Pre AP Chemistry -2.3A Types of Chemical Bonding- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.3A Types of Chemical Bonding- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.3A Types of Chemical Bonding- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
The table shows some properties of four substances.

Which substances are ionic?
A) 1, 3 and 4
B) 1 and 3 only
C) 2 and 4
D) 2 only
▶️ Answer/Explanation
Ans: D
Ionic compounds typically have:
1. High melting points (eliminates substance 3)
2. Poor conductivity when solid (ions can’t move)
3. Good conductivity when molten (ions are free to move)
Only substance 2 fits all these criteria. Substance 4 conducts in solid state, suggesting metallic bonding. Substance 1 is likely covalent (poor conductor in both states). Therefore, only substance 2 is ionic.
The diagram shows part of an ionic lattice structure.
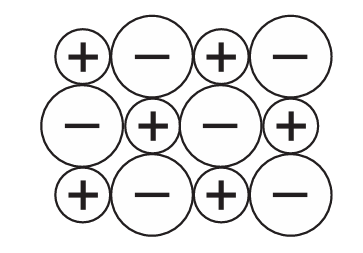
Which compound does the diagram represent?
A) potassium bromide
B) sodium oxide
C) magnesium chloride
D) carbon monoxide
▶️ Answer/Explanation
Ans: A
The question implies the diagram shows a simple 1:1 ionic lattice (one positive ion for each negative ion). Let’s analyze the options:
A) KBr – potassium forms 1+ ions, bromine forms 1- ions → 1:1 ratio
B) Na₂O – sodium forms 1+ ions, oxygen forms 2- ions → 2:1 ratio
C) MgCl₂ – magnesium forms 2+ ions, chlorine forms 1- ions → 1:2 ratio
D) CO – covalent molecular compound, not ionic
Only potassium bromide (KBr) forms a simple 1:1 ionic lattice among the options.
Which statement describes the structure of an ionic compound?
A. It is a giant lattice of oppositely charged ions.
B. It is a giant lattice of positive ions in a ‘sea’ of electrons.
C. It is a giant molecule of oppositely charged ions.
D. It is a simple molecule of oppositely charged ions.
▶️ Answer/Explanation
Ans: A
Ionic compounds consist of a giant lattice structure where positively charged ions (cations) and negatively charged ions (anions) are held together by strong electrostatic forces. Option B describes metallic bonding, while options C and D incorrectly refer to “molecules,” which do not apply to ionic compounds. Thus, the correct description is A.
The electronic structures of two atoms, P and Q, are shown.

P and Q combine together to form a compound.
What is the type of bonding in the compound and what is the formula of the compound?

▶️ Answer/Explanation
Ans: A
From the electronic configurations:
1. Atom P has 2 valence electrons (group 2 metal, likely Mg).
2. Atom Q has 6 valence electrons (group 16 non-metal, likely O).
P loses 2 electrons to form \(\text{P}^{2+}\), and Q gains 2 electrons to form \(\text{Q}^{2-}\).
The resulting ionic compound has the formula \(\text{PQ}\) (e.g., MgO).
Thus, the bonding is ionic, and the formula is \(\text{PQ}\).
The structure of copper is described as a lattice of positive ions in a ‘sea of electrons’.
Which statements are correct?
- Copper has a high melting point because of the strong electrostatic attraction between the positive ions and the ‘sea of electrons’.
- Copper is malleable because the layers of atoms in the lattice can slide over each other.
- Copper atoms can be oxidized to form copper ions by losing electrons.
A) 1, 2, and 3
B) 1 and 2 only
C) 1 and 3 only
D) 2 and 3 only
▶️ Answer/Explanation
Ans: A
Copper is a metallic solid with the following properties:
1. It has a high melting point due to strong metallic bonds between positive ions and delocalized electrons (statement 1 is correct).
2. It is malleable because layers of ions can slide without breaking bonds (statement 2 is correct).
3. Copper oxidizes to \(\text{Cu}^{2+}\) by losing electrons (statement 3 is correct).
Therefore, all three statements (1, 2, and 3) are correct.
Which statement about metals is correct?
A) Layers of positive ions can slide over each other making metals malleable.
B) Metallic bonding consists of a lattice of negative ions in a sea of delocalised electrons.
C) Metallic bonding consists of a lattice of positive ions in a sea of delocalised negative ions.
