Pre AP Chemistry -2.2E Molecular Geometry (VSEPR Theory)- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2E Molecular Geometry (VSEPR Theory)- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2E Molecular Geometry (VSEPR Theory)- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
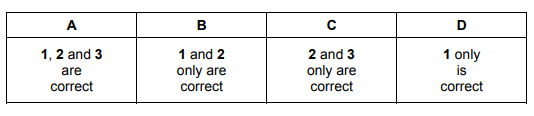
Which molecules have no overall dipole moment?
1 boron trifluoride
2 methane
3 phosphorus pentafluoride
▶️ Answer/Explanation
Step 1: Condition for zero dipole moment
A molecule has no overall dipole moment if:
- bond dipoles are present but
- the molecular shape is symmetrical so the dipoles cancel.
Step 2: Examine each molecule
- BF₃ – trigonal planar and symmetrical → dipoles cancel → no dipole moment.
- CH₄ – tetrahedral and symmetrical → dipoles cancel → no dipole moment.
- PF₅ – trigonal bipyramidal and symmetrical → dipoles cancel → no dipole moment.
Step 3: Conclusion
All three molecules have no overall dipole moment.
Ans: A (1, 2 and 3)
Which molecule does not have any 90° or 180° bond angles?
A \(C_{2}H_{6}\) B \(CO_{2}\) C \(PF_{5}\) D \(SF_{6}\)
▶️ Answer/Explanation
Step 1: Determine molecular geometry
- C₂H₆ – each carbon is tetrahedral, bond angles ≈ 109.5°. No 90° or 180° angles.
- CO₂ – linear, bond angle = 180°.
- PF₅ – trigonal bipyramidal, contains 90°, 120° and 180° angles.
- SF₆ – octahedral, contains 90° and 180° angles.
Step 2: Identify the correct molecule
Only C₂H₆ has bond angles of about 109.5° and therefore does not contain 90° or 180° bond angles.
Ans: A
The characteristic smell of garlic is due to alliin.
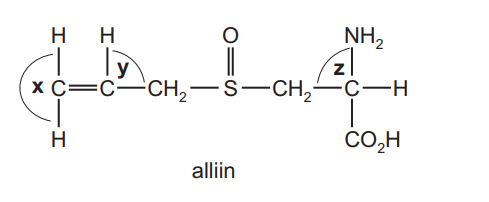
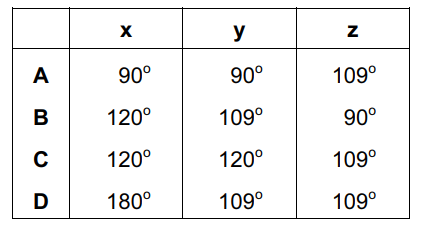
What are the approximate bond angles x, y and z in a molecule of alliin?
▶️ Answer/Explanation
Step 1: Identify hybridisation and geometry
- x: The carbon is sp² hybridised in a C=C double bond → trigonal planar → bond angle ≈ 120°.
- y: The second carbon in the double bond is also sp² hybridised → trigonal planar → bond angle ≈ 120°.
- z: The carbon attached to NH₂, H, CO₂H and CH₂ is sp³ hybridised → tetrahedral → bond angle ≈ 109.5°.
Step 2: Match with the options
x ≈ 120°
y ≈ 120°
z ≈ 109.5°
This corresponds to option C.
Ans: C
Which series shows molecules in order of increasing bond angle?
A CH4 → BF3 → NH3
B H2O → CO2 → BF3
C NH3 → CH4 → CO2
D NH3 → CH4 → H2O
▶️ Answer/Explanation
Step 1: Determine molecular shapes and bond angles
- NH₃: trigonal pyramidal → bond angle ≈ 107°.
- CH₄: tetrahedral → bond angle ≈ 109.5°.
- CO₂: linear → bond angle = 180°.
Step 2: Arrange in increasing order
NH₃ (107°) < CH₄ (109.5°) < CO₂ (180°)
This sequence corresponds to option C.
Ans: C
For which molecule is the dipole moment zero?
