Pre AP Chemistry -2.2A Intermolecular Forces- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2A Intermolecular Forces- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2A Intermolecular Forces- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
In which substance are the only intermolecular forces temporary dipole-induced dipole attractions?
A hydrogen chloride
B methanol
C octane
D water
▶️ Answer/Explanation
Step 1: Recall types of intermolecular forces
- Hydrogen bonding – occurs when H is bonded to N, O or F.
- Permanent dipole–dipole forces – occur in polar molecules.
- Temporary dipole-induced dipole forces (London forces) – occur in all molecules but are the only force present in non-polar molecules.
Step 2: Analyse each substance
- Hydrogen chloride (HCl) – polar molecule → permanent dipole-dipole forces present.
- Methanol (CH3OH) – contains O–H bond → hydrogen bonding present.
- Octane (C8H18) – non-polar hydrocarbon → only London dispersion (temporary dipole-induced dipole) forces.
- Water (H2O) – strong hydrogen bonding between molecules.
Step 3: Identify the correct substance
Only octane has temporary dipole-induced dipole attractions as the only intermolecular forces.
Ans: C
In which of the following, when in liquid form, are there only intermolecular forces based on temporary dipoles between the particles?
A bromine
B ethanol
C hydrogen chloride
D water
▶️ Answer/Explanation
Step 1: Recall intermolecular forces
- Temporary dipole–induced dipole forces (London dispersion) occur in all molecules but are the only forces present in non-polar molecules.
- Hydrogen bonding occurs when hydrogen is bonded to N, O or F.
- Permanent dipole–dipole forces occur in polar molecules.
Step 2: Analyse each substance
- Bromine (Br2) – non-polar molecule → only temporary dipole-induced dipole forces.
- Ethanol (C2H5OH) – contains O–H bond → hydrogen bonding present.
- Hydrogen chloride (HCl) – polar molecule → permanent dipole–dipole interactions.
- Water (H2O) – strong hydrogen bonding between molecules.
Step 3: Identify the correct answer
Only bromine has temporary dipole-induced dipole forces as the only intermolecular forces.
Ans: A
Copper has a high melting point.
What is the reason for the high melting point of copper?
A strong attractive forces between copper atoms only
B strong attractive forces between copper ions and delocalised electrons
C strong attractive forces between copper ions only
D strong attractive forces between copper atoms and delocalised electrons
▶️ Answer/Explanation
Step 1: Understand metallic bonding
In metals, atoms lose outer electrons to form a lattice of positive metal ions surrounded by a sea of delocalised electrons.
Step 2: Nature of the attractive force
There is a strong electrostatic attraction between:
- positive metal ions
- delocalised electrons
This strong attraction requires a large amount of energy to overcome.
Step 3: Effect on melting point
Because the metallic bonding is strong, a high temperature is needed to break these interactions, giving copper a high melting point.
Ans: B
The strong hydrogen bonding present in liquid water causes an increase in which properties?
1 viscosity
2 boiling point
3 surface tension

▶️ Answer/Explanation
Step 1: Understand hydrogen bonding
Hydrogen bonding occurs when hydrogen is bonded to highly electronegative atoms such as oxygen, and there is attraction between molecules.
In water, extensive hydrogen bonding forms a network between molecules.
Step 2: Effect on physical properties
- Viscosity increases because molecules stick together more strongly.
- Boiling point increases because more energy is required to separate molecules.
- Surface tension increases due to strong intermolecular attraction at the liquid surface.
Step 3: Identify correct statements
All three properties increase due to hydrogen bonding.
Ans: A (1, 2 and 3)
The boiling points of methane, ethane, propane and butane are given.
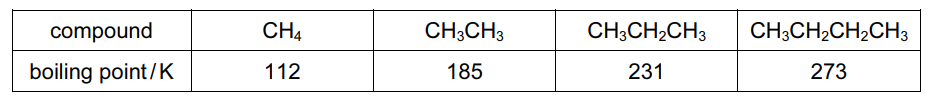
Which statement explains the increase in boiling point from methane to butane?
