Pre AP Chemistry -2.3B Properties of Ionic and Covalent Compounds- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.3B Properties of Ionic and Covalent Compounds- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.3B Properties of Ionic and Covalent Compounds- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
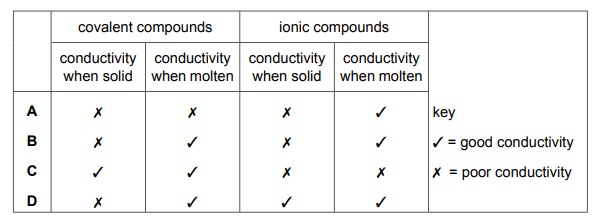
Which row describes the electrical conductivity of covalent and ionic compounds when solid and when molten?
▶️ Answer/Explanation
Ans: A
1. Ionic Compounds: – Solid: No conductivity (ions fixed in lattice) – Molten: Conducts (mobile ions can carry charge)
2. Covalent Compounds: – Solid: No conductivity (no free electrons/ions) – Molten: No conductivity (still no charged particles)
3. Row Analysis: – Only Row A matches this behavior pattern – Other rows incorrectly show conductivity for covalent compounds or solid ionic compounds
4. Key Concept: – Conductivity requires mobile charge carriers – Ionic compounds provide these only when molten/dissolved – Simple covalent compounds never conduct
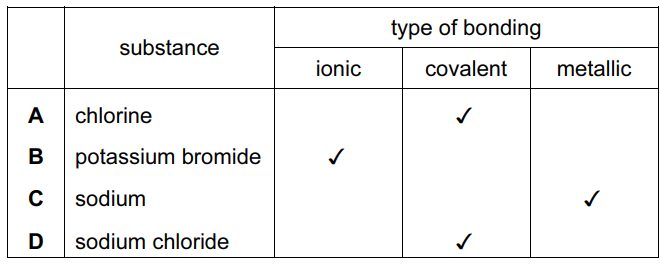
For which substance is the type of bonding not correct?
▶️ Answer/Explanation
Ans: D
1. Analysis of each option:
– A) CO2: Correctly identified as covalent (non-metal + non-metal)
– B) MgO: Correctly identified as ionic (metal + non-metal)
– C) H2O: Correctly identified as covalent (non-metal + non-metal)
– D) NaCl: Incorrectly labeled as covalent (should be ionic)
2. Why D is wrong:
– Sodium (metal) + Chlorine (non-metal) always form ionic bonds
– NaCl forms a crystal lattice through electron transfer (Na→Na+ + Cl→Cl–)
3. Key distinction:
– Covalent bonds involve electron sharing (between non-metals)
– Ionic bonds involve electron transfer (between metals and non-metals)
Phosphorus forms a compound with hydrogen called phosphine, PH₃. This compound can react with a hydrogen ion, H⁺. Which type of interaction occurs between PH₃ and H⁺?
▶️ Answer/Explanation
Ans: A
Phosphine (PH₃) has a lone pair of electrons on the phosphorus atom. When it reacts with H⁺ (which has an empty 1s orbital), the lone pair is donated to form a dative covalent bond (also called a coordinate bond). This results in the formation of PH₄⁺ (phosphonium ion). Since the bond is formed by sharing a lone pair, the correct answer is A.
In which pairs are both species free radicals?
1 Cl and O
2 \(Cl^–\) and \(O_2^–\)
3 Cl and \(O^–\)
4 Cl⁻ and O₂⁺
▶️ Answer/Explanation
Ans: A
A free radical is a species with an unpaired electron. Analyzing the pairs:
1. Cl and O – Both are neutral atoms with unpaired electrons (radicals).
3. Cl and \(O^–\) – Cl is a radical, and \(O^–\) has an unpaired electron (radical).
4. Cl⁻ and O₂⁺ – Cl⁻ has no unpaired electrons, but O₂⁺ has one unpaired electron (radical).
Thus, pairs 1, 3, and 4 include at least one radical, making option A correct.
In which species is there a lone pair of electrons?
▶️ Answer/Explanation
Ans: C
To find a lone pair, we calculate the total valence electrons for each species. Carbon has 4 valence electrons and hydrogen has 1. For \(CH_3^-\), total electrons = \(4 + (3 \times 1) + 1 = 8\). These form 3 C-H bonds (using 6 electrons), leaving a pair of electrons unbonded on the carbon atom. The other species either have no extra electrons (\(CH_4\), \(CH_3\)) or a deficit (\(CH_3^+\)), so they lack a lone pair.
