Pre AP Chemistry -2.3C Ionic Bond Strength and Electrostatic Forces- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.3C Ionic Bond Strength and Electrostatic Forces- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.3C Ionic Bond Strength and Electrostatic Forces- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which statement about the bonding in sodium chloride is correct?
A) The sodium and chlorine atoms share pairs of electrons.
B) The chlorine atoms give electrons to the sodium atoms to form positive and negative ions.
C) There is covalent bonding between sodium and chlorine.
D) The positive and negative ions have noble gas electronic configurations.
▶️ Answer/Explanation
Ans: D
Analysis of sodium chloride (NaCl) bonding:
1. Ionic nature: Sodium donates 1 electron to chlorine, forming Na⁺ and Cl⁻ ions (opposite of option B).
2. Electron configurations:
• Na⁺ achieves neon configuration (2,8)
• Cl⁻ achieves argon configuration (2,8,8)
3. Why other options are wrong:
• A) and C) incorrectly describe covalent/sharing (ionic compounds transfer electrons)
• B) Reverses the electron transfer direction
Which statement is correct?
A Air is a mixture of gaseous elements only.
B Alloys are formed when a metal is ionically bonded to other elements.
C Carbon dioxide is a mixture of carbon and oxygen.
D Potassium bromide is an ionic compound
▶️ Answer/Explanation
Ans: D
1. Analysis of Each Statement:
- A: Incorrect – Air contains compounds (CO₂, H₂O vapor) not just elements (N₂, O₂)
- B: Incorrect – Alloys involve metallic bonding, not ionic bonding
- C: Incorrect – CO₂ is a compound (fixed ratio), not a mixture
- D: Correct – KBr forms when potassium (metal) donates electrons to bromine (non-metal)
2. Ionic Bonding Verification:
- K (Group 1) loses 1 electron → K⁺
- Br (Group 7) gains 1 electron → Br⁻
- Electrostatic attraction between ions forms ionic bond
3. Conclusion:
Only statement D is correct as it accurately describes the ionic nature of potassium bromide.
Chemical Insight: The ionic character of KBr is evidenced by its high melting point (734°C) and ability to conduct electricity when molten – properties typical of ionic compounds.
Some properties of substances W, X, Y and Z are shown.
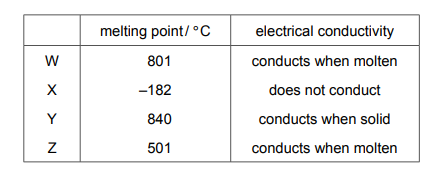
Which substances are ionic?
A) W, X and Y
B) W and Y only
C) W and Z
D) X and Z
▶️ Answer/Explanation
Ans: C
Identifying ionic compounds based on the properties:
- W (High MP, Conducts when molten):
- High melting point and conductivity in molten state are characteristic of ionic compounds.
- X (Low MP, Doesn’t conduct):
- Low melting point and no conductivity suggest a molecular substance.
- Y (High MP, Doesn’t conduct):
- High MP could suggest ionic, but no conductivity even when molten rules this out (likely a covalent network solid).
- Z (High MP, Conducts when solid):
- This suggests a metallic structure, but some ionic compounds can show conductivity in solid state if they have mobile ions.
Conclusion: Only W clearly shows ionic properties, while Z may also be ionic (despite some metallic characteristics). Therefore, C (W and Z) is the most correct answer.
The boiling point of sodium is 890°C. What happens to sodium atoms as the temperature of a sample of sodium changes from 950°C to 900°C?
- The atoms move more quickly and bonds are formed.
- The atoms move more quickly and bonds are neither broken nor formed.
- The atoms move more slowly and bonds are formed.
- The atoms move more slowly and bonds are neither broken nor formed.
▶️ Answer/Explanation
Ans: D
Key analysis of the temperature change:
- Temperature decrease: From 950°C (gas phase) to 900°C (still above boiling point but cooling)
- Atomic motion: As temperature decreases, atoms move more slowly (kinetic energy reduces)
- Bonding status:
- Above boiling point (890°C), sodium exists as individual atoms in gas phase
- No metallic bonds exist in gaseous state
- No new bonds form during this cooling (still above boiling point)
Therefore, the correct description is D – atoms move more slowly as temperature decreases, and no bonds are formed or broken during this phase change above the boiling point.