D) Metals conduct electricity because positive ions are free to move.
▶️ Answer/Explanation
Ans: A
1. Malleability (A): Metals are malleable because layers of positive ions can slide over one another without breaking bonds due to the sea of delocalized electrons holding them together.
2. Metallic Bonding (B & C): The correct description is a lattice of positive ions in a sea of delocalized electrons (not negative ions), so B and C are incorrect.
3. Electrical Conductivity (D): Metals conduct electricity due to the movement of delocalized electrons, not positive ions, making D incorrect.
$\mathrm{X}$ is a solid at room temperature.
$X$ has a high melting point.
Solid $X$ conducts electricity.
Which diagram shows how the particles are arranged in solid $X$ ?
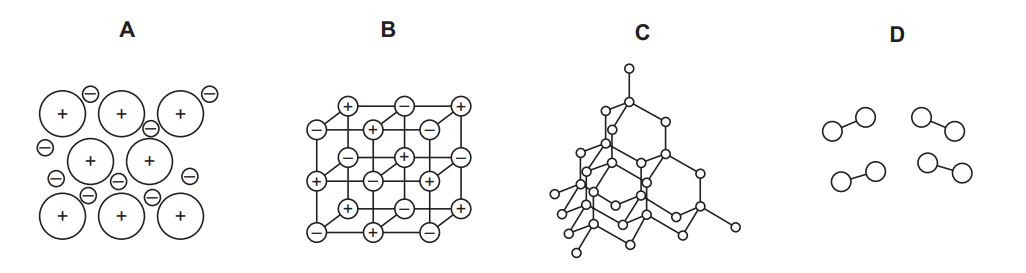
▶️ Answer/Explanation
Ans: A
Given the properties of $\mathrm{X}$:
– Solid at room temperature with a high melting point suggests a giant structure (ionic or metallic).
– Electrical conductivity in solid state rules out ionic compounds (they conduct only when molten/dissolved).
– Thus, $\mathrm{X}$ must be a metallic solid, where positive ions are arranged in a lattice with a ‘sea’ of delocalized electrons (Diagram A).
Diagrams B (simple molecular), C (ionic), and D (covalent network) do not fit all the given properties.
The diagram represents the general structure of a solid Z.
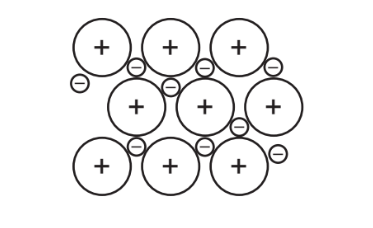
What is Z?
A) aluminium
B) iodine
C) silicon dioxide
D) sulfur
▶️ Answer/Explanation
Ans: A
The diagram shows a giant metallic lattice, which is characteristic of metals like aluminium.
1. Aluminium (A) forms a closely packed 3D lattice of positive ions surrounded by delocalized electrons.
2. Iodine (B) exists as diatomic molecules (\(I_2\)) in a molecular lattice.
3. Silicon dioxide (C) has a giant covalent structure with a tetrahedral arrangement.
4. Sulfur (D) typically forms \(S_8\) rings, not a continuous lattice.
Thus, the correct answer is A (aluminium).
Which structure represents the sodium chloride lattice?
