Pre AP Chemistry -2.2C Periodic Trends- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2C Periodic Trends- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2C Periodic Trends- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which option describes the electronic configurations of three different elements from the same group of the Periodic Table?
A) 2 2,2 2,8,8,2
B) 2 2,8 2,8,2
C) 2,1 2,8,1 2,8,8,1
D) 2,1 2,2 2,3
▶️ Answer/Explanation
Ans: C
Elements in the same group have the same number of valence electrons. Let’s analyze each option:
- Option A: Shows configurations with 2, 2, and 2 valence electrons respectively – but the second element (2,2) is in Group 2 while the third (2,8,8,2) is in Group 2 but the first (2) is helium (Group 18).
- Option B: Shows configurations with 2, 8, and 2 valence electrons – different numbers, so not same group.
- Option C: All configurations end with 1 valence electron (2,1; 2,8,1; 2,8,8,1) – these are all Group 1 elements (alkali metals).
- Option D: Shows 1, 2, and 3 valence electrons respectively – different groups.
Only option C shows three elements all with 1 valence electron, meaning they’re in the same group (Group 1).
Which statement about the Periodic Table is correct?
A) All the metals in the Periodic Table are transition elements.
B) The halogens are elements in Group I of the Periodic Table.
C) The elements become more metallic across a period from Group I to Group VII.
D) The Periodic Table can be used to predict the properties of the elements.
▶️ Answer/Explanation
Ans: D
Let’s evaluate each option:
- A: Incorrect – There are many metals (like alkali and alkaline earth metals) that aren’t transition elements.
- B: Incorrect – Halogens are in Group VII, not Group I.
- C: Incorrect – Elements become less metallic (more non-metallic) across a period from left to right.
- D: Correct – The Periodic Table organizes elements by atomic number and shows periodic trends in properties, allowing predictions about element behavior.
The Periodic Table’s main purpose is to show periodic trends and allow prediction of element properties based on position.
Which compound is made from elements that are all in the same period?
A) Al2(SO4)3
B) C2H5OH
C) LiNO3
D) Na3AlF6
▶️ Answer/Explanation
Ans: C
To solve this, we need to check which compound contains elements all from the same period (row) in the Periodic Table.
Option A: Al (Period 3), S (Period 3), O (Period 2) – different periods
Option B: C (Period 2), H (Period 1), O (Period 2) – different periods
Option C: Li (Period 2), N (Period 2), O (Period 2) – all same period
Option D: Na (Period 3), Al (Period 3), F (Period 2) – different periods
Only LiNO3 contains elements that are all in Period 2.
Part of the Periodic Table is shown.
Which element is a metal?

▶️ Answer/Explanation
Ans: A
From the periodic table shown in the question (not visible in text), we can deduce:
1. Metals are typically found on the left side and middle of the periodic table.
2. Non-metals are found on the right side.
3. The question indicates that element A is a metal, which would be positioned in the metal region of the periodic table.
Therefore, the correct answer is option A.
Four mixtures each contain a halogen and an aqueous sodium halide. Which row describes what happens in the mixtures shown?

▶️ Answer/Explanation
Ans: C
1. Halogen Reactivity Series:
F > Cl > Br > I (decreasing oxidizing power)
A halogen can displace any halide below it in the series 2. Key Observations:
• Cl2 + 2NaBr → 2NaCl + Br2 (orange solution)
• Br2 + 2NaI → 2NaBr + I2 (brown solution)
• No reaction when less reactive halogen is added 3. Color Changes:
• Chlorine water: Pale yellow-green
• Bromine solution: Orange
• Iodine solution: Brown 4. Why Option C:
Correctly identifies both successful displacement reactions where the added halogen is more reactive than the halide present
Which statements about elements in the Periodic Table are correct?
- Elements in the same group have the same number of electrons in their outer shell.
- Elements in the same period have the same number of occupied electron shells.
- The elements are arranged in order of their atomic mass.
- Every period contains eight elements.
A 1 and 2
B 1 and 3
C 2 and 4
D 3 and 4
▶️ Answer/Explanation
Ans: A
Analysis of each statement about Periodic Table organization:
| Statement | Evaluation | Explanation |
|---|---|---|
| 1 | Correct | Group number = valence electrons (main group elements) |
| 2 | Correct | Period number = principal quantum number (n) of outermost shell |
| 3 | Incorrect | Arranged by atomic number (protons), not mass |
| 4 | Incorrect | Period lengths vary (2, 8, 8, 18, 18, 32, 32 elements) |
Key periodic trends:
- Groups (vertical):
- Same valence electron configuration
- Similar chemical properties
- Periods (horizontal):
- Same number of electron shells
- Properties change progressively
Therefore, only statements 1 and 2 are correct, making option A the right answer.