A \(CH_3Cl\) B \(CH_2Cl_2\) C \(CHCl_3\) D \(CCl_4\)
▶️ Answer/Explanation
Step 1: Recall dipole moment concept
A molecule has a zero dipole moment when the bond dipoles cancel due to a symmetrical molecular geometry.
Step 2: Examine each molecule
- CH₃Cl – tetrahedral but not symmetrical → dipoles do not cancel → polar.
- CH₂Cl₂ – tetrahedral but two different substituents → dipoles do not cancel → polar.
- CHCl₃ – tetrahedral but substituents are not identical → dipoles do not cancel → polar.
- CCl₄ – perfectly symmetrical tetrahedral molecule → bond dipoles cancel.
Step 3: Conclusion
The molecule with zero dipole moment is CCl₄.
Ans: D
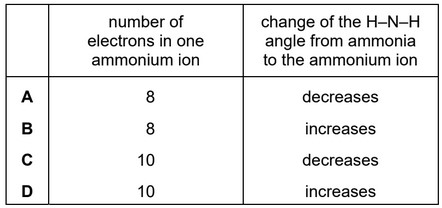
When ammonia, \(NH_3\), is dissolved in water, a small concentration of ammonium ions, \(NH_4^+\), is formed.
Which row is correct?
▶️ Answer/Explanation
Step 1: Number of electrons in ammonium ion
Nitrogen contributes 5 valence electrons and each hydrogen contributes 1 electron.
Total electrons = \(5 + 4(1) = 9\)
The ion has a +1 charge, meaning one electron is removed:
\(9 – 1 = 8\) electrons around nitrogen in bonding pairs.
Thus the ammonium ion contains 4 bonding pairs (8 electrons) around nitrogen.
Step 2: Compare bond angles
- NH₃: trigonal pyramidal with one lone pair → bond angle ≈ 107°.
- NH₄⁺: tetrahedral with no lone pairs → bond angle ≈ 109.5°.
Therefore the H–N–H bond angle increases from ammonia to ammonium.
Step 3: Identify correct row
Number of electrons = 8
Bond angle change = increases
This corresponds to option D.
Ans: D
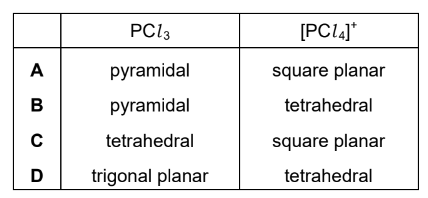
Phosphorus forms two chlorides. Phosphorus(III) chloride, PCl3, is a covalent liquid.
Phosphorus(V) chloride is an ionic solid. One of the ions present is [PCl4]+.
What is the shape of the PCl3 molecule and the [PCl4]+ ion?
▶️ Answer/Explanation
Step 1: Determine the shape of PCl3
- Phosphorus has 5 valence electrons.
- Three P–Cl bonds are formed.
- One lone pair remains on phosphorus.
This gives 4 electron regions (3 bonding pairs + 1 lone pair).
The electron geometry is tetrahedral but the molecular shape becomes trigonal pyramidal.
Step 2: Determine the shape of [PCl4]+
- Phosphorus forms four P–Cl bonds.
- There are no lone pairs on phosphorus.
- Total electron regions = 4 bonding pairs.
This gives a tetrahedral shape.
Step 3: Match with the correct option
PCl3 → trigonal pyramidal
[PCl4]+ → tetrahedral
Ans: B
Which molecules contain at least one bond angle of 120°?
1 C2H4
2 PF5
3 NCl3
▶️ Answer/Explanation
Step 1: Determine molecular shapes
- C₂H₄ – each carbon is sp² hybridised, trigonal planar geometry → bond angles ≈ 120°.
- PF₅ – trigonal bipyramidal structure → equatorial bond angles are 120°.
- NCl₃ – trigonal pyramidal structure → bond angles ≈ 107°, not 120°.
Step 2: Identify molecules with 120° bond angles
C₂H₄ and PF₅ contain at least one 120° bond angle.
Ans: B (1 and 2)
The eight species that follow all have covalent bonds.