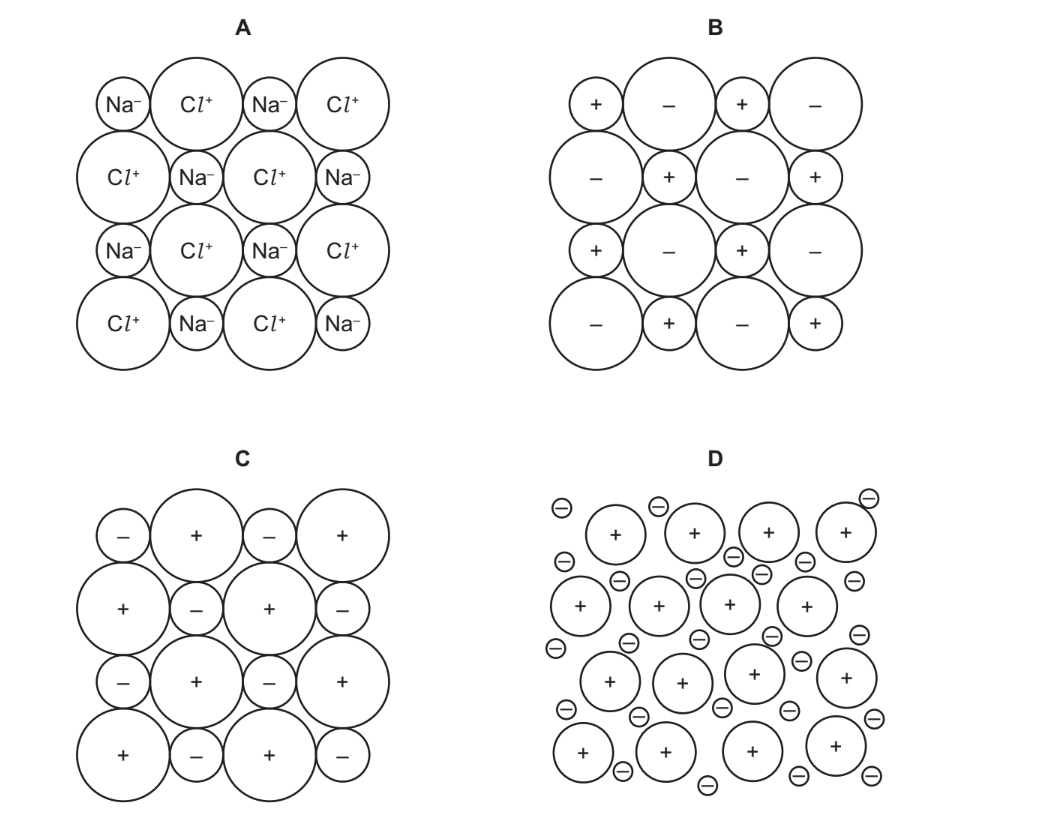
▶️ Answer/Explanation
Ans: B
The sodium chloride (NaCl) lattice has the following characteristics:
1. It forms a face-centered cubic (FCC) arrangement.
2. Each Na+ ion is surrounded by 6 Cl− ions, and vice versa (6:6 coordination).
3. Structure B correctly shows this alternating 3D pattern of Na+ and Cl− ions.
The other structures (A, C, D) either show incorrect coordination numbers or non-ionic arrangements.
Which diagram shows the covalent bonding in a molecule of carbon dioxide?
A) O–C–O
B) O=C–O
C) O=C=O
D) O≡C≡O
▶️ Answer/Explanation
Ans: C
Carbon dioxide (CO₂) has a linear molecular geometry with double bonds between the carbon atom and each oxygen atom. Each oxygen shares two pairs of electrons with the central carbon atom, forming two double bonds (O=C=O).
Option A shows single bonds which is incorrect. Option B shows one double and one single bond which doesn’t represent CO₂’s symmetrical structure. Option D shows a triple bond which doesn’t occur in CO₂.
Which diagram represents the outer-shell electron arrangement in a nitrogen molecule?

▶️ Answer/Explanation
Ans: D
A nitrogen molecule (\(N_2\)) forms a triple covalent bond, sharing three pairs of electrons. Each nitrogen atom has 5 valence electrons, and in \(N_2\), they share 3 electrons each to complete their octet. Diagram D correctly shows 6 shared electrons (3 pairs) between the two nitrogen atoms, with 2 lone pairs on each nitrogen. This satisfies the octet rule and represents the Lewis structure of \(N_2\).
How many electrons are used to form covalent bonds in a molecule of methanol, CH3OH?
A) 5
B) 6
C) 8
D) 10
▶️ Answer/Explanation
Ans: D
In methanol (CH3OH), the covalent bonds are formed as follows:
1. C-H bonds: 3 single bonds (each uses 2 electrons) → Total = 6 electrons.
2. C-O bond: 1 single bond → 2 electrons.
3. O-H bond: 1 single bond → 2 electrons.
Summing up, \(6 + 2 + 2 = 10\) electrons are used in covalent bonding.
A covalent molecule Q contains only six shared electrons.
What is Q?
A) ammonia, \(NH_3\)
B) chlorine, \(Cl_2\)
C) methane, \(CH_4\)
D) water, \(H_2O\)
▶️ Answer/Explanation
Ans: A
To determine the correct molecule:
- Ammonia (\(NH_3\)) has 3 N-H bonds, contributing 6 shared electrons (2 per bond).