Which particle contains nitrogen in the same oxidation state as in the ion N₂O₂²⁻?
▶️ Answer/Explanation
Ans: D
First, find the oxidation state of N in N₂O₂²⁻. Let oxidation state of N be x. The ion has charge -2. So, 2x + 2(-2) = -2 → 2x – 4 = -2 → 2x = 2 → x = +1. Now check options: A) NH₂F: H is +1, F is -1, so x + 2(+1) + (-1) = 0 → x + 2 – 1 = 0 → x = -1. B) N₂O₄: 2x + 4(-2)=0 → 2x -8=0 → x=+4. C) NO₃⁻: x + 3(-2) = -1 → x -6 = -1 → x=+5. D) HNF₂: H is +1, F is -1 (each), so x + (+1) + 2(-1) = 0 → x +1 -2 = 0 → x = +1. So D (HNF₂) has N in +1 state, same as N₂O₂²⁻.
Electronegativity differences can be used to help determine the oxidation number of an atom in different species. A number of rules are used which include:
- The more electronegative atom is given a negative oxidation number.
- Hydrogen is more electronegative than Group 1 metals.
- Oxygen is more electronegative than hydrogen.
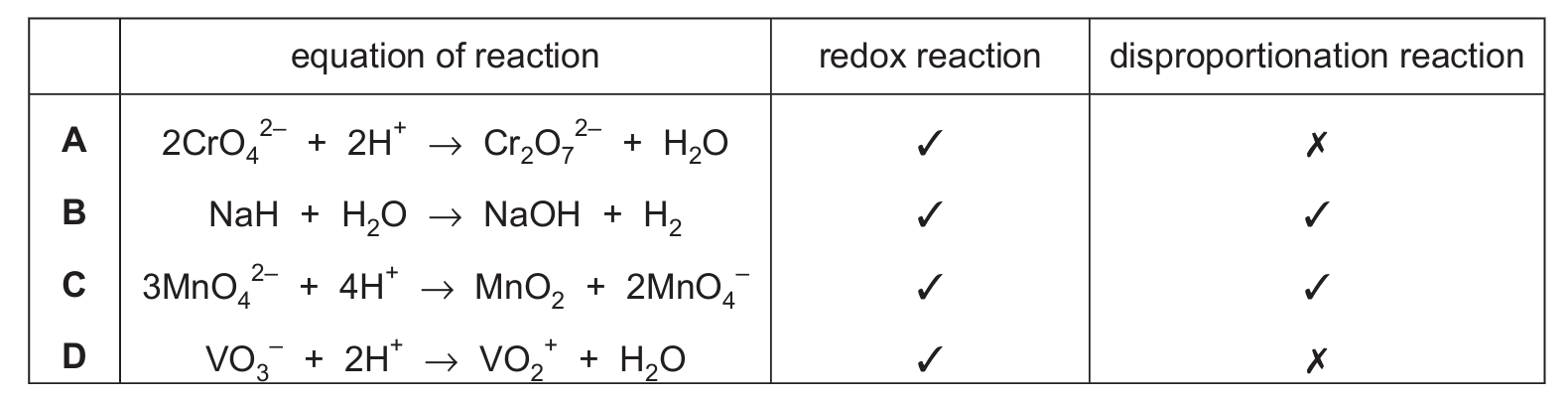
Which row is correct?
▶️ Answer/Explanation
Ans: C
In reaction C, \(3MnO_4^{2-} + 4H^+ \rightarrow MnO_2 + 2MnO_4^{-}\), manganese changes oxidation state. In \(MnO_4^{2-}\), Mn is +6. In \(MnO_2\), it’s +4 (reduction), and in \(MnO_4^-\), it’s +7 (oxidation). The same element is both oxidized and reduced, making it a disproportionation reaction, which is also a type of redox reaction.
The ionic equation shows iodide ions reacting with manganate(VII) ions in acidic solution.
\[uMnO_4^- + vH^+ + wI^- \rightarrow xMn^{2+} + yH_2O + zI_2\]
The letters \( u, v, w, x, y \) and \( z \) all represent whole numbers. Two or more of \( u, v, w, x, y \) and \( z \) are the same as each other.
