Pre AP Chemistry -2.2B Intermolecular Forces and Physical Properties- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2B Intermolecular Forces and Physical Properties- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2B Intermolecular Forces and Physical Properties- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Ethanal, CH3CHO, ethanol, C2H5OH, and methoxymethane, CH3OCH3, are three organic compounds.
Which compound has the highest boiling point and what is the interaction that causes this boiling point to be the highest?
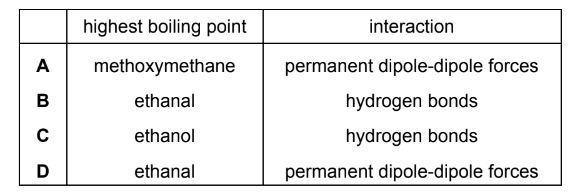
▶️ Answer/Explanation
Step 1: Identify intermolecular forces in each compound
- Ethanal (CH3CHO) → dipole–dipole forces
- Methoxymethane (CH3OCH3) → dipole–dipole forces
- Ethanol (C2H5OH) → hydrogen bonding
Step 2: Compare intermolecular force strength
Hydrogen bonding is stronger than dipole–dipole interactions. Stronger intermolecular forces require more energy to separate molecules, resulting in a higher boiling point.
Step 3: Determine the compound with the highest boiling point
Ethanol forms hydrogen bonds due to the O–H group, so it has the strongest intermolecular forces among the three compounds.
Conclusion:
Ethanol has the highest boiling point because of hydrogen bonding.
Ans: C
Which statements are correct?
1. The hydrogen bonds in ice are more regularly arranged than in water.
2. The solidification of water to form ice is exothermic.
3. Pure water is less dense than ice.
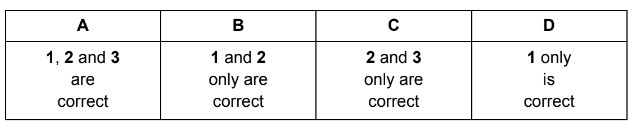
▶️ Answer/Explanation
Statement 1: True.
In ice, hydrogen bonds form a fixed and highly ordered lattice structure. In liquid water, the hydrogen bonds constantly break and reform, making the structure less regular.
Statement 2: True.
When water freezes to form ice, heat is released to the surroundings. Therefore, freezing (solidification) is an exothermic process.
Statement 3: False.
Ice is less dense than liquid water. This is why ice floats on water.
Correct statements: 1 and 2 only.
Ans: B
Which molecule has no overall dipole?
A. CH3Cl B. CH2Cl2 C. CHCl3 D. CCl4
▶️ Answer/Explanation
Step 1: Consider molecular geometry
All these molecules have a tetrahedral arrangement around the carbon atom.
Step 2: Check symmetry
- CH3Cl → different atoms around carbon → dipoles do not cancel → polar.
- CH2Cl2 → two Cl and two H → asymmetric → polar.
- CHCl3 → three Cl and one H → asymmetric → polar.
- CCl4 → four identical Cl atoms arranged symmetrically → dipoles cancel.
Step 3: Determine overall dipole
Because CCl4 is perfectly symmetrical, its bond dipoles cancel, giving no overall dipole.
Ans: D (CCl4)
Which compound has a boiling point that is influenced by hydrogen bonding?
A. CH3CHO B. CH3OCH3 C. HCO2CH3 D. HCO2H
▶️ Answer/Explanation
Step 1: Requirement for hydrogen bonding
Hydrogen bonding occurs when hydrogen is directly bonded to highly electronegative atoms such as O, N, or F.
Step 2: Check each compound
- CH3CHO – contains C=O but no O–H bond → cannot form hydrogen bonds between its own molecules.
- CH3OCH3 – contains oxygen but no O–H bond → no hydrogen bonding between molecules.
- HCO2CH3 – ester group → no O–H bond → no hydrogen bonding.
- HCO2H – contains an O–H group → forms strong hydrogen bonds.
Step 3: Conclusion
Formic acid (HCO2H) forms strong intermolecular hydrogen bonds, increasing its boiling point.
Ans: D (HCO2H)
Which molecule has the largest overall dipole?
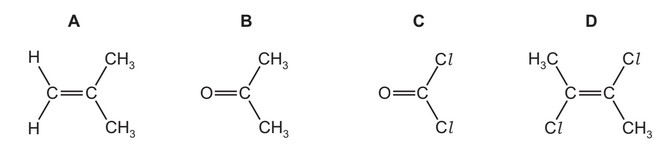
▶️ Answer/Explanation
Step 1: Consider bond polarity
The polarity of a molecule depends on the polarity of its bonds and the geometry of the molecule. If the molecule is symmetrical, the bond dipoles may cancel.
Step 2: Analyse each structure
- A: The substituents are arranged fairly symmetrically around the C=C bond, causing partial cancellation of dipoles.
- B: The molecule contains a strongly polar C=O bond and two CH3 groups that do not cancel the dipole. This gives a large net dipole moment.
- C: Although C–Cl bonds are polar, the two identical chlorine atoms tend to partially cancel each other due to the molecular geometry.
- D: The C–Cl dipoles oppose each other across the double bond, reducing the overall dipole.
Step 3: Determine the largest dipole
Because the strong C=O dipole is not cancelled by symmetry, molecule B has the largest overall dipole.
Ans: B
At room temperature and pressure, H2O is a liquid and H2S is a gas.
What is the reason for this difference?
A. O has higher first and second ionisation energies than S.
B. The covalent bond between O and H is stronger than the covalent bond between S and H.
C. There is significant hydrogen bonding between H2O molecules but not between H2S molecules.
D. The instantaneous dipole-induced dipole forces between H2O molecules are stronger than the instantaneous dipole-induced dipole forces between H2S molecules.