- Chlorine (\(Cl_2\)) has 1 Cl-Cl bond (2 shared electrons).
- Methane (\(CH_4\)) has 4 C-H bonds (8 shared electrons).
- Water (\(H_2O\)) has 2 O-H bonds (4 shared electrons).
Only ammonia (\(NH_3\)) matches the condition of having exactly six shared electrons.
Which statement describes the attractive forces between molecules?
- They are strong covalent bonds which hold molecules together.
- They are strong ionic bonds which hold molecules together.
- They are weak forces formed between covalently-bonded molecules.
- They are weak forces which hold ions together in a lattice.
▶️ Answer/Explanation
Ans: C
The attractive forces between molecules are called intermolecular forces, which are much weaker than covalent or ionic bonds. These forces (e.g., van der Waals, dipole-dipole, hydrogen bonds) act between covalently-bonded molecules. Options A and B are incorrect because covalent/ionic bonds are intramolecular (within molecules), not intermolecular. Option D is incorrect because ions are held by strong electrostatic forces, not weak ones.
Which row describes the formation of single covalent bonds in methane?
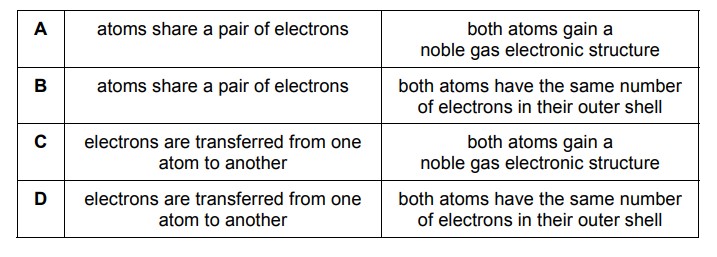
▶️ Answer/Explanation
Ans: A
In methane (CH4), carbon shares one electron with each of the four hydrogen atoms, forming four single covalent bonds. Each bond consists of a shared pair of electrons (one from carbon and one from hydrogen). The table row corresponding to this description is row A, as it correctly states that electrons are shared between carbon and hydrogen atoms.
Which statement explains why methane has a lower boiling point than water?
A) Methane has weaker covalent bonds than water.
B) Methane has weaker attractive forces than water.
C) Methane molecules are heavier than water molecules.
D) Methane molecules have more bonds than water molecules.
▶️ Answer/Explanation
Ans: B
The boiling point depends on the strength of intermolecular forces, not covalent bonds (A is incorrect). Methane (\(CH_4\)) has weaker London dispersion forces, while water (\(H_2O\)) has strong hydrogen bonding (B is correct). Methane is lighter than water (C is wrong), and it has fewer bonds (D is wrong). Thus, weaker attractive forces in methane lead to a lower boiling point.
Which diagram shows the outer shell electron arrangement in a molecule of methanol, $\mathrm{CH}_3 \mathrm{OH}$?

▶️ Answer/Explanation
Ans: A
Methanol ($\mathrm{CH}_3 \mathrm{OH}$) has the following electron arrangement:
1. Carbon forms 4 single bonds (3 with H and 1 with O).
2. Oxygen forms 2 single bonds (1 with C and 1 with H) and has 2 lone pairs.
3. Hydrogen atoms share 1 electron each (no lone pairs).
Diagram A correctly shows this structure, with lone pairs on oxygen and proper bonding.
The structure of ethanoic acid is shown.
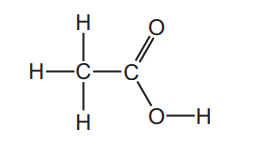
Which diagram shows the arrangement of outer shell electrons in a molecule of ethanoic acid?
