Pre AP Chemistry -2.2D Lewis Diagrams and Structural Isomerism- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2D Lewis Diagrams and Structural Isomerism- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2D Lewis Diagrams and Structural Isomerism- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which row gives the number of covalent bonds in one molecule of ammonia and in one molecule of hydrogen chloride?
| ammonia | hydrogen chloride | |
|---|---|---|
| A | 3 | 1 |
| B | 3 | 2 |
| C | 4 | 1 |
| D | 4 | 2 |
▶️ Answer/Explanation
Ans: A
Ammonia (NH₃) has 3 covalent bonds – one between nitrogen and each hydrogen atom. Nitrogen has 5 valence electrons and forms 3 single bonds, leaving one lone pair.
Hydrogen chloride (HCl) has just 1 covalent bond between the hydrogen and chlorine atoms. Hydrogen can only form one bond, and chlorine needs just one more electron to complete its outer shell.
Therefore, the correct combination is 3 bonds in ammonia and 1 bond in hydrogen chloride.
Ammonia, \(NH_3\), is a covalent molecule.
Which diagram shows the outer shell electron arrangement in a molecule of ammonia?

▶️ Answer/Explanation
Ans: C
Ammonia (\(NH_3\)) has a nitrogen atom with 5 valence electrons, forming 3 covalent bonds with hydrogen atoms (each contributes 1 electron). The nitrogen retains 1 lone pair of electrons. Diagram C correctly shows:
1. Three N-H bond pairs (shared electrons).
2. One lone pair on nitrogen.
The other diagrams either misplace electrons or incorrectly represent bonding.
The electronic structures of atoms X and Y are shown.
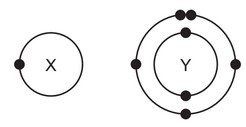
X and Y form a covalent compound. What is its formula?
A) \(XY_5\) B) \(XY_3\) C) \(XY\) D) \(X_3Y\)
▶️ Answer/Explanation
Ans: D
From the electronic structures:
- Atom X has 1 valence electron (needs to lose 1 or share to stabilize).
- Atom Y has 5 valence electrons (needs 3 more to complete its octet).
To form a stable covalent compound:
- Each Y shares 3 of its electrons with 3 X atoms.
- Each X shares its 1 electron with Y.
Thus, the formula is \(X_3Y\), where Y achieves an octet by bonding with three X atoms.
Other options are incorrect because:
- \(XY_5\) would require X to share 5 electrons (impossible with only 1 valence electron).
- \(XY_3\) would leave Y with an incomplete octet (only 6 electrons).
- \(XY\) would not satisfy Y’s octet requirement (only 6 electrons total).
The diagrams show the electron arrangements in the atoms of four elements.
Which element does not form a covalent bond?
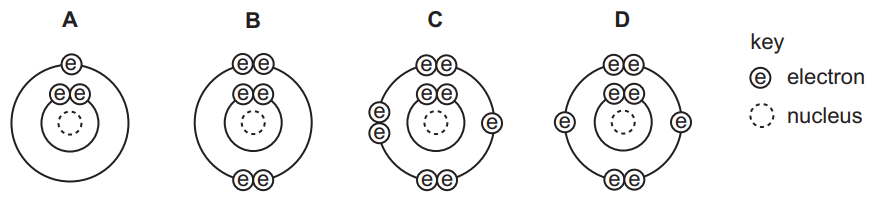
▶️ Answer/Explanation
Ans: A
The element represented by A is lithium (Li), which has one valence electron. To achieve stability, lithium tends to lose this electron, forming a positive ion (Li+). This behavior is characteristic of ionic bonding, not covalent bonding.
In contrast, elements like B, C, and D (likely nonmetals such as oxygen or fluorine) share electrons to complete their valence shells, forming covalent bonds. Since Li does not share electrons but instead transfers them, it does not form covalent bonds.
Which statement describes a homologous series?