▶️ Answer/Explanation
Step 1: Identify intermolecular forces
H2O molecules contain an O–H bond where hydrogen is bonded to a highly electronegative oxygen atom. This allows strong hydrogen bonding between water molecules.
Step 2: Compare with H2S
In H2S, hydrogen is bonded to sulfur. Sulfur is less electronegative than oxygen, so hydrogen bonding does not occur effectively. Only weaker intermolecular forces act between H2S molecules.
Step 3: Effect on physical state
Hydrogen bonding greatly increases intermolecular attraction, raising the boiling point of water so that it exists as a liquid at room temperature. H2S lacks these strong interactions and remains a gas.
Ans: C
Nitrogen, N2, and carbon monoxide, CO, both have Mr = 28.
The boiling point of N2 is 77 K. The boiling point of CO is 82 K.
What could be responsible for this difference in boiling points?
A. CO molecules have a permanent dipole, the N2 molecules are not polar.
B. N2 has σ and π bonding, CO has σ bonding only.
C. N2 has a strong N≡N bond, CO has a C=O bond.
D. The CO molecule has more electrons than the N2 molecule.
▶️ Answer/Explanation
Step 1: Compare molecular polarity
N2 is a homonuclear molecule (two identical nitrogen atoms), so it is completely non-polar and has no permanent dipole moment.
CO is a heteronuclear molecule (carbon bonded to oxygen). Because oxygen is more electronegative than carbon, the molecule has a small permanent dipole.
Step 2: Consider intermolecular forces
N2 molecules interact only through weak London dispersion forces.
CO molecules experience both dipole–dipole attractions and dispersion forces.
Step 3: Effect on boiling point
Stronger intermolecular forces in CO require slightly more energy to separate the molecules, so CO has a slightly higher boiling point.
Ans: A
P and Q are two liquid compounds with similar Mr values.
Molecules of P attract each other by hydrogen bonds. Molecules of Q attract each other by van der Waals’ forces only.
How do the properties of P and Q differ?
1 P has a higher surface tension than Q.
2 P is less soluble in water than Q.
3 P has a lower melting point than Q.
The responses A to D should be selected on the basis of

▶️ Answer/Explanation
Step 1: Compare intermolecular forces
Hydrogen bonding is much stronger than van der Waals’ (London dispersion) forces.
Step 2: Evaluate statement 1
Stronger intermolecular forces increase surface tension. Therefore, P (hydrogen bonding) has higher surface tension than Q. ✔ Statement 1 is correct.
Step 3: Evaluate statement 2
Compounds capable of hydrogen bonding generally dissolve well in water because water itself forms hydrogen bonds. Therefore, P is more soluble in water than Q. ✘ Statement 2 is incorrect.
Step 4: Evaluate statement 3
Stronger intermolecular forces increase melting point. Since P has hydrogen bonding, it should have a higher melting point, not lower. ✘ Statement 3 is incorrect.
Correct statements: 1 only.
Ans: D
Pollutant oxide Y, which contains non-metallic element X, is formed in a car engine. Further oxidation of Y to Z occurs in the atmosphere. In this further oxidation, 1 mol of Y reacts with 0.5 mol of gaseous oxygen molecules.
X could be either nitrogen or sulfur.
Which statements about X, Y and Z can be correct?
1 The oxidation number of X increases by two from Y to Z.
2 Y has an unpaired electron in its molecule.
3 Y is a polar molecule.
The responses A to D should be selected on the basis of

▶️ Answer/Explanation
Step 1: Identify Y and Z
In car engines, nitrogen monoxide (NO) is commonly formed. It can further oxidise in air:
2 NO + O2 → 2 NO2
Dividing by 2:
NO + 0.5 O2 → NO2
Therefore:
Y = NO and Z = NO2.
Step 2: Check oxidation number change
In NO:
O = −2 → N = +2
In NO2:
2O = −4 → N = +4
The oxidation number of nitrogen increases from +2 to +4 (increase of 2).
✔ Statement 1 is correct.
Step 3: Check for unpaired electron
NO has an odd number of electrons (11 valence electrons), so it contains an unpaired electron.
✔ Statement 2 is correct.
Step 4: Check polarity
NO is a heteronuclear diatomic molecule (N and O have different electronegativities), so the molecule has a dipole moment.
✔ Statement 3 is correct.
Conclusion:
Statements 1, 2 and 3 are correct.
Ans: A
Which molecular structure will have the smallest overall dipole?
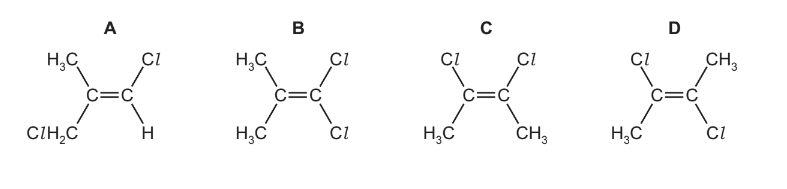
▶️ Answer/Explanation
Step 1: Understand molecular dipole
The overall dipole moment depends on the vector sum of individual bond dipoles. If the molecule is arranged symmetrically, the dipoles cancel and the net dipole becomes very small.
Step 2: Compare structures
- Structures A, B, and C have asymmetric arrangements of substituents, so the C–Cl bond dipoles do not cancel effectively.
- Structure D has substituents arranged in a more symmetrical pattern across the C=C bond, causing the dipoles to oppose each other.
Step 3: Determine smallest dipole
Because of this symmetry, the dipole moments cancel to the greatest extent in structure D, giving the smallest overall dipole.
Ans: D