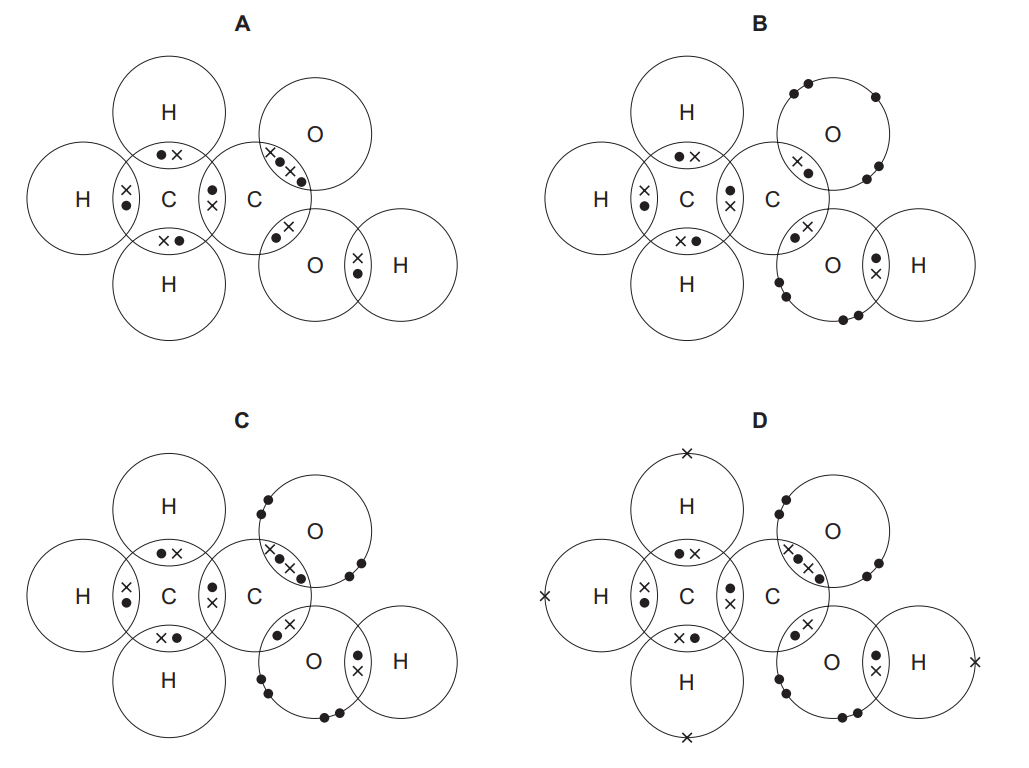
▶️ Answer/Explanation
Ans: C
To determine the correct electron arrangement:
- Ethanoic acid (\(CH_3COOH\)) has a carboxyl group (\(-COOH\)) and a methyl group (\(-CH_3\)).
- The C=O double bond in the carboxyl group consists of 4 shared electrons (2 pairs).
- The O-H bond has 2 shared electrons, and the C-O bond has 2 shared electrons.
- Option C correctly shows these bonds with the appropriate lone pairs on oxygen.
Thus, the correct answer is C, as it accurately represents the outer shell electron arrangement in ethanoic acid.
The table shows the electronic structure of four atoms.
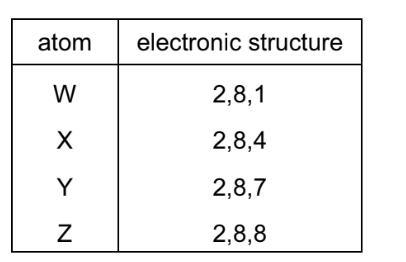
Which two atoms combine to form a covalent compound?
- W and X
- W and Y
- X and Y
- X and Z
▶️ Answer/Explanation
Ans: C
Covalent compounds form when two non-metal atoms share electrons to achieve stable configurations. From the table:
- X (2,6) needs 2 electrons to complete its outer shell (oxygen-like).
- Y (2,4) needs 4 electrons (carbon-like).
They can share electrons (e.g., forming CO2), making X and Y the correct pair. Options A, B, and D involve metals (W/Z), which typically form ionic bonds, not covalent.
Which statement describes the attractive forces between molecules (intermolecular forces)?
A) They are strong covalent bonds which hold molecules together.
B) They are strong ionic bonds which hold molecules together.
C) They are weak forces formed between covalently-bonded molecules.
D) They are weak forces which hold ions together in a lattice.
▶️ Answer/Explanation
Ans: C
Intermolecular forces are weak attractions between molecules (e.g., van der Waals forces, hydrogen bonds). They differ from intramolecular forces (covalent/ionic bonds) that hold atoms together within a molecule.
A and B are incorrect because covalent/ionic bonds are intramolecular, not intermolecular. D is wrong because ionic lattices involve ions, not molecules. Thus, C is correct—intermolecular forces are weak and act between covalently-bonded molecules.