A Closer packing of molecules results in stronger van der Waals’ forces.
B More covalent bonds are present and therefore more energy is required to break the bonds.
C More electrons in the molecules results in stronger van der Waals’ forces.
D More hydrogen atoms in the molecules results in stronger hydrogen bonding.
▶️ Answer/Explanation
Step 1: Identify the intermolecular forces
Methane, ethane, propane and butane are non-polar molecules. The only intermolecular forces present are temporary dipole-induced dipole forces (London dispersion forces).
Step 2: Effect of increasing molecular size
From methane to butane:
- The number of electrons increases.
- The electron cloud becomes more easily polarised.
- This strengthens van der Waals’ forces between molecules.
Step 3: Result on boiling point
Stronger intermolecular attractions require more energy to overcome, so the boiling point increases.
Ans: C
Which molecules have an overall dipole moment?
1 carbon monoxide, CO
2 phosphine, PH3
3 carbon dioxide, CO2
▶️ Answer/Explanation
Step 1: Recall dipole moment concept
A molecule has an overall dipole moment if:
- it contains polar bonds, and
- the bond dipoles do not cancel due to molecular geometry.
Step 2: Analyse each molecule
- CO: A polar diatomic molecule → has a dipole moment.
- PH3: Trigonal pyramidal shape due to a lone pair on P → dipoles do not cancel → molecule is polar.
- CO2: Linear molecule → bond dipoles cancel → no overall dipole moment.
Step 3: Identify correct molecules
Molecules with an overall dipole moment: 1 and 2.
Ans: B
Elements X, Y and Z are all in the first two periods of the Periodic Table. Their Pauling electronegativity values, \(E_N\), are shown.
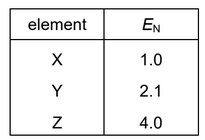
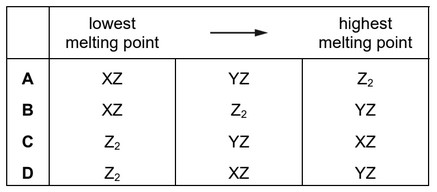
Substances exist with formulae XZ, YZ and Z2.
Which row puts these substances in order of increasing melting point?
▶️ Answer/Explanation
Step 1: Identify the elements
- X has electronegativity ≈ 1.0 → similar to Li
- Y has electronegativity ≈ 2.1 → similar to H
- Z has electronegativity ≈ 4.0 → similar to F
Step 2: Determine bonding types
- Z2 → F2, a non-polar molecule with weak London forces → very low melting point.
- YZ → HF, polar molecule with strong hydrogen bonding → higher melting point.
- XZ → LiF, an ionic compound with strong electrostatic attraction → very high melting point.
Step 3: Arrange in increasing melting point
Z2 < YZ < XZ
This corresponds to option C.
Ans: C
The boiling points of some hydrogen halides are shown.
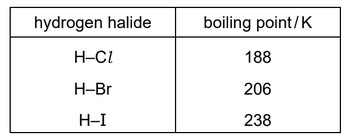
What is the explanation for the trend in boiling point for the hydrogen halides from HCl to HI?
A The bond energies of the hydrogen halides increase from HCl to HI.
B There is an increase in the strength of the intermolecular forces of attraction from HCl to HI.
C The intermolecular hydrogen bonds become stronger from HCl to HI.
D There is an increase in the bond polarity from HCl to HI.
▶️ Answer/Explanation
Step 1: Identify intermolecular forces
HCl, HBr and HI are polar molecules but they do not form hydrogen bonds.
The main intermolecular forces present are:
- permanent dipole–dipole interactions
- London dispersion forces
Step 2: Effect of increasing atomic size
From Cl → Br → I:
- the atoms become larger
- the number of electrons increases
- the electron cloud becomes more easily polarised
This strengthens London dispersion forces.
Step 3: Result on boiling point
Stronger intermolecular attractions require more energy to overcome, so the boiling point increases from HCl → HBr → HI.
Ans: B
For which pair is the boiling point of the first compound higher than the boiling point of the second compound?