A) a family of elements in the same group of the Periodic Table
B) a family of elements with similar chemical properties
C) a family of compounds with the same functional group
D) a family of compounds with similar physical properties
▶️ Answer/Explanation
Ans: C
A homologous series is a family of organic compounds that:
- Have the same functional group
- Show a gradual change in physical properties
- Have similar chemical properties
- Differ by a CH2 unit in their molecular formulae
Key points:
- Option A describes groups in the Periodic Table, not homologous series
- Option B is partially correct but too vague – it’s about compounds, not elements
- Option D is partially correct but not the complete definition
- Option C is the most accurate as the functional group defines the series (e.g., alcohols, alkanes)
Examples of homologous series include alkanes (CH4, C2H6, etc.) and alcohols (CH3OH, C2H5OH, etc.).
PVA is a polymer. The monomer has the structure shown.
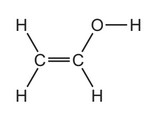
To which homologous series does this compound belong?
▶️ Answer/Explanation
Ans: A
The monomer structure shows:
1. A hydroxyl (–OH) group, characteristic of alcohols.
2. A carbon-carbon double bond (C=C), characteristic of alkenes.
Thus, it belongs to both alcohol and alkene series, but the primary functional group (alcohol) takes precedence in classification. The homologous series is determined by the –OH group, making it an alcohol.
Three statements about ammonia molecules and ammonium ions are given.
1. In aqueous solution, ammonia molecules form coordinate bonds with hydroxide ions.
2. Ammonium ions are Brønsted–Lowry acids.
3. The H–N–H bond angle is larger in the ammonium ion than in the ammonia molecule.
Which statements are correct?
▶️ Answer/Explanation
Ans: C
Statement 1: Incorrect. Ammonia (\(NH_3\)) forms coordinate bonds with \(H^+\) ions (protons) to form ammonium ions (\(NH_4^+\)), not hydroxide ions (\(OH^-\)).
Statement 2: Correct. Ammonium ions (\(NH_4^+\)) can donate a proton (\(H^+\)), making them Brønsted–Lowry acids.
Statement 3: Correct. The \(H-N-H\) bond angle in \(NH_4^+\) (≈109.5°, tetrahedral) is larger than in \(NH_3\) (≈107°, trigonal pyramidal) due to the absence of a lone pair in \(NH_4^+\).
Thus, only statements 2 and 3 are correct, making C the correct answer.
Kerosene is used as an aircraft fuel. Q is one of the molecules in kerosene and has the skeletal formula shown below.
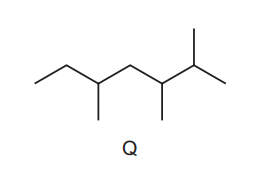
Other structural isomers of this molecule are also found in kerosene.
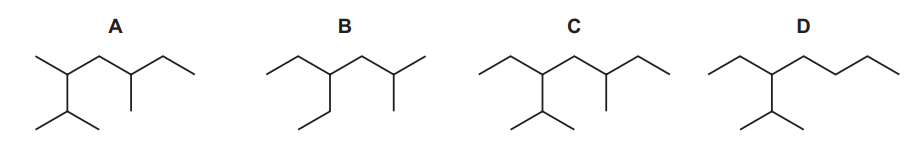
Which structure is a structural isomer of Q?
▶️ Answer/Explanation
Step 1: Understand structural isomers
Structural isomers have the same molecular formula but a different arrangement of atoms.
Step 2: Count carbon atoms in Q
From the skeletal structure of Q, the molecule contains the same total number of carbon atoms as the correct isomer.
Step 3: Compare the options
- Options A, B and C either change the number of carbon atoms in the main chain or do not match the same molecular formula.
- Option D has the same number of carbon atoms as Q but arranged differently, which satisfies the definition of a structural isomer.
Conclusion:
Structure D is a structural isomer of Q.
Ans: D
Structural isomerism and stereoisomerism should be considered in answering this question.
Compound J is reacted with KOH dissolved in ethanol. Three isomeric alkenes with molecular formula C4H8 are formed.
What is J?
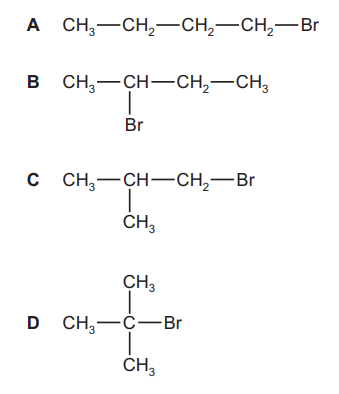
▶️ Answer/Explanation
Step 1: Identify the reaction
KOH in ethanol causes elimination (E2), removing HBr from a haloalkane to form an alkene.