What is the lowest possible value of \( v \)?
▶️ Answer/Explanation
Ans: D
The balanced half-equations are:
Reduction: \(MnO_4^- + 8H^+ + 5e^- \rightarrow Mn^{2+} + 4H_2O\)
Oxidation: \(2I^- \rightarrow I_2 + 2e^-\)
To balance electrons, multiply reduction by 2 and oxidation by 5: \(2MnO_4^- + 16H^+ + 10e^- + 10I^- \rightarrow 2Mn^{2+} + 8H_2O + 5I_2\).
Thus, the lowest value for \(v\) (H⁺ coefficient) is 16.
Which types of bonding are present in ammonium carbonate, (NH4)2CO3?
1 ionic
2 covalent
3 co-ordinate (dative covalent)
The responses A to D should be selected on the basis of

▶️ Answer / Explanation
Ionic bonding: occurs between \(NH_4^+\) ions and \(CO_3^{2-}\) ions in the ionic lattice.
Covalent bonding: present within the ammonium ion (N–H bonds) and within the carbonate ion (C–O bonds).
Co-ordinate (dative) bonding: in the ammonium ion, nitrogen donates a lone pair to \(H^+\) to form one N→H bond.
Therefore all three bonding types are present.
Correct Answer: A
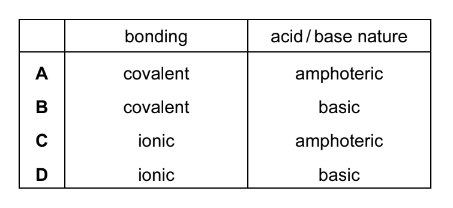
Which description of the bonding and acid/base nature of aluminium oxide is correct?
▶️ Answer / Explanation
Aluminium oxide (\(Al_2O_3\)) contains ionic bonding between \(Al^{3+}\) and \(O^{2-}\) ions in a giant ionic lattice.
However, because the aluminium ion has a high charge density, it polarises the oxide ions, giving the compound some covalent character.
Chemically, aluminium oxide reacts with both acids and bases, so it is amphoteric.
Correct description: ionic bonding and amphoteric behaviour.
Answer: C
Why is the ionic radius of a chloride ion larger than the ionic radius of a sodium ion?
- A chloride ion has one more occupied electron shell than a sodium ion.
- Chlorine has a higher proton number than sodium.
- Ionic radius increases regularly across the third period.
- Sodium is a metal, chlorine is a non-metal.
▶️ Answer / Explanation
Na⁺ electron configuration: \(1s^2 2s^2 2p^6\) → 2 electron shells.
Cl⁻ electron configuration: \(1s^2 2s^2 2p^6 3s^2 3p^6\) → 3 electron shells.
Because the chloride ion has one extra electron shell, its electrons are farther from the nucleus, giving it a larger ionic radius.
Correct Answer: A
Which statement about the properties associated with the different types of bonding involved is correct?
▶️ Answer/Explanation
Ans: C
Ionic compounds conduct electricity only in molten or aqueous states due to free-moving ions, whereas metals conduct in solid state via delocalized electrons. Thus, statement C is correct. Option A is incorrect as not all O-H containing compounds form H-bonds (e.g., \( \text{H}_2\text{O}_2 \)). Option B is false (e.g., \( \text{NH}_4\text{Cl} \) has both bonds). Option D is incorrect since covalent networks (e.g., diamond) have high melting points without H-bonds.
V and Z are both elements in Period 3 of the Periodic Table. Each element forms one stable ion that does not contain another element. The atomic radius of each element and the ionic radius of the ion described above is shown.

Which statement is correct?
A. Ions of V and Z have the same number of full electron shells.
B. Ions of Z are positively charged.
C. Z has a greater electronegativity than V.
D. V has more outer electrons than Z
▶️ Answer/Explanation
Ans: C
The data shows Z has a smaller atomic radius than V, and its ionic radius is larger than its atomic radius. This pattern is characteristic of a non-metal forming an anion (e.g., \( \ce{Cl} \) forming \( \ce{Cl-} \)), while V, whose ionic radius is smaller than its atomic radius, is a metal forming a cation (e.g., \( \ce{Na} \) forming \( \ce{Na+} \)). Electronegativity increases across a period, so Z (a non-metal) has a greater electronegativity than V (a metal).