A \(CH_3CH_2OH\) and \(CH_3CH_2SH\)
B \(CH_3CO_2CH_3\) and \(CH_3CH_2CO_2H\)
C \(CH_3OCH_3\) and \(CH_3CH_2OH\)
D \(CH_3CH_2CHO\) and \(CH_3CH_2CO_2H\)
▶️ Answer/Explanation
Step 1: Compare intermolecular forces.
- A \(CH_3CH_2OH\) (ethanol) forms strong hydrogen bonds because of the O–H group. \(CH_3CH_2SH\) (ethanethiol) has weaker intermolecular forces since S–H hydrogen bonding is much weaker.
Therefore ethanol has a higher boiling point than ethanethiol.
Step 2: Check the other options.
- B Carboxylic acids form strong hydrogen-bonded dimers, so \(CH_3CH_2CO_2H\) has a higher boiling point than the ester.
- C Ethanol forms hydrogen bonds while dimethyl ether cannot, so ethanol has the higher boiling point.
- D Carboxylic acids again have strong hydrogen bonding, so \(CH_3CH_2CO_2H\) has a higher boiling point.
Only option A satisfies the condition that the first compound has the higher boiling point.
Ans: A
Which type of interaction exists between water molecules and metal cations in aqueous solution?
A dipole–dipole interactions
B hydrogen bonds
C ion–dipole interactions
D ionic bonds
▶️ Answer/Explanation
Step 1: Nature of water molecules
Water (H2O) is a polar molecule with a permanent dipole.
The oxygen atom carries a partial negative charge (δ−), while the hydrogen atoms carry partial positive charges (δ+).
Step 2: Interaction with metal cations
Metal cations (e.g., Na+, Mg2+) have a positive charge.
The negative end of the water molecule (oxygen) is attracted to the positive ion.
Step 3: Type of interaction
This attraction between an ion and a polar molecule is called an ion–dipole interaction.
Ans: C
Liquids X and Y do not react with one another. They have identical boiling points.
When a particular volume of X is shaken with a similar volume of Y, they form a liquid mixture Z. The average intermolecular forces in liquid Z are stronger than the average of the forces in X and the forces in Y.
Which deductions from this information are correct?
1 The mixing of X and Y is exothermic.
2 The vapour pressure of liquid Z will be less than that of either liquid X or liquid Y at the same temperature.
3 The boiling point of liquid Z will be lower than that of either liquid X or liquid Y at the same pressure.
▶️ Answer/Explanation
Step 1: Effect of stronger intermolecular forces
If the intermolecular forces between X and Y in mixture Z are stronger than those in the pure liquids, energy is released when the molecules attract each other.
Therefore, the mixing process is exothermic.
✔ Statement 1 is correct.
Step 2: Effect on vapour pressure
Stronger intermolecular forces hold molecules more tightly in the liquid phase.
This makes it harder for molecules to escape into the vapour phase, so the vapour pressure decreases.
✔ Statement 2 is correct.
Step 3: Effect on boiling point
Stronger intermolecular forces require more energy to overcome.
This causes the boiling point to increase, not decrease.
✖ Statement 3 is incorrect.
Correct statements: 1 and 2
Ans: B
What is the oxidation number of sulfur in each species?
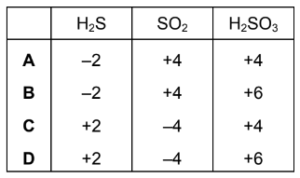
▶️ Answer/Explanation
Step 1: Apply oxidation number rules
- Hydrogen usually has oxidation number +1.
- Oxygen usually has oxidation number −2.
- The sum of oxidation numbers in a neutral molecule equals 0.
Step 2: Calculate for each compound
H₂S
2(+1) + S = 0
S = −2
SO₂
S + 2(−2) = 0
S = +4
H₂SO₃
2(+1) + S + 3(−2) = 0
2 + S − 6 = 0
S = +4
Step 3: Compare with the table
The correct set is:
H₂S = −2
SO₂ = +4
H₂SO₃ = +4
Ans: A