Step 2: Identify possible alkenes (C4H8)
- But-1-ene
- cis-But-2-ene
- trans-But-2-ene
These are the three possible alkene isomers with formula C4H8.
Step 3: Determine which haloalkane can form all three
Only 2-bromobutane can eliminate hydrogen atoms from two different β-carbons to produce:
- But-1-ene
- cis-But-2-ene
- trans-But-2-ene
Step 4: Identify the correct option
Option B represents 2-bromobutane.
Ans: B
In this question, alkenes and cyclic alkanes should be considered.
How many structural isomers of C4H8 are there?
A 3 B 4 C 5 D 6
▶️ Answer/Explanation
Step 1: Determine possible types of compounds
The molecular formula C4H8 follows the general formula CnH2n, which corresponds to either:
- Alkenes (one C=C double bond)
- Cycloalkanes (a ring structure)
Step 2: List alkene structural isomers
- But-1-ene
- But-2-ene
- 2-methylpropene
Step 3: List cyclic alkane structural isomers
- Cyclobutane
- Methylcyclopropane
Step 4: Count total structural isomers
Total = 3 (alkenes) + 2 (cycloalkanes) = 5
Ans: C
Structural isomerism only should be considered when answering this question.
Molecule X contains three C=C double bonds. One mole of X is reacted with three moles of HBr. The carbon skeleton is unchanged.
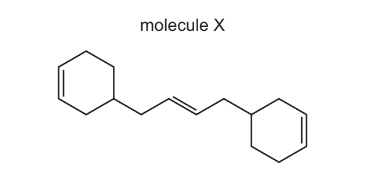
How many different products are formed?
A 3 B 4 C 6 D 8
▶️ Answer/Explanation
Step 1: Identify the reaction
Hydrogen bromide adds across each C=C double bond in an electrophilic addition reaction.
Since molecule X contains three C=C double bonds, three molecules of HBr react to give three additions.
Step 2: Possible orientations of addition
For each double bond, HBr can add in two possible ways depending on which carbon receives H and which receives Br.
However, because the carbon skeleton has symmetry, some of these products are identical.
Step 3: Count distinct structural products
After considering symmetry and ignoring stereoisomerism (since only structural isomers are considered), the total number of different products formed is 4.
Ans: B
How many structural isomers are there of trichloropropane, C3H5Cl3?
A 3 B 4 C 5 D 6
▶️ Answer/Explanation
Step 1: Identify the carbon skeleton
Propane has three carbon atoms arranged as:
CH3–CH2–CH3
Three hydrogen atoms are replaced by chlorine atoms.
Step 2: Consider different substitution patterns
The three Cl atoms can be distributed among the three carbons in different ways:
- 1,1,1-trichloropropane
- 1,1,2-trichloropropane
- 1,1,3-trichloropropane
- 1,2,2-trichloropropane
- 1,2,3-trichloropropane
Structures like 2,2,3 are equivalent to 1,2,2 due to symmetry of the propane chain.
Step 3: Count unique structures
Total structural isomers = 5.
Ans: C
Structural isomerism only should be considered when answering this question.
All the isomeric alcohols with the molecular formula C5H12O are added separately to warm alkaline aqueous iodine.
How many of the isomers give a yellow precipitate?
A 0 B 1 C 2 D 3
▶️ Answer/Explanation
Step 1: Recall the iodoform test
A yellow precipitate of CHI3 forms when a compound contains either:
- a methyl ketone group (–COCH3) or
- a secondary alcohol with the group CH3–CH(OH)–
Step 2: Consider alcohol isomers of C5H12O
There are eight alcohol structural isomers of C5H12O.
Only alcohols containing the fragment CH3–CH(OH)– will give the iodoform test.
Step 3: Identify those alcohols
- Pentan-2-ol
- 3-methylbutan-2-ol
Both contain the required CH3–CH(OH)– group.
Step 4: Count
Number of alcohols giving the iodoform test = 2.
Ans: C
Structural isomerism and stereoisomerism should be considered when answering this question.
2-bromopentane is heated with an excess of ethanolic sodium hydroxide.
How many different hydrocarbons are produced?