Which statements describe changes that occur from left to right across a period of the Periodic Table?
1 The atomic number of the elements increases.
2 The metallic character of the elements decreases.
3 The physical state of the elements changes from gas to solid.
A 2 only
B 1 and 2
C 1 and 3
D 2 and 3
▶️ Answer/Explanation
Ans: B (1 and 2)
Periodic trends analysis:
- Statement 1 (Correct):
- Atomic number increases by 1 across each period
- This is the fundamental organization principle of the Periodic Table
- Statement 2 (Correct):
- Metallic character decreases left to right
- Elements change from metals → metalloids → nonmetals
- Due to increasing electronegativity and ionization energy
- Statement 3 (Incorrect):
- Physical state doesn’t follow a consistent gas→solid pattern
- Example: Period 2 starts with solid Li, ends with gas Ne
- Physical state depends on specific elements, not periodic position
Therefore, only statements 1 and 2 correctly describe periodic trends.
Which row about elements in the Periodic Table is correct?
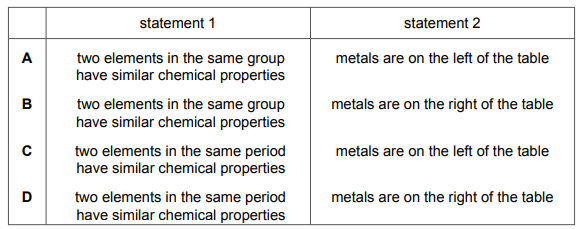
▶️ Answer/Explanation
Ans: A
Analysis of each periodic table property:
| Option | Evaluation | Scientific Basis |
|---|---|---|
| A | Correct |
|
| B | Incorrect | Non-metals are poor conductors, not good |
| C | Incorrect | Metals form basic oxides, not acidic |
| D | Incorrect | Group 0 elements are unreactive, not very reactive |
Key periodic table facts:
- Metal properties:
- Malleable and ductile
- Form basic oxides (e.g., Na2O, CaO)
- Good conductors of heat/electricity
- Non-metal properties:
- Brittle when solid
- Form acidic oxides (e.g., SO3, P4O10)
- Poor conductors (except graphite)
- Noble gases:
- Group 0/18 elements
- Extremely low reactivity (full outer electron shells)
- Monatomic gases at room temperature
The image shows Option A as the only completely correct description of these periodic table properties.
Which statement about the Periodic Table is correct?
A Elements with the highest atomic number in each period are metallic.
B Elements with the lowest group numbers are non-metals.
C Elements with similar chemical properties are placed in groups.
D Elements with similar physical properties are placed in periods.
▶️ Answer/Explanation
Ans: C
1. Periodic Table Organization:
– Groups (vertical columns) contain elements with similar chemical properties due to identical valence electrons
– Periods (horizontal rows) show trends in physical properties
2. Evaluating Options:
– A: Incorrect – Noble gases (Group 18) are non-metallic
– B: Incorrect – Groups 1-2 contain highly reactive metals
– C: Correct – Fundamental principle of periodic table organization
– D: Incorrect – Physical properties change gradually across periods
The positions of four elements in the Periodic Table are shown.
Which element is a gas that displaces iodine from sodium iodide?

▶️ Answer/Explanation
Ans: C (Chlorine)
Key analysis:
- Displacement reaction requirement:
Cl2(g) + 2NaI(aq) → 2NaCl(aq) + I2(aq)
- Only chlorine gas (Cl2) can displace iodine from NaI
- This demonstrates chlorine’s higher reactivity in Group 17
- Periodic Table positions:
- Chlorine is above iodine in Group 17 (halogens)
- Higher halogens can displace lower ones from their salts
- Why other options are incorrect:
- A: Too reactive (alkali metal)
- B: Noble gas – doesn’t react
- D: Less reactive than iodine (can’t displace it)
Key points:
- Chlorine is the only gas among the options
- It’s more electronegative than iodine (can oxidize I– to I2)
- The reaction shows a classic halogen displacement
Therefore, Option C (chlorine) is the correct answer.
The elements in Period 3 of the Periodic Table are shown.

Which statements about the elements in Period 3 are correct?
- 1. Na, Mg and Al are metals.
- 2. S, Cl and Ar are non-metals.
- 3. Si, P and S are metals.