The boiling points of Br₂, ICl and IBr are given in the table.

Which row explains:
• why the boiling point of ICl is greater than Br₂?
• why the boiling point of IBr is greater than ICl ? 
▶️ Answer/Explanation
Ans: B
ICl has a higher boiling point than Br₂ because ICl is a polar molecule with permanent dipole-dipole forces, whereas Br₂ is non-polar and only has weaker London (dispersion) forces. IBr has a higher boiling point than ICl because IBr has a greater number of electrons (IBr: 198; ICl: 178), resulting in stronger London forces. The difference in electronegativity for both ICl and IBr is similar (approx. 0.5), so the polarity is comparable, making the difference in electron count and the resulting London forces the dominant factor for the second comparison.
Which statement is correct?
A Ammonia reacts with alkalis to form the ammonium ion.
B Ammonium chloride contains ionic, covalent and co-ordinate bonds.
C The ammonium ion reacts with acids to produce ammonia.
D The bond angle in the ammonium ion is approximately 107°.
▶️ Answer / Explanation
A. Ammonia reacts with acids (not alkalis) to form the ammonium ion \(NH_4^+\). ✘
B. In ammonium chloride:
• Ionic bonding exists between \(NH_4^+\) and \(Cl^-\).
• Covalent bonds exist in the N–H bonds.
• One N–H bond forms by donation of a lone pair (co-ordinate bond). ✔
C. Ammonium ions react with alkalis, not acids, to produce ammonia. ✘
D. The ammonium ion is tetrahedral with a bond angle of about 109.5°, not 107°. ✘
Answer: B
Which row describes the structure and bonding of SiO2 and SiCl4?
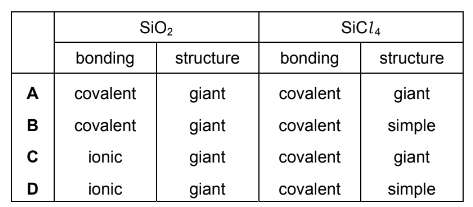
▶️ Answer / Explanation
SiO2: forms a giant covalent (macromolecular) structure. Each silicon atom is covalently bonded to four oxygen atoms in a 3D network.
SiCl4: consists of simple molecular covalent molecules with weak intermolecular forces between molecules.
Therefore the correct description is:
SiO2 → giant covalent structure
SiCl4 → simple molecular covalent structure
Answer: B
The definitions of many chemical terms can be illustrated by chemical equations.
Which terms can be illustrated by an equation that includes the formation of a positive ion?
1 first ionisation energy
2 heterolytic fission of a covalent bond
3 enthalpy change of atomisation
The responses A to D should be selected on the basis of
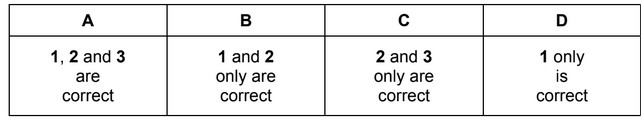
▶️ Answer / Explanation
1. First ionisation energy
\(X(g) \rightarrow X^+(g) + e^-\)
A positive ion is formed. ✔
2. Heterolytic fission
\(A–B \rightarrow A^+ + B^-\) or \(A^- + B^+\)
This process forms ions, including a positive ion. ✔
3. Enthalpy of atomisation
\(X_2(g) \rightarrow 2X(g)\)
This produces neutral atoms, not ions. ✘
Correct statements: 1 and 2
Answer: B
Which molecule contains a nitrogen atom with sp hybridised orbitals?
A CH3CH2NH2 B HNO3 C HCN D NH3
▶️ Answer / Explanation
sp hybridisation occurs when an atom has two electron domains, giving a linear geometry (180°).
A. CH3CH2NH2 → N forms 3 σ bonds + 1 lone pair → sp³. ✘
B. HNO3 → N is trigonal planar → sp². ✘
C. HCN → N is involved in a C≡N triple bond with one lone pair → 2 regions of electron density → sp. ✔
D. NH3 → 3 bonds + 1 lone pair → sp³. ✘
Answer: C
In the sodium chloride lattice the number of chloride ions that surround each sodium ion is called the co-ordination number of the sodium ions.
What are the co-ordination numbers of the sodium ions and the chloride ions in the sodium chloride lattice?