A 1 B 2 C 3 D 4
▶️ Answer/Explanation
Step 1: Identify the reaction
Ethanolic NaOH promotes elimination (E2), removing HBr to form alkenes.
Step 2: Possible elimination positions
Hydrogen can be removed from carbons adjacent to the carbon bearing Br.
This forms:
- Pent-1-ene
- Pent-2-ene
Step 3: Consider stereoisomerism
Pent-2-ene exists as two stereoisomers:
- cis-pent-2-ene
- trans-pent-2-ene
Step 4: Count total hydrocarbons
- Pent-1-ene
- cis-pent-2-ene
- trans-pent-2-ene
Total hydrocarbons formed = 3.
Ans: C
Structural isomerism and stereoisomerism should be considered when answering this question.
3-methylhexan-3-ol reacts with hot, concentrated sulfuric acid to form several isomeric compounds with the molecular formula C7H14.
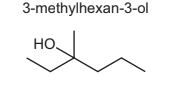
How many isomeric compounds could be formed in this reaction?
A 3 B 4 C 5 D 6
▶️ Answer/Explanation
Step 1: Identify the reaction
Hot concentrated H2SO4 causes dehydration of alcohols, forming alkenes.
The product therefore has the formula C7H14.
Step 2: Determine possible double-bond positions
Elimination can occur by removing hydrogen atoms from different β-carbon atoms around the carbon bearing the –OH group.
This produces several different structural alkene isomers.
Step 3: Consider stereoisomerism
Some of the alkenes formed can exist as E/Z (cis-trans) isomers, increasing the total number of possible products.
Step 4: Count total possible isomers
- Different structural alkenes from dehydration
- Plus possible E/Z stereoisomers
Total number of possible isomeric products = 5.
Ans: C
A new jet fuel has been produced that is a mixture of different structural isomers of compound Q.
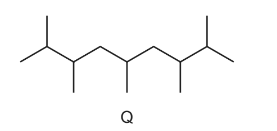
Which skeletal formula represents a structural isomer of Q?
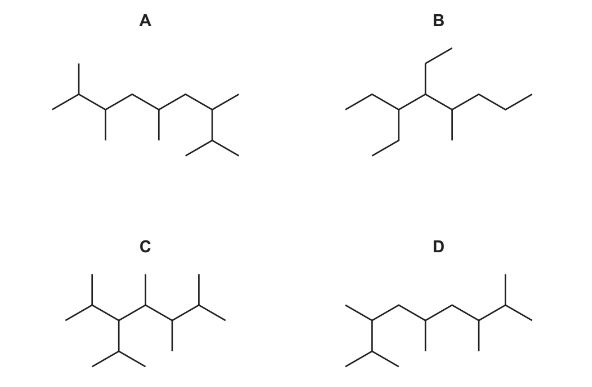
▶️ Answer/Explanation
Step 1: Recall definition of structural isomers
Structural isomers have the same molecular formula but a different arrangement of atoms.
Step 2: Analyse compound Q
From the skeletal formula of Q, count the total number of carbon atoms and the positions of the branches.
Step 3: Compare with options A–D
- Options A, B and D change the total carbon arrangement or do not maintain the same molecular formula.
- Option C has the same number of carbon atoms as Q but a different branching arrangement.
Step 4: Conclusion
Since option C has the same molecular formula but a different structure, it is the correct structural isomer.
Ans: C
X has the molecular formula C5H12O. X has a branched carbon skeleton and a secondary alcohol functional group.
How many structural isomers fit this description of X?
A 1 B 2 C 3 D 4
▶️ Answer/Explanation
Step 1: Recall definition of secondary alcohol
In a secondary alcohol, the carbon bearing the –OH group is attached to two other carbon atoms.
Step 2: Consider the carbon skeleton
The molecule must have:
- 5 carbon atoms
- a branched carbon chain
- a secondary –OH group
Step 3: Examine possible branched skeletons
For C5H12O, branched structures are mainly derived from the 2-methylbutane skeleton.
Placing the –OH group so that the carbon is bonded to two other carbons gives only one valid structure:
3-methyl-2-butanol
Any other placement either:
- produces a primary alcohol, or
- produces a tertiary alcohol.
Step 4: Count valid structures
Only one structure satisfies all conditions.