A. 1 and 2 only
B. 1 and 3 only
C. 2 and 3 only
D. 1, 2 and 3
▶️ Answer/Explanation
Ans: A
Analysis of Period 3 elements:
| Element Group | Classification | Notes |
|---|---|---|
| Na, Mg, Al | Metals | Conduct electricity, malleable |
| Si | Metalloid | Semiconductor properties |
| P, S, Cl, Ar | Non-metals | Poor conductors, brittle |
Evaluation of statements:
- Correct: Na, Mg, Al are all metals
- Correct: S, Cl, Ar are non-metals
- Incorrect: Si is a metalloid, P and S are non-metals
Therefore, only statements 1 and 2 are correct, making option A the right answer.
Part of the Periodic Table is shown.
Which element forms an acidic oxide?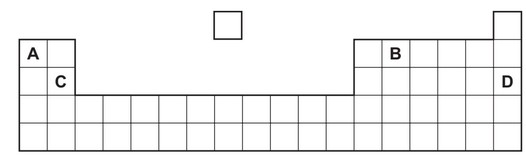
▶️ Answer/Explanation
Ans: B
Periodic Trends Analysis:
- Element Identification:
- B is carbon (C) – Group 14 nonmetal
- Forms CO2 and CO (both acidic oxides)
- Acidic Oxide Characteristics:
- React with water to form acids (CO2 + H2O → H2CO3)
- React with bases to form salts
- Typical of nonmetal oxides
- Other Elements:
- A (Al) – Forms amphoteric oxide (Al2O3)
- C (Mg) – Forms basic oxide (MgO)
- D (Na) – Forms strongly basic oxide (Na2O)
Chemical Evidence:
- CO2 turns limewater milky (acidic property test)
- Forms carbonic acid in water (pH ≈ 4)
- Reacts with NaOH: CO2 + 2NaOH → Na2CO3 + H2O
Therefore, only B (carbon) forms a definitively acidic oxide among the options.
Which statements describe the Periodic Table?
1 The elements are arranged in order of their nucleon number.
2 The elements are arranged in order of their proton number.
3 It is used to predict the properties of elements.
A 1 and 3 B 1 only C 2 and 3 D 2 only
▶️ Answer/Explanation
Ans: C
Statement Analysis
Statement 1: “Elements arranged by nucleon number”
- False – Nucleon number (mass number) is not the basis
- Example: Tellurium (atomic number 52) comes before iodine (53) despite having higher mass
Statement 2: “Elements arranged by proton number”
- True – Atomic number (proton count) determines position
- Fundamental organizing principle since Moseley’s work (1913)
Statement 3: “Used to predict properties”
- True – Periodic trends allow prediction of:
- Reactivity (e.g., Group 1 metals get more reactive down group)
- Melting points (e.g., higher for metals in middle of d-block)
- Ionization energies (periodic pattern across periods)
| Option | Evaluation |
|---|---|
| A (1 and 3) | Incorrect – Statement 1 is false |
| B (1 only) | Incorrect – Statement 1 is false |
| C (2 and 3) | Correct – Both statements true |
| D (2 only) | Incomplete – Misses predictive power |
Conclusion: The Periodic Table is arranged by proton number (atomic number) and enables property prediction through periodic trends, making Option C correct.
A period of the Periodic Table is shown.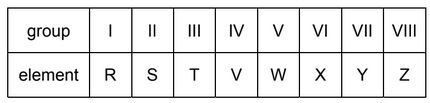
The letters are not their chemical symbols.
Which statement is correct?
A Element R does not conduct electricity.
B Elements R and Y react together to form an ionic compound.
C Element Z exists as a diatomic molecule.
D Element Z reacts with element T.
▶️ Answer/Explanation
Ans: B (Elements R and Y react together to form an ionic compound)
Periodic Table Analysis:
The elements shown represent a typical period with:
- R: Alkali metal (Group 1)
- S: Alkaline earth metal (Group 2)
- T: Transition metal (Groups 3-12)
- U: Metalloid
- V: Non-metal
- W: Non-metal
- X: Non-metal
- Y: Halogen (Group 17)
- Z: Noble gas (Group 18)
Evaluation of Statements:
- A: Incorrect – R (alkali metal) conducts electricity
- B: Correct – Alkali metal (R) + Halogen (Y) → Ionic compound (e.g., NaCl)
- C: Incorrect – Z (noble gas) exists as monatomic, not diatomic
- D: Incorrect – Z (noble gas) is inert and doesn’t react with T
Chemical Explanation:
- Alkali metals (R) readily lose 1 electron to form cations
- Halogens (Y) readily gain 1 electron to form anions
- This electron transfer creates strong ionic bonds
- Example: R (Na-like) + Y (Cl-like) → RY (NaCl-like)
Why Other Options Are Wrong:
- A: All metals conduct electricity
- C: Only halogens/hydrogen/nitrogen/oxygen form diatomic molecules
- D: Noble gases are chemically inert
Therefore, the only correct statement is B, as it correctly describes ionic compound formation between an alkali metal and a halogen.
