
Ans: A
To identify the test-tube containing iron:
- Reactivity analysis: The metals react with HCl in this order of vigor: Mg > Zn > Fe > Cu (copper doesn’t react)
- Observation matching:
- Tube A shows moderate bubbling (matches iron’s reaction)
- Tube B shows no reaction (copper)
- Tube C shows vigorous reaction (magnesium)
- Tube D shows fast but less vigorous than C (zinc)
The moderate reaction rate in Tube A perfectly matches iron’s reactivity with HCl.
The diagram shows apparatus used to separate petroleum into four fractions.
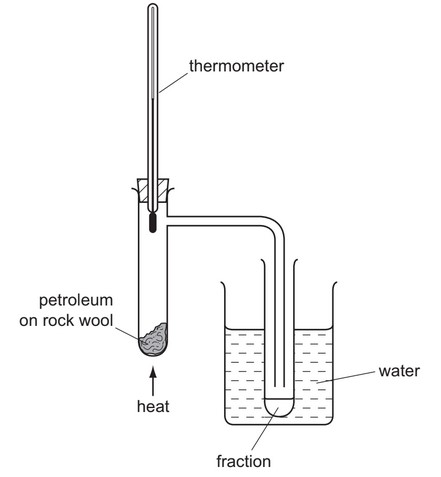
Which fraction contains the smallest hydrocarbon molecules?
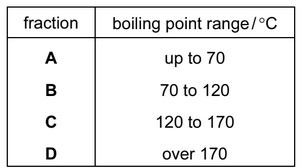
▶️ Answer/Explanation
Ans: A
The fraction with the smallest hydrocarbon molecules is A (refinery gases) because:
1. Petroleum is separated by fractional distillation based on boiling points.
2. Smaller hydrocarbon molecules have:
– Lower boiling points (condense at the top of the fractionating column)
– Fewer carbon atoms (typically C₁-C₄ for refinery gases)
3. The diagram shows fraction A being collected first at the top of the column.
4. Larger molecules (B, C, D) condense lower down as they have higher boiling points.
Which fraction from the fractional distillation of petroleum does not match its correct use?

▶️ Answer/Explanation
Ans: C
In petroleum fractional distillation:
1. Refinery gas (A) is correctly used for bottled gas.
2. Gasoline (B) is properly matched with fuel for cars.
3. Kerosene (C) is incorrectly matched with lubricants – it’s actually used for jet fuel and heating.
4. Bitumen (D) is correctly used for road surfacing.
Thus, option C shows the incorrect pairing as lubricants are typically derived from heavier fractions.