Ans: A
How many isomeric esters have the molecular formula C4H8O2?
A 2 B 3 C 4 D 5
▶️ Answer/Explanation
Step 1: General ester structure
An ester has the functional group R–COO–R′.
Step 2: Distribute the 4 carbon atoms
The total number of carbons in the acid part and alcohol part must equal 4.
Possible combinations:
- 1 carbon in acid + 3 carbons in alcohol
- 2 carbons in acid + 2 carbons in alcohol
- 3 carbons in acid + 1 carbon in alcohol
Step 3: List the possible esters
- Methyl propanoate
- Ethyl ethanoate
- Propyl methanoate
- Isopropyl methanoate
Total number of structural ester isomers = 4.
Ans: C
In 1869 Ladenburg suggested a structure for benzene, C6H6, in which one hydrogen atom is attached to each carbon atom.
A compound C6H4Cl2 could be formed with the same carbon skeleton as the Ladenburg structure.
How many structural isomers would this compound have?
A 3 B 4 C 5 D 6
▶️ Answer/Explanation
Step 1: Understand the Ladenburg structure
Ladenburg proposed that benzene has a prism structure, where six carbon atoms form a triangular prism instead of a flat ring.
Step 2: Replace two hydrogen atoms with chlorine
In C6H4Cl2, two of the six hydrogen atoms are replaced by chlorine atoms.
Step 3: Consider different positions
Because the prism structure has several distinct carbon positions, the two chlorine atoms can be attached in different ways that are not equivalent.
Step 4: Count possible arrangements
With the geometry of the triangular prism, there are six different structural arrangements for the two chlorine atoms.
Ans: D
Considering only structural isomers, what is the number of alcohols of each type with the formula C5H12O?
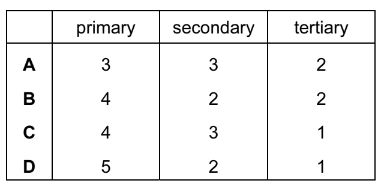
▶️ Answer/Explanation
Step 1: Identify types of alcohols
- Primary alcohol: –OH attached to a carbon bonded to only one other carbon.
- Secondary alcohol: –OH attached to a carbon bonded to two other carbons.
- Tertiary alcohol: –OH attached to a carbon bonded to three other carbons.
Step 2: Count structural isomers for C5H12O
By considering different carbon skeletons and positions of the –OH group:
- Primary alcohols: 4 possible structures
- Secondary alcohols: 3 possible structures
- Tertiary alcohols: 1 possible structure
Step 3: Match with options
The correct distribution is 4 primary, 3 secondary, and 1 tertiary alcohol.
Ans: C
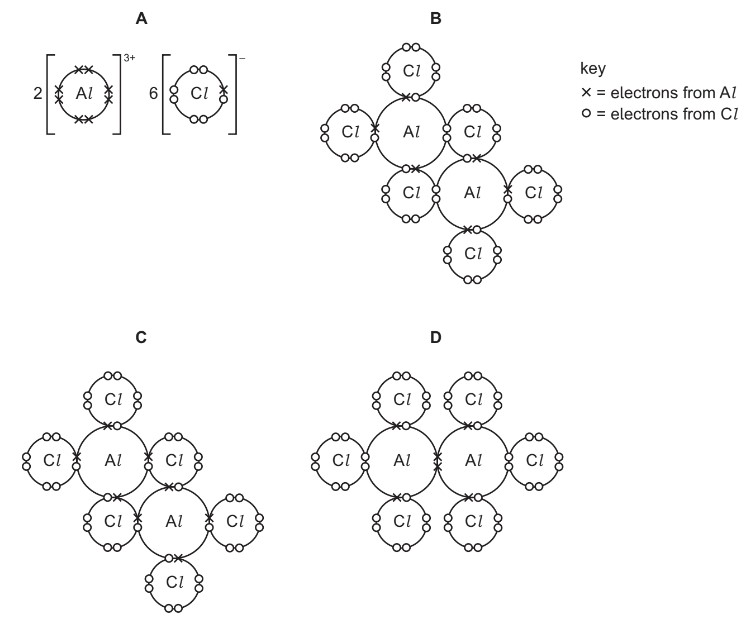
Which dot-and-cross diagram is correct for Al2Cl6?