Some properties of substances W, X, Y and Z are shown.

Which substances are ionic?
A W, X and Y
B W and Y only
C W and Z
D X and Z
▶️ Answer/Explanation
Ans: C
Identifying ionic substances:
- W: High melting point and conducts when molten → ionic (e.g., NaCl)
- Z: Soluble in water and conducts in solution → ionic (e.g., KBr)
- X: Insoluble and doesn’t conduct → likely covalent network (e.g., diamond)
- Y: Low melting point → molecular covalent (e.g., I2)
Key ionic characteristics present in W and Z:
- High melting points (strong ionic bonds)
- Electrical conductivity in molten/dissolved state (mobile ions)
Thus, Option C (W and Z) correctly identifies the ionic substances.
The boiling point of sodium is 890°C. What happens to sodium atoms as the temperature of a sample of sodium changes from 950°C to 900°C?
A) The atoms move more quickly and bonds are formed.
B) The atoms move more quickly and bonds are neither broken nor formed.
C) The atoms move more slowly and bonds are formed.
D) The atoms move more slowly and bonds are neither broken nor formed.
▶️ Answer/Explanation
Ans: D
1. Temperature is decreasing from 950°C (above boiling point) to 900°C (still above boiling point).
2. Since the temperature remains above the boiling point (890°C), sodium remains in gaseous state throughout.
3. As temperature decreases:
– Atoms move more slowly (kinetic energy decreases)
– No bonds form (still in gaseous phase)
– No bonds break (already in atomic gas form)
4. Thus, only option D correctly describes both the slowing motion and absence of bond formation/breaking.
Which row describes the properties of potassium bromide?
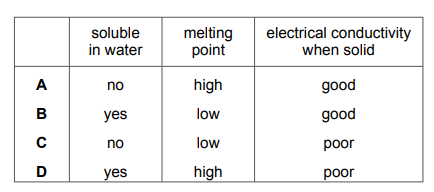
▶️ Answer/Explanation
Ans: D
Analysis of potassium bromide (KBr) properties:
- Electrical Conductivity:
- Solid KBr does not conduct electricity (ions are fixed in position).
- Molten KBr does conduct electricity (ions are free to move).
- Melting Point:
- KBr has a high melting point (~734°C) due to strong ionic bonds.
- Solubility:
- KBr is soluble in water (polar solvent).
Only Row D correctly states:
- Conducts when molten (✔️)
- High melting point (✔️)
- Soluble in water (✔️)
Which statement about the ions formed by the elements in Group VII of the Periodic Table is correct?
- All the ions have the same charge of 1–.
- All the ions have the same number of electron shells.
- Each ion is formed by losing one electron.
- Each ion has seven electrons in its outer electron shell.
▶️ Answer/Explanation
Ans: A
Key characteristics of Group VII (Halogens) ions:
- Option A: Correct. Halogens gain 1 electron to achieve a stable octet, forming 1– ions (e.g., F⁻, Cl⁻, Br⁻).
- Option B: Incorrect. The number of electron shells increases down the group (e.g., F⁻ has 2 shells, I⁻ has 5).
- Option C: Incorrect. Halogens gain electrons to form anions, not lose them.
- Option D: Incorrect. Ions have 8 outer electrons (octet) after gaining 1 electron.
Thus, only A accurately describes Group VII ions.
Cesium fluoride is an ionic compound. Which statements about cesium fluoride are correct?
- 1. It conducts electricity when solid.
- 2. It has a high melting point.
- 3. It is soluble in water.
- 4. It is highly volatile.