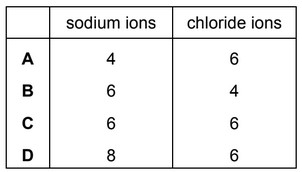
▶️ Answer / Explanation
In the NaCl lattice, each Na⁺ ion is surrounded by 6 Cl⁻ ions in an octahedral arrangement.
Similarly, each Cl⁻ ion is surrounded by 6 Na⁺ ions.
Therefore:
Co-ordination number of Na⁺ = 6
Co-ordination number of Cl⁻ = 6
Answer: C
What is the correct number of bonds of each type in the Al2Cl6 molecule?
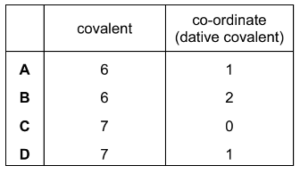
▶️ Answer / Explanation
AlCl3 dimerises to form Al2Cl6.
In this structure:
• Each Al atom forms two normal covalent bonds with terminal Cl atoms.
• Two Cl atoms act as bridging atoms forming dative (coordinate) bonds to aluminium.
Counting the bonds:
Normal covalent bonds = 4
Co-ordinate (dative) bonds = 2
Answer: B
Some car paints contain small flakes of silica, \(SiO_{2}\).
In the structure of solid \(SiO_{2}\)
● each silicon atom is bonded to x oxygen atoms,
● each oxygen atom is bonded to y silicon atoms,
● each bond is a z type bond.
What is the correct combination of x, y and z in these statements?
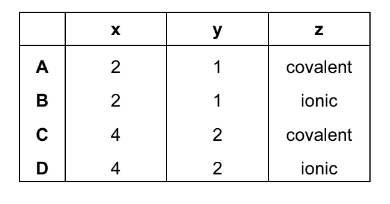
▶️ Answer / Explanation
Solid \(SiO_{2}\) has a giant covalent (network) structure.
• Each Si atom bonds to 4 O atoms in a tetrahedral arrangement.
• Each O atom bonds to 2 Si atoms, linking the network.
• The bonds are covalent.
Therefore:
x = 4
y = 2
z = covalent
Answer: C
The table shows the physical properties of four substances. Which substance has a giant covalent structure?
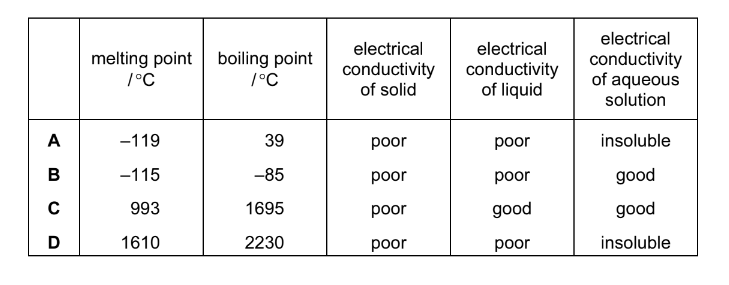
▶️ Answer / Explanation
Giant covalent structures have:
• Very high melting and boiling points
• Poor electrical conductivity
• Usually insoluble in water
A: Low melting/boiling points → simple molecular. ✘
B: Conducts in aqueous solution → ionic substance. ✘
C: Conducts when molten and in solution → ionic lattice. ✘
D: Very high melting/boiling points, poor conductivity, insoluble → typical giant covalent structure. ✔
Answer: D
Which types of bonding are present in ammonium carbonate, \((NH_4)_2CO_3\)?
1 ionic
2 covalent
3 co-ordinate (dative covalent)
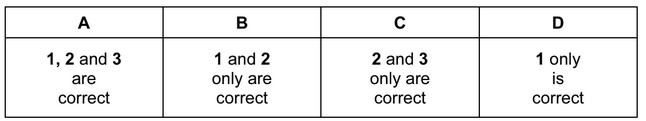
▶️ Answer / Explanation
Ionic bonding: present between \(NH_4^+\) ions and \(CO_3^{2-}\) ions.
Covalent bonding: present inside the polyatomic ions: • N–H bonds in \(NH_4^+\)
• C–O bonds in \(CO_3^{2-}\)
Co-ordinate (dative) bonding: formed when nitrogen donates a lone pair to \(H^+\) to form the \(NH_4^+\) ion.
Therefore all three bonding types are present.
Answer: A