In which pair do the species have different shapes from each other?
A BeCl2 and CO2
B CH4 and NH4+
C NH3 and BF3
D SCl2 and H2O
▶️ Answer/Explanation
Step 1: Determine molecular shapes
- BeCl₂ → linear (180°)
- CO₂ → linear (180°)
Option A: same shape.
- CH₄ → tetrahedral
- NH₄⁺ → tetrahedral
Option B: same shape.
- NH₃ → trigonal pyramidal (lone pair on N)
- BF₃ → trigonal planar (no lone pairs on B)
Option C: different shapes.
- SCl₂ → bent
- H₂O → bent
Option D: same shape.
Therefore the correct pair with different shapes is:
NH₃ and BF₃
Ans: C
Histidine is an amino acid.
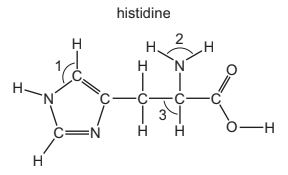
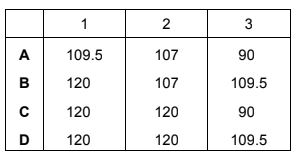
What are the approximate bond angles 1, 2 and 3?
▶️ Answer/Explanation
Step 1: Identify hybridisation at each labelled atom
- Angle 1: Located in the aromatic imidazole ring where atoms are sp² hybridised. Trigonal planar geometry → bond angle ≈ 120°.
- Angle 2: Around the nitrogen in the –NH2 group. Nitrogen has three bonds and one lone pair, giving a trigonal pyramidal arrangement → bond angle ≈ 107°.
- Angle 3: Around the tetrahedral carbon in the side chain (sp³ hybridised). Bond angle ≈ 109.5°.
Step 2: Match with the given options
1 ≈ 120°
2 ≈ 107°
3 ≈ 109.5°
This corresponds to option B.
Ans: B
Which molecule or ion contains the smallest bond angle?
A C2H4 B CH3COCH3 C NH4+ D NH3
▶️ Answer/Explanation
Step 1: Determine molecular geometries
- C₂H₄ – trigonal planar around carbon → bond angle ≈ 120°.
- CH₃COCH₃ – contains trigonal planar carbonyl carbon → bond angles ≈ 120°.
- NH₄⁺ – tetrahedral → bond angle ≈ 109.5°.
- NH₃ – trigonal pyramidal with one lone pair → bond angle ≈ 107°.
Step 2: Compare the bond angles
120° > 109.5° > 107°
The smallest bond angle occurs in NH₃ due to lone pair–bond pair repulsion compressing the bond angles.
Ans: D
Which molecules and ions have a bond angle of 120°?
1 BF3
2 CH3–
3 NH3
▶️ Answer/Explanation
Step 1: Determine shapes of the species
- BF₃: Boron has three bonding pairs and no lone pairs → trigonal planar → bond angle ≈ 120°.
- CH₃⁻: Carbon has three bonding pairs and one lone pair → trigonal pyramidal → bond angle ≈ 107°, not 120°.
- NH₃: Nitrogen has three bonding pairs and one lone pair → trigonal pyramidal → bond angle ≈ 107°, not 120°.
Step 2: Identify correct species
Only BF₃ has a bond angle of approximately 120°.
This corresponds to option D.
Ans: D
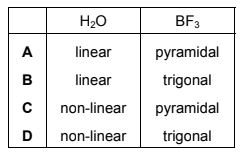
What are the shapes of the molecules of water and boron trifluoride?
▶️ Answer/Explanation
Step 1: Determine the shape of H₂O
- Oxygen has 6 valence electrons.
- Two O–H bonds are formed.
- Two lone pairs remain on oxygen.
This gives four electron regions (2 bonding pairs + 2 lone pairs).
The electron geometry is tetrahedral, but the molecular shape becomes bent (V-shaped) with a bond angle of about 104.5°.
Step 2: Determine the shape of BF₃
- Boron has 3 valence electrons.
- It forms three B–F bonds.
- There are no lone pairs on boron.