Which molecule contains only single covalent bonds?
A) \(Cl_2\)
B) \(CO_2\)
C) \(N_2\)
D) \(O_2\)
▶️ Answer/Explanation
Ans: A
1. \(Cl_2\) (A): Chlorine forms a single covalent bond (Cl-Cl), as each atom shares one electron.
2. \(CO_2\) (B): Contains double bonds (C=O), not single bonds.
3. \(N_2\) (C): Has a triple bond (N≡N), not single bonds.
4. \(O_2\) (D): Contains a double bond (O=O), not single bonds.
Thus, only \(Cl_2\) has exclusively single covalent bonds.
Why does ICl have a higher boiling point than Br₂?
▶️ Answer/Explanation
Ans: B
ICl has a higher boiling point than Br₂ because it is a polar molecule (due to the electronegativity difference between I and Cl), leading to stronger permanent dipole-dipole interactions. Br₂ is nonpolar and only has weaker London dispersion forces, even though both have similar molecular masses. Thus, the polar nature of ICl (Option B) is the correct explanation.
The diagram shows part of the lattice structures of solids X and Y.
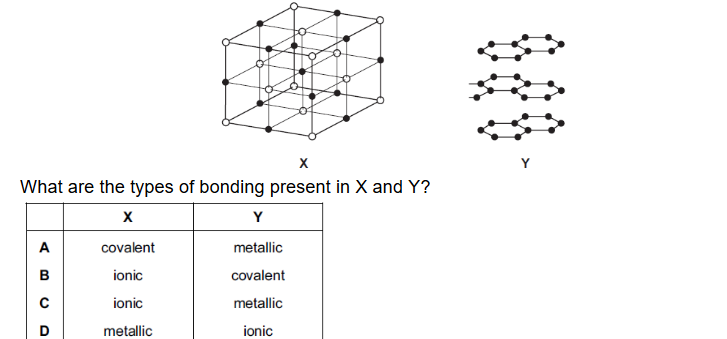
▶️ Answer / Explanation
X: The structure shows alternating particles arranged in a cubic lattice, typical of an ionic lattice where positive and negative ions alternate.
Y: The layered hexagonal sheets represent a giant covalent structure (similar to graphite), where atoms are covalently bonded within layers.
Therefore:
X → ionic bonding
Y → covalent bonding
Correct Answer: B
Silver and iodine are both shiny crystalline solids.
Which forces exist between neighboring iodine molecules in solid iodine and particles in solid silver?
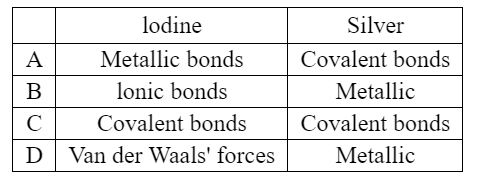
▶️ Answer / Explanation
Iodine: Solid iodine consists of \(I_2\) molecules. The molecules are held together by weak intermolecular (van der Waals / London dispersion) forces.
Silver: Silver is a metal. The particles are held together by metallic bonding (attraction between positive metal ions and delocalised electrons).
Therefore the correct combination is:
van der Waals forces in iodine and metallic bonding in silver.
Answer: D
For which substance is the type of bonding not correct?
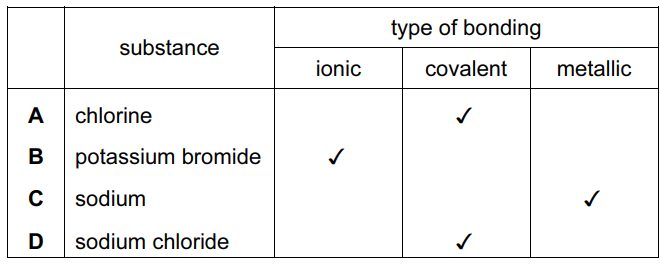
▶️ Answer / Explanation
A. Chlorine exists as \(Cl_2\) molecules with covalent bonding. ✔
B. Potassium bromide (\(KBr\)) consists of \(K^+\) and \(Br^-\) ions, so it has ionic bonding. ✔
C. Sodium is a metal and should have metallic bonding, not covalent bonding as shown. ✘
D. Sodium chloride (\(NaCl\)) forms an ionic lattice with ionic bonding. ✔
Incorrect bonding: C
Answer: C
Which row explains the malleability and electrical conductivity of a solid metal?