A. 1 and 2 B. 1 and 4 C. 2 and 3 D. 3 and 4
▶️ Answer/Explanation
Ans: C
1. Ionic Compound Properties:
– High melting point (Statement 2 correct) due to strong ionic bonds
– Soluble in water (Statement 3 correct) as water molecules can solvate ions
2. Why Other Statements Are Incorrect:
– Statement 1: Only conducts when molten/dissolved (ions must be free to move)
– Statement 4: Ionic compounds are non-volatile (strong bonds prevent evaporation)
3. Key Characteristics:
– Melting point: ~682°C
– Water solubility: 367 g/100 mL (20°C)
– Electrical conductivity: Only in aqueous/molten state
Therefore, only statements 2 and 3 are correct (Option C).
Which row describes the properties of potassium iodide, KI?

▶️ Answer/Explanation
Ans: D
Analysis of potassium iodide (KI) properties:
1. Physical state: Ionic compounds like KI are solid at room temperature with high melting/boiling points.
2. Electrical conductivity:
• Solid state: No conductivity (ions fixed in lattice)
• Molten/dissolved: Conducts (mobile ions)
3. Structure: Giant ionic lattice (not simple molecules)
Therefore, only option D correctly describes all these properties of KI.
Which row describes how an ionic bond forms between a sodium atom and a chlorine atom?

▶️ Answer/Explanation
Ans: D
1. Electron Configuration Analysis:
- Sodium (Na): 1s² 2s² 2p⁶ 3s¹ → 1 valence electron
- Chlorine (Cl): 1s² 2s² 2p⁶ 3s² 3p⁵ → 7 valence electrons
2. Ionic Bond Formation Process:
- Na donates its 3s¹ electron to achieve stable Ne configuration (2,8)
- Cl accepts 1 electron to achieve stable Ar configuration (2,8,8)
- Resulting ions: Na⁺ (cation) and Cl⁻ (anion)
3. Option Verification:
- Only Row D correctly shows:
- Electron transfer from Na to Cl
- Formation of oppositely charged ions
- Electrostatic attraction between ions
Chemical Insight: The ionic bond in NaCl results in a crystal lattice structure with high melting point (801°C) and solubility in water – characteristic properties of ionic compounds.
4. Conclusion:
The complete electron transfer process described in Option D accurately represents ionic bond formation between sodium and chlorine.
Lithium and fluorine react to form lithium fluoride.
A student writes three statements about the reaction.
1 Lithium atoms lose an electron when they react.
2 Each fluoride ion has one more electron than a fluorine atom.
3 Lithium fluoride is a mixture of elements.
Which statements are correct?
A 1 and 2 only B 1 and 3 only C 2 and 3 only D 1, 2 and 3
▶️ Answer/Explanation
Ans: A
Analysis of each statement:
- Statement 1: Correct
Lithium (Li) loses its single valence electron to form Li⁺ ion: Li → Li⁺ + e⁻ - Statement 2: Correct
Fluorine (F) gains one electron to form F⁻ ion: F + e⁻ → F⁻ - Statement 3: Incorrect
Lithium fluoride (LiF) is an ionic compound, not a mixture of elements
Key points about the reaction:
– Li (metal) donates electron to F (non-metal)
– Forms ionic bond through electron transfer
– Results in crystalline ionic compound (not mixture)
Therefore, only statements 1 and 2 are correct.
The element livermorium, Lv, was discovered in the year 2000.
Which statement predicts what will happen to an Lv atom when it forms an \(Lv^{2–}\) ion?
- The atom will gain two electrons.
- The atom will lose two electrons.
- The atom will lose two protons.
- The atom will gain two protons.
▶️ Answer/Explanation
Ans: A
Understanding ion formation:
- Negative ions: Form when atoms gain electrons (not protons)
- \(Lv^{2–}\): The 2– charge indicates the atom gained 2 electrons
- Key concepts:
- Proton number never changes in normal chemical reactions
- Electron transfer (not proton) creates ions
- Negative charge = electron gain
Why other options are incorrect:
- B: Would create \(Lv^{2+}\) (positive ion)
- C/D: Proton number changes would alter the element’s identity (nuclear reaction)
Thus, the correct answer is A – livermorium gains two electrons to form the \(Lv^{2–}\) ion.
Lithium reacts with fluorine to form the compound lithium fluoride.
Which statement about this reaction is correct?
A Each fluorine atom gains one electron.
B Each fluorine atom gains two or more electrons.