Which statement about the Periodic Table is correct?
A Most metallic elements are on the left.
B Elements in the same period have the same number of outer electrons.
C Elements on the left are usually gases.
D The relative atomic mass of the elements increases from right to left.
▶️ Answer/Explanation
Ans: A
Detailed Analysis of Each Option:
| Option | Evaluation | Explanation |
|---|---|---|
| A | Correct |
|
| B | Incorrect |
|
| C | Incorrect |
|
| D | Incorrect |
|
Periodic Trends Supporting Option A:
- Metallic Character:
- Highest in bottom-left (Francium most metallic)
- Decreases moving up and right
- Electron Configuration:
- Left-side elements readily lose electrons (metallic bonding)
- Right-side elements tend to gain electrons (non-metals)
Visual Representation:
Metallicity Trend: Metals (left) → Metalloids (diagonal) → Non-metals (right)
Therefore, the only completely correct statement is A, as it accurately describes the distribution of metallic elements in the Periodic Table.
The diagram shows elements W, X, Y and Z in a section of the Periodic Table.

Which statement about the reactivity of the elements is correct?
A) X is more reactive than Y, and W is more reactive than Z.
B) X is more reactive than Y, and Z is more reactive than W.
C) Y is more reactive than X, and W is more reactive than Z.
D) Y is more reactive than X, and Z is more reactive than W.
▶️ Answer/Explanation
Ans: C
1. Element Identification:
• W: Fluorine (F) – Group 17
• X: Sodium (Na) – Group 1
• Y: Potassium (K) – Group 1
• Z: Chlorine (Cl) – Group 17 2. Reactivity Trends:
• Group 1 (Alkali Metals):
– Reactivity increases down the group (K > Na)
– Due to decreasing ionization energy
• Group 17 (Halogens):
– Reactivity decreases down the group (F > Cl)
– Due to decreasing electronegativity 3. Correct Relationships:
• Y (K) > X (Na) in reactivity
• W (F) > Z (Cl) in reactivity
Thus confirming option C 4. Experimental Evidence:
• K reacts violently with water vs Na’s vigorous reaction
• F reacts with nearly all elements vs Cl’s selective reactivity
Which statement about elements in Group VIII of the Periodic Table is correct?
A. They all have a full outer shell of electrons.
B. They all react with Group I elements to form ionic compounds.
C. They are all diatomic molecules.
D. They are all liquids at room temperature.
▶️ Answer/Explanation
Ans: A
Analysis of Group VIII (Noble Gases) properties:
| Option | Evaluation | Scientific Explanation |
|---|---|---|
| A | Correct | Noble gases have complete valence shells:
|
| B | Incorrect | Noble gases are inert and don’t readily form compounds (except Xe/Kr under extreme conditions) |
| C | Incorrect | Noble gases exist as monatomic (single-atom) molecules |
| D | Incorrect | Physical states at room temperature:
|
Key characteristics of noble gases:
- Electron Configuration: Stable octet (or duplet for He)
- Reactivity: Extremely low due to filled valence shells
- Physical State: All gases at standard conditions
- Atomicity: Monatomic molecular form
Therefore, only statement A correctly describes the defining feature of Group VIII elements – their complete outer electron shells.
Which set of elements shows the change from metallic to non-metallic character across a period of the Periodic Table?
- beryllium → magnesium → calcium
- fluorine → bromine → iodine
- oxygen → boron → lithium
- sodium → silicon → chlorine
▶️ Answer/Explanation
Ans: D (sodium → silicon → chlorine)
Analysis of each option:
| Option | Elements | Pattern | Evaluation |
|---|---|---|---|
| A | Be → Mg → Ca | Group 2 metals (vertical) | All metals |
| B | F → Br → I | Group 17 (vertical) | All nonmetals |
| C | O → B → Li | Reverse period | Nonmetal → metalloid → metal |
| D | Na → Si → Cl | Period 3 (horizontal) | Metal → metalloid → nonmetal |
Only option D correctly shows the left-to-right transition across a period from metallic (Na) through metalloid (Si) to nonmetallic (Cl) character.
Which pair of elements reacts together most violently?