This gives a trigonal planar geometry with bond angles of about 120°.
Step 3: Conclusion
H₂O → bent
BF₃ → trigonal planar
This corresponds to option D.
Ans: D
Histamine is produced in the body to help fight infection. Its shape allows it to fit into receptors which expand blood vessels.
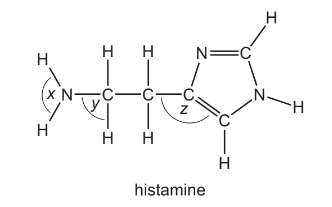
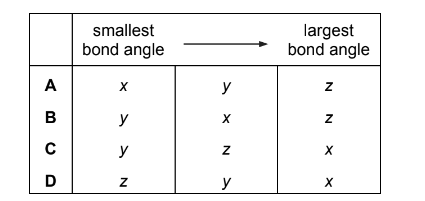
What are the bond angles x, y and z in histamine, from the smallest to the largest?
▶️ Answer/Explanation
Step 1: Identify the geometry at each labelled position
- x is around the nitrogen in the –NH2 group. Nitrogen has 3 bonding pairs and 1 lone pair, giving a trigonal pyramidal shape with bond angle ≈ 107°.
- y is around a carbon with four single bonds (sp³ hybridised). This gives a tetrahedral shape with bond angle ≈ 109.5°.
- z is in the aromatic ring where atoms are sp² hybridised. This gives a trigonal planar geometry with bond angle ≈ 120°.
Step 2: Arrange from smallest to largest
x ≈ 107° < y ≈ 109.5° < z ≈ 120°
This corresponds to option A.
Ans: A
Urea is a product of animal metabolism. It can also be used as a fertiliser.
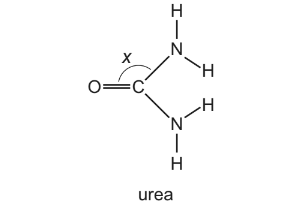
The diagram shows angle x in this molecule.
Which statements about the structure of urea are correct?
1 Angle x is approximately 120°.
2 The molecule has two π bonds.
3 The molecule has only three lone pairs of electrons.
▶️ Answer/Explanation
Step 1: Geometry around the carbon atom
The carbon atom in urea is bonded to:
- one oxygen by a C=O double bond
- two nitrogen atoms by C–N single bonds
This gives three regions of electron density, meaning the carbon is sp² hybridised with a trigonal planar geometry.
Therefore the bond angle at x is approximately 120°.
Statement 1 → Correct
Step 2: Number of π bonds
The structure contains only one double bond (C=O). Each double bond contains one π bond.
So the molecule has one π bond, not two.
Statement 2 → Incorrect
Step 3: Count lone pairs
- Oxygen has 2 lone pairs
- Each nitrogen has 1 lone pair
Total lone pairs:
2 (O) + 1 (N) + 1 (N) = 4 lone pairs
So the molecule does not have only three lone pairs.
Statement 3 → Incorrect
Correct combination: Only statement 1 is correct.
According to the table of responses, this corresponds to Option D.
Ans: D
\(AlCl_{3}\) vapour forms molecules with formula \(Al_{2}Cl_{6}\) as it is cooled.
What happens to the bond angles during the change from \(AlCl_{3}\) to \(Al_{2}Cl_{6}\)?
A Some decrease, some remain the same.
B Some increase, some remain the same.
C They all decrease.
D They all increase.
▶️ Answer/Explanation
Step 1: Structure of AlCl₃
- AlCl₃ is trigonal planar.
- Bond angles are approximately 120°.
Step 2: Structure of Al₂Cl₆
When cooled, two AlCl₃ molecules dimerise to form \(Al_{2}Cl_{6}\).
- Two chlorine atoms form bridges between the aluminium atoms.
- The geometry around aluminium becomes closer to tetrahedral.
- Bond angles become smaller than 120°.
Step 3: Compare bond angles
In AlCl₃ → 120°
In Al₂Cl₆ → smaller angles (around tetrahedral arrangements)
Therefore, the bond angles decrease during dimer formation.