▶️ Answer/Explanation
Step 1: Understand the structure of AlCl3
Aluminium chloride is electron-deficient because aluminium forms only three bonds and has an incomplete octet.
Step 2: Dimer formation
Two AlCl3 molecules combine to form a dimer, Al2Cl6. Two chlorine atoms act as bridging atoms between the aluminium atoms.
Step 3: Coordinate bonding
Each bridging chlorine donates a lone pair to an aluminium atom, forming a dative (coordinate) bond. Thus the structure contains two bridging Cl atoms connecting the two Al atoms.
Step 4: Identify correct diagram
The correct diagram must show:
- Two aluminium atoms
- Four terminal chlorine atoms
- Two bridging chlorine atoms forming coordinate bonds
Only diagram B correctly represents this arrangement.
Ans: B
In which structure are three atoms bonded together in a straight line?
A poly(ethene), –(–CH2CH2–)n–
B propane, C3H8
C silicon tetrachloride, SiCl4
D sulfur hexafluoride, SF6
▶️ Answer/Explanation
Step 1: Recall molecular geometries
- Poly(ethene): carbon atoms are tetrahedral → bond angles ≈109.5°, not linear.
- Propane (C3H8): carbon atoms are also tetrahedral → atoms are not arranged in a straight line.
- SiCl4: tetrahedral geometry → no straight line of three bonded atoms.
- SF6: octahedral geometry → opposite fluorine atoms lie directly across from each other, forming a straight line through sulfur.
Step 2: Identify linear arrangement
In SF6, F–S–F atoms are arranged in a straight line (180°).
Ans: D
Which molecules contain at least one unpaired electron?
1 NO
2 NO2
3 NH3
▶️ Answer/Explanation
Step 1: Check total valence electrons
- NO: N (5) + O (6) = 11 electrons → odd number → contains an unpaired electron.
- NO2: N (5) + 2×O (12) = 17 electrons → odd number → contains an unpaired electron.
- NH3: N (5) + 3×H (3) = 8 electrons → all paired → no unpaired electrons.
Step 2: Determine correct molecules
Only NO and NO2 contain unpaired electrons.
Ans: B (1 and 2)
In which species is there a lone pair of electrons?
A. CH3 B. CH3+ C. CH3– D. CH4
▶️ Answer/Explanation
Step 1: Examine each species
- CH3 (methyl radical): has an unpaired electron, not a lone pair.
- CH3+ (methyl cation): carbon has three bonds and no lone pairs.
- CH3– (methyl anion): carbon has three bonds and one lone pair of electrons.
- CH4: carbon forms four bonds and has no lone pairs.
Step 2: Identify the correct species
Only CH3– contains a lone pair of electrons on the carbon atom.
Ans: C
Which molecule contains six bonding electrons?
A C2H4 B H2S C NCl3 D SF6
▶️ Answer/Explanation
Step 1: Recall bonding electrons
Each covalent bond contains 2 bonding electrons.
Step 2: Calculate bonding electrons in each molecule
- C2H4: 1 C=C double bond (4 electrons) + 4 C–H bonds (8 electrons) = 12 bonding electrons.
- H2S: 2 S–H bonds → 4 bonding electrons.
- NCl3: 3 N–Cl bonds → 3 × 2 = 6 bonding electrons.
- SF6: 6 S–F bonds → 12 bonding electrons.
Step 3: Identify the correct molecule
Only NCl3 contains six bonding electrons.
Ans: C
In which species does the underlined atom have an incomplete outer shell?
A BF3 B CH3− C F2O D H3O+
▶️ Answer/Explanation
Step 1: Recall the octet rule
Most atoms tend to have 8 electrons in their outer shell (octet). If an atom has fewer than 8 electrons around it, it has an incomplete outer shell.
Step 2: Examine each option
- BF3: Boron forms three B–F bonds. Boron has only 6 electrons around it → incomplete octet.
- CH3−: Carbon forms three bonds and has one lone pair → complete octet.
- F2O: Oxygen has two bonds and two lone pairs → complete octet.
- H3O+: Oxygen has three bonds and one lone pair → complete octet.
Step 3: Conclusion
Only boron in BF3 has an incomplete outer shell.
Ans: A