- chlorine and lithium
- chlorine and potassium
- iodine and lithium
- iodine and potassium
▶️ Answer/Explanation
Ans: B
Analysis of reactivity trends:
| Option | Reactivity Factors | Violence of Reaction |
|---|---|---|
| A (Cl + Li) |
| Vigorous but less than B |
| B (Cl + K) |
| Most violent (flames/sparks) |
| C (I + Li) |
| Moderate reaction |
| D (I + K) |
| Vigorous but less than B |
Key chemical principles:
- Alkali metal reactivity:
- Increases down Group 1: K > Li
- K has weaker metallic bonding (larger atomic radius)
- Halogen reactivity:
- Decreases down Group 17: Cl > I
- Cl higher electronegativity than I
- Reaction violence:
- 2K + Cl2 → 2KCl (extremely exothermic)
- Produces violet flame and explosive sounds
Therefore, chlorine and potassium (B) react most violently due to the combination of the most reactive alkali metal and most reactive halogen in the options.
Part of the Periodic Table is shown.
Which element is used to provide an inert atmosphere?
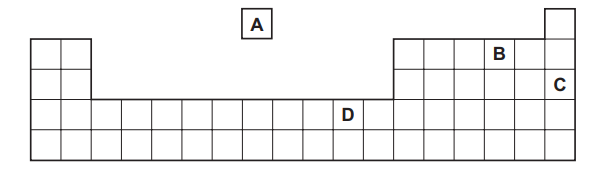
▶️ Answer/Explanation
Ans: C
1. Identifying the Elements:
– A: Sodium (Na) – reactive metal
– B: Chlorine (Cl) – reactive non-metal
– C: Argon (Ar) – noble gas
– D: Potassium (K) – reactive metal
2. Inert Atmosphere Requirements:
Noble gases (Group 18) are chemically inert due to complete valence shells.
3. Industrial Applications:
Argon (Ar) is commonly used in:
– Welding to prevent oxidation
– Light bulbs to prevent filament combustion
– Food packaging to prevent spoilage
The elements sodium to argon form Period 3 of the Periodic Table.
Which row describes the trend across Period 3 from left to right?
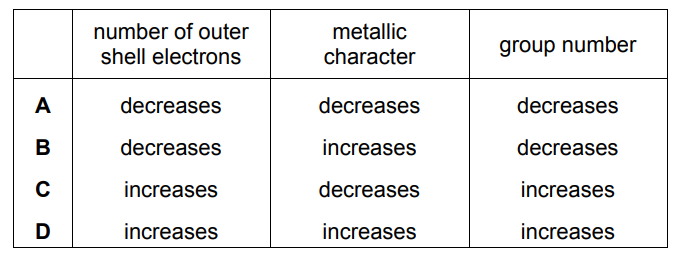
▶️ Answer/Explanation
Ans: C
Key trends across Period 3 (Na to Ar):
| Property | Trend | Explanation |
|---|---|---|
| Atomic radius | Decreases | Increasing nuclear charge pulls electrons closer |
| Electronegativity | Increases | Greater nuclear attraction for bonding electrons |
| Metallic character | Decreases | Transition from metals → metalloids → non-metals |
| Outer electrons | Increase (1→8) | Each element adds one more electron |
Option C correctly shows:
- Decreasing atomic radius
- Increasing electronegativity
- Decreasing metallic character
This matches all major periodic trends across Period 3.
Which statement explains why elements in Group VIII of the Periodic Table are unreactive?
- They are monatomic gases.
- They form stable diatomic molecules.
- They have a full outer shell of electrons.
- They share electrons with each other.
▶️ Answer/Explanation
Ans: C
The noble gases (Group VIII) are unreactive because:
- Electron Configuration:
- They possess a complete valence shell (octet for most, duet for helium)
- This stable configuration doesn’t require gaining/losing/sharing electrons
- Why Other Options Are Incorrect:
- A: Monatomic nature is a consequence of their stability, not the cause
- B: Noble gases don’t form diatomic molecules (except under extreme conditions)
- D: They don’t share electrons as they’re already stable
- Chemical Significance:
- Their full outer shell gives them high ionization energies and zero electronegativity
- This explains their lack of chemical reactivity under normal conditions
Thus, the fundamental reason for their unreactivity is having a full outer shell of electrons (Option C).
Part of the Periodic Table is shown.
Which element forms an acidic oxide?