Ans: C
Valence shell electron pair repulsion (VSEPR) theory should be used to answer this question.
Which species are trigonal planar?
1 \(BH_3\)
2 \(CH_3^+\)
3 \(PH_3\)
The responses A to D should be selected on the basis of
▶️ Answer/Explanation
Step 1: Apply VSEPR theory
Molecular shape depends on the number of electron pairs around the central atom.
- BH₃: Boron forms three bonds and has no lone pairs → trigonal planar → bond angles ≈ 120°.
- CH₃⁺: Carbon forms three bonds and has no lone pairs → trigonal planar → bond angles ≈ 120°.
- PH₃: Phosphorus forms three bonds and has one lone pair → trigonal pyramidal → bond angles ≈ 93°.
Step 2: Identify trigonal planar species
BH₃ and CH₃⁺ are trigonal planar.
PH₃ is not trigonal planar.
Correct combination: 1 and 2
Ans: B
In which pair do the molecules have the same shape as each other?
- H2O and CO2
- H2O and SCl2
- NH3 and BH3
- SCl2 and BeCl2
▶️ Answer/Explanation
Step 1: Determine molecular shapes using VSEPR theory
- H₂O: 2 bonding pairs + 2 lone pairs → bent (V-shaped).
- CO₂: 2 bonding regions, no lone pairs → linear.
Option A: Shapes are different.
- SCl₂: 2 bonding pairs + 2 lone pairs → bent.
Option B: Both H₂O and SCl₂ are bent → same shape.
- NH₃: trigonal pyramidal.
- BH₃: trigonal planar.
Option C: Shapes are different.
- BeCl₂: linear.
Option D: Shapes are different.
Therefore, the correct pair with the same shape is:
H₂O and SCl₂
Ans: B
Sodium borohydride, NaBH4, and boron trifluoride, BF3, are compounds of boron.
What are the shapes around boron in the borohydride ion and in boron trifluoride?
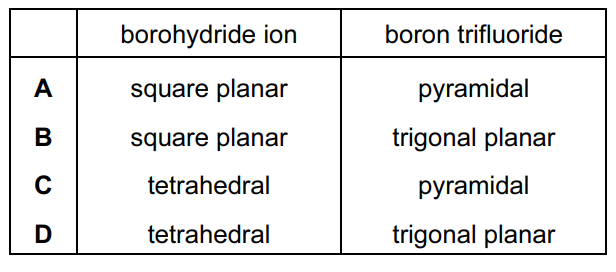
▶️ Answer/Explanation
Step 1: Shape of the borohydride ion, BH4−
- Boron forms four B–H bonds.
- There are no lone pairs on boron.
- Total electron regions = 4.
According to VSEPR theory, this gives a tetrahedral geometry with bond angles ≈ 109.5°.
Step 2: Shape of boron trifluoride, BF3
- Boron forms three B–F bonds.
- There are no lone pairs on boron.
- Total electron regions = 3.
This gives a trigonal planar geometry with bond angles ≈ 120°.
Step 3: Conclusion
BH4− → tetrahedral
BF3 → trigonal planar
Ans: D
Two conversions are outlined below.
NH4+ → NH3
C2H4 → C2H6
What similar feature do these two conversions have?
- a lone pair of electrons in the product
- change in oxidation state of an element
- decrease in bond angle of the species involved
- disappearance of a π bond
▶️ Answer/Explanation
Step 1: Analyse the first conversion
NH4+ → NH3
- NH4+ has a tetrahedral shape with bond angles ≈ 109.5°.
- NH3 has a trigonal pyramidal shape due to one lone pair.
- The lone pair–bond pair repulsion reduces the bond angle to about 107°.
Therefore, the bond angle decreases.
Step 2: Analyse the second conversion
C2H4 → C2H6
- C2H4 (ethene) has sp² hybridised carbon with bond angles ≈ 120°.
- C2H6 (ethane) has sp³ hybridised carbon with bond angles ≈ 109.5°.
Again, the bond angle decreases.
Step 3: Identify the common feature
In both reactions, the bond angles become smaller.
Ans: C