| malleability | electrical conductivity | |
|---|---|---|
| A | Delocalised electrons can move freely through the structure. | Delocalised electrons can move freely through the structure. |
| B | Delocalised electrons can move freely through the structure. | Positive ions can move freely through the structure. |
| C | Rows of positive ions can slide over each other. | Delocalised electrons can move freely through the structure. |
| D | Rows of positive ions can slide over each other. | Positive ions can move freely through the structure. |
▶️ Answer/Explanation
Ans: C
Metals are malleable because their positive ions are arranged in layers that can slide over each other when force is applied, while the sea of delocalized electrons maintains the metallic bonding.
Electrical conductivity in metals is due to the movement of delocalized electrons (not positive ions, making options B and D incorrect). Option A incorrectly attributes malleability to electron movement rather than ion layer movement.
Four substances, P, Q, R and S, are described.
- P is diatomic.
- Q is a good conductor of electricity when solid and when molten.
- R is a silver solid with a very high melting point.
- S reacts with oxygen to form a brown gas.
Which substances are metals?
A) P and Q
B) P and S
C) Q and R
D) R and S
▶️ Answer/Explanation
Ans: C
Let’s analyze each substance’s properties to identify metals:
P (diatomic): Most metals aren’t diatomic (except some like Hg in vapor). This suggests a non-metal (like O₂, N₂).
Q (conducts electricity when solid/molten): This is characteristic of metals (metallic bonding with delocalized electrons).
R (silver solid, high melting point): Typical metal properties (silvery luster, high m.p. due to strong metallic bonds).
S (forms brown gas with O₂): Likely a non-metal (e.g., nitrogen forms brown NO₂).
Therefore:
– Q and R show metallic properties
– P and S are non-metals
Metals are characterized by:
– Electrical conductivity
– Malleability/ductility
– Shiny luster
– High melting points (usually)
Which row contains a description of metallic bonding and a property that is explained by reference to metallic bonding?
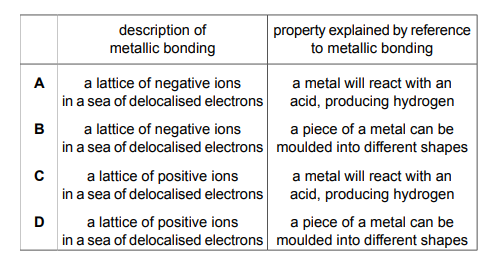
▶️ Answer/Explanation
Ans: D
Metallic bonding involves a lattice of positive ions surrounded by a “sea” of delocalized electrons. This structure explains key metallic properties:
- Electrical conductivity (Row D): Delocalized electrons move freely, carrying current.
- Malleability: Layers of ions can slide without breaking bonds.
Rows A–C are incorrect because:
- A describes ionic bonding (high MP due to strong electrostatic forces).
- B describes covalent bonding (localized electrons).
- C incorrectly links metallic bonding to brittleness (a property of ionic compounds).
Which statement describes the structure of copper?
- It has a lattice of negative ions in a ‘sea of electrons’.
- It has a lattice of negative ions in a ‘sea of protons’.
- It has a lattice of positive ions in a ‘sea of electrons’.
- It has a lattice of positive ions in a ‘sea of protons’.
▶️ Answer/Explanation
Ans: C
Copper is a metallic solid with a lattice of positive ions (Cu+) surrounded by a delocalized ‘sea of electrons’.
- A and B are incorrect because metals form positive ions, not negative ones.
- D is wrong because protons are not delocalized—only valence electrons are.
- C is correct: the ‘sea of electrons’ explains copper’s conductivity and malleability.
Which row contains a description of metallic bonding and a property that is explained by reference to metallic bonding?

▶️ Answer/Explanation
Ans: D
1. Metallic bonding involves a lattice of positive ions surrounded by a “sea” of delocalized electrons.
2. Electrical conductivity is a key property explained by metallic bonding, as delocalized electrons move freely under a potential difference.
3. Rows A, B, and C either describe incorrect bonding types or unrelated properties.
4. Row D correctly describes metallic bonding and links it to electrical conductivity, making it the correct choice.