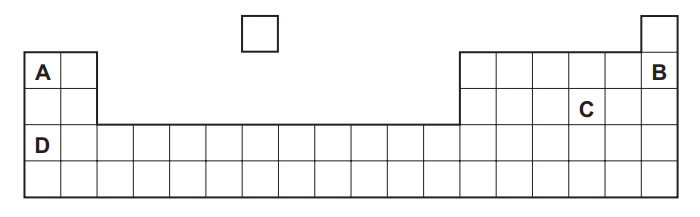
▶️ Answer/Explanation
Ans: C
Periodic Table Analysis:
- Element Identification:
- C is sulfur (S) – Group 16 nonmetal
- Forms SO2 and SO3 (both acidic oxides)
- Acidic Oxide Properties:
- SO2 + H2O → H2SO3 (sulfurous acid)
- SO3 + H2O → H2SO4 (sulfuric acid)
- React with bases to form sulfites/sulfates
- Other Elements:
- A (Mg) – Forms basic oxide (MgO)
- B (Al) – Forms amphoteric oxide (Al2O3)
- D (Si) – Forms weakly acidic oxide (SiO2)
Chemical Evidence:
- Sulfur oxides turn blue litmus red
- Form strong acids in water (pH < 3)
- React completely with NaOH: SO2 + 2NaOH → Na2SO3 + H2O
Therefore, C (sulfur) forms the most strongly acidic oxide among the options.
Which statement about trends in the Periodic Table is not correct?
A Elements in the same period have the same number of electron shells.
B The elements change from metals to non-metals from left to right.
C The number of protons in an atom of an element increases from left to right.
D The oxides of the elements change from acidic to basic from left to right.
▶️ Answer/Explanation
Ans: D
Periodic Trend Analysis
| Option | Evaluation | Explanation |
|---|---|---|
| A | Correct | Period number = number of electron shells |
| B | Correct | Left: Metals (Na, Mg) → Right: Non-metals (P, S, Cl) |
| C | Correct | Atomic number increases left to right |
| D | Incorrect | Oxides change from basic to acidic, not acidic to basic |
Oxide Trend Details
The correct oxide trend across a period is:
Basic oxides → Amphoteric oxides → Acidic oxides
Examples in Period 3:
- Na2O (strongly basic)
- Al2O3 (amphoteric)
- SO3 (strongly acidic)
Conclusion: Option D reverses the actual trend of oxide character across periods. The correct statement should be: “The oxides change from basic to acidic from left to right,” making D the incorrect option.
Which statement about the Periodic Table is not correct?
A It can be used to find the atomic number of an element.
B It can be used to find the physical state of an element.
C It can be used to find the symbol of an element.
D It can be used to predict the properties of an element.
▶️ Answer/Explanation
Ans: B
Analysis of Each Statement:
| Option | Correctness | Explanation |
|---|---|---|
| A | Correct | The atomic number (proton number) is the fundamental organizing principle of the Periodic Table, appearing with each element. |
| B | Incorrect | The Periodic Table does not show physical states:
|
| C | Correct | Element symbols are a standard feature of all Periodic Tables (e.g., Fe for iron, O for oxygen). |
| D | Correct | Periodic trends allow prediction of properties like reactivity, electronegativity, and bonding type based on position. |
Why Option B is the Correct Answer:
- Physical State Variability:
- Elements can exist in different states under different conditions (e.g., oxygen is gas at room temp but liquid at -183°C)
- No standard representation of states in the table layout
- What the Periodic Table Actually Shows:
- Atomic number and mass
- Element symbols and names
- Electron configuration patterns
- Periodic trends in chemical properties
Key Exception:
While most periodic tables don’t show physical states, some educational versions might color-code states at standard temperature and pressure (STP). However, this is not a fundamental feature of the Periodic Table itself.
Therefore, B is the correct choice as it identifies the statement that is not true about the standard Periodic Table.
Which element is in the same period of the Periodic Table as silicon?
- Germanium
- Scandium
- Sodium
- Strontium
▶️ Answer/Explanation
Ans: C
1. Periodic Table Position:
• Silicon (Si): Period 3, Group 14
• Sodium (Na): Period 3, Group 1 2. Why Sodium is Correct:
• Both have electrons filling the 3rd energy level
• Same principal quantum number (n=3) 3. Other Elements’ Periods:
• Germanium (Ge): Period 4 (same group as Si)
• Scandium (Sc): Period 4
• Strontium (Sr): Period 5 4. Key Characteristics of Period 3:
• Contains 8 elements (Na to Ar)
• Shows transition from metallic to nonmetallic character
• Includes important elements like Al, P, and S
Iodine, I, has a lower relative atomic mass than tellurium, Te, but is placed after it in the Periodic Table.
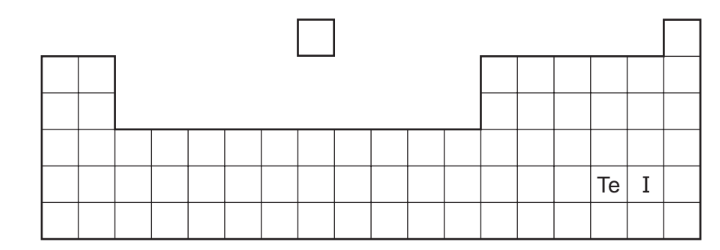
Which statement explains why iodine is placed after tellurium in the Periodic Table?
