Pre AP Chemistry -2.2F Molecular Polarity- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.2F Molecular Polarity- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.2F Molecular Polarity- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which of the following diatomic molecules is nonpolar?
(A) \(HCl\)
(B) \(HF\)
(C) \(Cl_2\)
(D) \(NO\)
▶️ Answer / Explanation
A bond is nonpolar when the two atoms sharing it have equal electronegativity, so the electron pair is shared symmetrically with no partial charges (\(\delta^+\) / \(\delta^-\)).
\(Cl_2\) consists of two identical chlorine atoms → same electronegativity → zero dipole moment → nonpolar.
\(HCl\), \(HF\), and \(NO\) all involve atoms with different electronegativities, creating polar bonds and therefore polar molecules.
Answer: C
What two factors determine whether a molecule is polar or nonpolar?
(A) Molecular mass and number of atoms
(B) Bond polarity and molecular geometry (shape)
(C) Number of lone pairs and atomic radius
(D) Temperature and pressure
▶️ Answer / Explanation
Molecular polarity depends on two things working together:
1. Bond polarity — each bond must have a dipole (difference in electronegativity between the bonded atoms).
2. Molecular geometry — even if all bonds are polar, they may cancel out if the shape is perfectly symmetrical.
Answer: B
\(CO_2\) has two polar \(C{=}O\) bonds, yet the molecule has a net dipole moment of zero. Which property explains this?
(A) The molecule is bent
(B) The molecule is linear and symmetric so dipoles cancel
(C) Carbon and oxygen have the same electronegativity
(D) Lone pairs cancel
▶️ Answer / Explanation
\(CO_2\) is a linear molecule (O=C=O). The two bond dipoles point in opposite directions and cancel.
Answer: B
In which direction does the dipole arrow point in a polar bond?
(A) Toward less electronegative atom
(B) Toward more electronegative atom
(C) Perpendicular to the bond
(D) Random direction
▶️ Answer / Explanation
The dipole arrow points toward the more electronegative atom.
Answer: B
Which molecule below is polar?
(A) \(BeCl_2\)
(B) \(BCl_3\)
(C) \(CCl_4\)
(D) \(NH_3\)
▶️ Answer / Explanation
\(NH_3\) has a trigonal pyramidal shape due to a lone pair on nitrogen, creating an asymmetric molecule and a net dipole.
Answer: D
Both \(SO_2\) and \(CO_2\) contain one central atom double-bonded to two oxygen atoms, yet \(SO_2\) is polar and \(CO_2\) is nonpolar. What is the best explanation?
(A) Oxygen is more electronegative when bonded to sulfur than to carbon
(B) \(SO_2\) has a lone pair on sulfur making it bent; \(CO_2\) is linear and symmetric
(C) Sulfur forms stronger bonds than carbon, creating larger dipoles
(D) \(CO_2\) has more electrons, causing its dipoles to cancel
▶️ Answer / Explanation
The key is electron geometry vs. molecular geometry:
• \(CO_2\): linear → dipoles cancel → nonpolar.
• \(SO_2\): bent due to a lone pair → dipoles do not cancel → polar.
Answer: B
The table below lists four molecules. Which molecule has polar bonds but is overall nonpolar?
| Molecule | Shape |
|---|---|
| \(H_2O\) | Bent |
| \(CHCl_3\) | Tetrahedral |
| \(CCl_4\) | Tetrahedral |
| \(HCN\) | Linear |
(A) \(H_2O\)
(B) \(CHCl_3\)
(C) \(CCl_4\)
(D) \(HCN\)
▶️ Answer / Explanation
\(CCl_4\) has four polar C–Cl bonds but a perfectly symmetric tetrahedral geometry, so all dipoles cancel.
Answer: C
A molecule has the formula \(AX_2E_2\). What is the molecular geometry and polarity?
(A) Linear; nonpolar
(B) Bent; polar
(C) Trigonal planar; nonpolar
(D) Tetrahedral; polar
▶️ Answer / Explanation
\(AX_2E_2\) has tetrahedral electron geometry but bent molecular geometry (like \(H_2O\)).
The asymmetry causes a net dipole.
Answer: B
A student claims: “If a molecule contains only nonpolar bonds, it must be nonpolar.”