A Iodine has fewer neutrons than tellurium.
B Iodine has fewer protons than tellurium.
C Iodine has more neutrons than tellurium.
D Iodine has more protons than tellurium.
▶️ Answer/Explanation
Ans: D
This question demonstrates the fundamental principle of Periodic Table organization:
| Element | Atomic Number | Atomic Mass | Position |
|---|---|---|---|
| Tellurium (Te) | 52 | 127.60 | Before Iodine |
| Iodine (I) | 53 | 126.90 | After Tellurium |
Key concepts:
- Periodic Law: Elements are arranged by atomic number (proton count), not atomic mass
- Anomalous Pair: Te (52p, 76n) vs I (53p, 74n) – I has lower mass but higher atomic number
- Chemical Properties: Iodine’s 53 protons give it different chemical behavior than tellurium
Why other options are incorrect:
- A: Neutron count doesn’t determine position (Te-76n, I-74n)
- B: Incorrect proton comparison (I has more protons)
- C: Actually, iodine has fewer neutrons
Therefore, option D correctly identifies that iodine’s higher proton count (atomic number) determines its position, despite its lower atomic mass.
The Periodic Table lists all the known elements.
Elements are arranged in order of …….1……. number.
The melting points of Group I elements …….2……. down the group.
The melting points of Group VII elements …….3……. down the group.
Which words correctly complete gaps 1, 2 and 3?
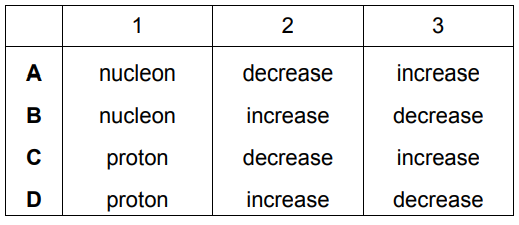
▶️ Answer/Explanation
Ans: C (proton, decrease, increase)
Complete explanation:
- Gap 1: proton
- Elements are arranged by increasing proton (atomic) number
- This is the fundamental organizing principle of the Periodic Table
- Gap 2: decrease
- Group I (alkali metals) melting points decrease down the group
- Due to increasing atomic size weakening metallic bonds
- Example: Li (181°C) → Na (98°C) → K (64°C)
- Gap 3: increase
- Group VII (halogens) melting points increase down the group
- Due to increasing van der Waals forces between larger molecules
- Example: F₂ (-220°C) → Cl₂ (-101°C) → Br₂ (-7°C)
This combination correctly describes both the organization of the Periodic Table and the contrasting trends in Groups I and VII.
Which trend occurs across the period from sodium to argon?
- a change from metal to non-metal
- an increase in melting point
- a more violent reaction with water
- an increase in electrical conductivity
▶️ Answer/Explanation
Ans: A
Analysis of periodic trends from Na (Period 3 start) to Ar (Period 3 end):
| Element Sequence | Classification | Key Properties |
|---|---|---|
| Na, Mg, Al | Metals | Good conductors, malleable, basic oxides |
| Si | Metalloid | Semiconductor, amphoteric oxide |
| P, S, Cl | Non-metals | Poor conductors, acidic oxides |
| Ar | Noble gas | Non-metal, inert, no oxide formation |
Evaluating each option:
- A (Correct):
- Clear transition: Na/Mg/Al (metals) → Si (metalloid) → P/S/Cl (non-metals) → Ar (noble gas)
- Demonstrates the metal-nonmetal transition across periods
- B (Incorrect):
- Melting points: Na(98°C) → Mg(650°C) → Al(660°C) → Si(1414°C) → P(44°C) → S(115°C) → Cl(-101°C) → Ar(-189°C)
- Peaks at silicon then decreases
- C (Incorrect):
- Reactivity with water decreases: Na (violent) → Mg (slow) → Al (protective oxide layer prevents reaction)
- Non-metals don’t react with water in same way
- D (Incorrect):
- Conductivity decreases: Good (metals) → Moderate (Si) → Poor (non-metals) → None (Ar)
Therefore, only option A correctly describes the trend from sodium to argon.
Which property of elements increases across a period of the Periodic Table?