(A) Always true
(B) Always false
(C) True because nonpolar bonds have zero dipole moments
(D) Only true for diatomic molecules
▶️ Answer / Explanation
If all bonds are nonpolar, each bond dipole is zero. The sum of zero dipoles is always zero.
Answer: A
Which bond is the most polar?
(A) C–H
(B) N–H
(C) O–H
(D) C–O
▶️ Answer / Explanation
Bond polarity increases with electronegativity difference.
O–H has the largest difference, so it is the most polar bond.
Answer: C
\(SF_4\) has the molecular formula \(AX_4E_1\). What is its molecular geometry and is it polar or nonpolar?
(A) Tetrahedral; nonpolar — four identical bonds cancel symmetrically
(B) See-saw; polar — the lone pair creates an asymmetric arrangement
(C) Square planar; nonpolar — the four F atoms arrange symmetrically
(D) Trigonal bipyramidal; nonpolar — the lone pair occupies an equatorial position and cancels dipoles
▶️ Answer / Explanation
\(SF_4\) has 5 electron domains (4 bonding + 1 lone pair) → trigonal bipyramidal electron geometry.
The lone pair occupies an equatorial position, producing a see-saw molecular geometry. The arrangement is asymmetric so dipoles do not cancel.
Answer: B
Consider the molecule \(XeF_2\). Xenon has 3 lone pairs and 2 bonding pairs. What are the electron geometry, molecular geometry, and polarity of \(XeF_2\)?
(A) Trigonal bipyramidal; linear; nonpolar
(B) Octahedral; bent; polar
(C) Trigonal bipyramidal; bent; polar
(D) Linear; linear; nonpolar
▶️ Answer / Explanation
\(XeF_2\) has 5 electron domains → trigonal bipyramidal electron geometry.
The three lone pairs occupy equatorial positions leaving two F atoms in axial positions (180° apart).
Molecular geometry is linear and the dipoles cancel → nonpolar.
Answer: A
Two molecules, are shown.
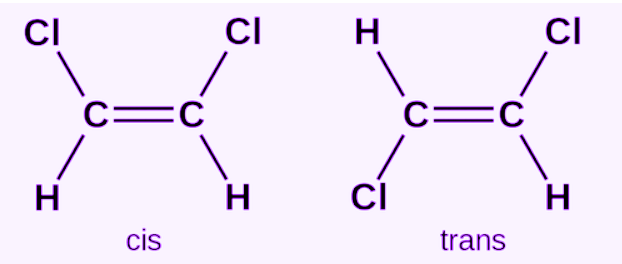
The cis isomer has Cl and Cl on the same side; the trans isomer has them on opposite sides.
Which statement about their polarities is correct?
(A) Both are nonpolar because they contain the same atoms
(B) Both are polar because all their bonds are polar
(C) The cis isomer is polar; the trans isomer may be nonpolar if dipoles cancel
(D) The trans isomer is always more polar
▶️ Answer / Explanation
Polarity depends on geometry.
• In the cis isomer, dipoles reinforce → polar.
• In the trans isomer, opposing dipoles may cancel → less polar or nonpolar.
Answer: C
A molecule \(AB_3\) is experimentally found to have a net dipole moment of zero. Which geometry could it have?
(A) Trigonal pyramidal
(B) Trigonal planar
(C) Trigonal planar or T-shaped
(D) Trigonal pyramidal or T-shaped
▶️ Answer / Explanation
Trigonal planar structures with identical atoms are perfectly symmetrical so dipoles cancel.
Trigonal pyramidal and T-shaped geometries are asymmetric and therefore polar.
Answer: B
A chemist replaces one F atom in \(CF_4\) with Cl to form \(CF_3Cl\). What happens to the polarity?
(A) Nonpolar — tetrahedral geometry cancels dipoles
(B) Nonpolar — C–F and C–Cl bonds have identical polarity
(C) Polar — symmetry is broken and dipoles do not cancel
(D) Polar — London dispersion forces create the dipole
▶️ Answer / Explanation
\(CF_4\) is nonpolar because all four C–F dipoles cancel symmetrically.
Replacing one F with Cl breaks the symmetry, so the dipoles no longer cancel.
The molecule becomes polar.
Answer: C