A metallic character
B number of electron shells
C number of outer shell electrons
D tendency to form positive ions
▶️ Answer/Explanation
Ans: C
1. Periodic Trends:
Across a period (left to right):
– Outer electrons: Increase from 1 to 8 (Option C correct)
– Metallic character: Decreases (Option A wrong)
– Electron shells: Constant within a period (Option B wrong)
– Positive ion formation: Decreases (Option D wrong)
2. Electronic Configuration:
Each element adds 1 more valence electron than the previous
Example: Period 2 elements have 2nd shell e– counts from 1 (Li) to 8 (Ne)
3. Why Others Are Incorrect:
Metallic character and positive ion tendency decrease as electronegativity increases
Part of the Periodic Table is shown.
Which element is a soft solid that reacts violently with cold water?
▶️ Answer/Explanation
Ans: D (Rubidium – Rb)
Key characteristics of the correct element:
- Alkali metal properties:
- Soft, silvery solids (can be cut with a knife)
- Extremely reactive with water
- Single valence electron makes them highly reactive
- Violent water reaction:
2Rb(s) + 2H2O(l) → 2RbOH(aq) + H2(g) + heat
- Produces hydrogen gas (may ignite)
- Exothermic reaction (may cause explosion)
- Forms alkaline hydroxide solution
- Periodic Table position:
- Group 1 element (alkali metals)
- Below potassium in reactivity series
Why other options are incorrect:
- A: Transition metal (less reactive with water)
- B: Alkaline earth metal (reacts slowly with water)
- C: Halogen (non-metal, doesn’t react this way)
Therefore, Option D (Rb) is the correct answer as it shows all the described properties.
In the Periodic Table, how does the metallic character of the elements vary from left to right across a period?
- It decreases.
- It increases.
- It increases then decreases.
- It stays the same.
▶️ Answer/Explanation
Ans: A
The metallic character decreases across a period due to:
- Atomic Structure Changes:
- Increasing nuclear charge pulls electrons closer
- Decreasing atomic radius makes electron loss harder
- Trends in Properties:
- Left side: Alkali/alkaline earth metals (strong metallic character)
- Middle: Transition metals (moderate metallic character)
- Right side: Nonmetals (no metallic character)
- Why Other Options Are Incorrect:
- B: Opposite of observed trend
- C: No periodic trend shows this pattern
- D: Clearly changes across periods
Key evidence:
• Ionization energy increases left to right (harder to remove electrons)
• Electronegativity increases (greater electron attraction)
• Electrical conductivity decreases
Thus, metallic character decreases from left to right across a period (Option A).
Which statement about the Periodic Table is correct?
- Elements in the same period have the same number of outer electrons.
- The elements on the left are usually gases.
- The most metallic elements are on the left.
- The relative atomic mass of the elements increases from right to left.
▶️ Answer/Explanation
Ans: C
1. Periodic Trends Analysis:
• Metallic character decreases left to right across periods and increases down groups
• Leftmost elements (Groups 1-2) are highly metallic (e.g., Na, K, Ca) 2. Why Other Options Are Incorrect:
• A: Elements in same group (not period) share outer electrons
• B: Left-side elements are mostly solids (except H gas)
• D: Atomic mass generally increases left to right 3. Metallic Properties:
• Good conductors of heat/electricity
• Malleable and ductile
• Form positive ions (lose electrons) 4. Visualizing the Trend:
Metallic character peak at bottom-left (Fr) and minimum at top-right (He)
The results of three tests on a solution of compound X are shown in the table.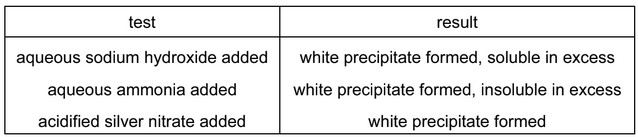
What is compound X?
A aluminium bromide
B aluminium chloride
C zinc bromide
D zinc chloride
▶️ Answer/Explanation
Ans: B (aluminium chloride)
Analysis of test results:
| Test | Observation | Explanation |
|---|---|---|
| NaOH(aq) | White precipitate | AlCl3 + 3NaOH → Al(OH)3↓ + 3NaCl (white gelatinous ppt) |
| NH3(aq) | White precipitate | AlCl3 + 3NH3 + 3H2O → Al(OH)3↓ + 3NH4Cl |
| AgNO3(aq) | White precipitate | AlCl3 + 3AgNO3 → 3AgCl↓ + Al(NO3)3 (white curdy ppt) |
Key identification points:
- All three tests produce white precipitates characteristic of Al3+ and Cl–
- Zinc compounds would give soluble complex with excess NaOH
- Bromides would give cream-colored AgBr precipitate
Therefore, Option B (aluminium chloride) is the only compound that matches all test results.