C Each fluorine atom loses one electron.
D Each fluorine atom loses two or more electrons.
▶️ Answer/Explanation
Ans: A
This ionic bond formation involves:
- Lithium (Group 1) donates its single valence electron to achieve stability
- Fluorine (Group 17) gains one electron to complete its outer shell (octet)
Why other options are incorrect:
- B: Fluorine only needs one electron to complete its valence shell
- C/D: Fluorine is highly electronegative and gains (not loses) electrons
The resulting compound LiF consists of Li+ cations and F– anions held together by strong electrostatic forces.
Calcium reacts with chlorine to produce calcium chloride. What happens when a calcium ion forms during this reaction?
A) The calcium atom gains one electron.
B) The calcium atom gains two electrons.
C) The calcium atom loses one electron.
D) The calcium atom loses two electrons.
▶️ Answer/Explanation
Ans: D
1. Calcium is in Group 2 of the periodic table with 2 valence electrons.
2. To achieve stability, calcium loses 2 electrons to form Ca2+ ion.
3. Each chlorine atom (Group 17) gains 1 electron, so two chlorine atoms are needed per calcium atom.
4. The resulting compound is CaCl2 (calcium chloride).
5. Electron transfer:
Ca → Ca2+ + 2e–
2Cl + 2e– → 2Cl–
Thus, calcium loses two electrons during ion formation.
Ions are formed by elements losing or gaining electrons.
Which statement is correct?
- Metal atoms gain electrons to form positive ions.
- Non-metal atoms lose electrons to form positive ions.
- The charge on an ion is always either +1 or -1.
- Group I ions have the same electronic structure as noble gases.
▶️ Answer/Explanation
Ans: D
Analysis of each statement:
- Statement A:
- Metal atoms lose electrons to form positive ions (cations).
- This statement is incorrect because it reverses the process.
- Statement B:
- Non-metal atoms gain electrons to form negative ions (anions).
- This statement is incorrect for the same reason as A.
- Statement C:
- Ions can have various charges (e.g., Fe²⁺, Fe³⁺, SO₄²⁻).
- This statement is too restrictive and therefore incorrect.
- Statement D:
- Group I elements (alkali metals) lose 1 electron to form +1 ions.
- This leaves them with a full outer shell, identical to noble gas configuration.
- This is the only correct statement.
Example: Sodium (Na) loses 1 electron → Na⁺ (same electron configuration as neon).
Sodium reacts with chlorine to form sodium chloride.
Which statements describe what happens to the sodium atoms in this reaction?
- 1. Sodium atoms form positive ions.
- 2. Sodium atoms form negative ions.
- 3. Sodium atoms gain electrons.
- 4. Sodium atoms lose electrons.
A 1 and 3 B 1 and 4 C 2 and 3 D 2 and 4
▶️ Answer/Explanation
Ans: B
1. Reaction Mechanism:
– Sodium (Na) loses 1 valence electron to form Na+ cation (Statement 1 and 4 correct)
– Chlorine (Cl) gains this electron to form Cl– anion
2. Why Other Options Are Incorrect:
– Sodium cannot form negative ions (Statement 2 wrong)
– Sodium loses electrons (Statement 3 wrong)
3. Key Concepts:
– Alkali metals (Group 1) always lose electrons to form +1 ions
– The electron transfer creates strong ionic bonds in NaCl
Therefore, only statements 1 and 4 are correct (Option B).
Which statement describes positive ions?
A) Positive ions have more electrons than neutrons.
B) Positive ions have more protons than neutrons.
C) Positive ions have more electrons than protons.
D) Positive ions have more protons than electrons.
▶️ Answer/Explanation
Ans: D
1. Positive Ion Formation: – Formed when atoms lose electrons – Results in fewer electrons than protons
2. Charge Imbalance: – Protons (positive) remain constant – Electron loss creates net positive charge
3. Why D is Correct: – Only statement showing proton-electron relationship correctly – “More protons than electrons” defines positive ions
4. Other Options Analysis: – A: Irrelevant – compares electrons to neutrons – B: Incorrect – proton-neutron ratio varies by isotope – C: Wrong – describes negative ions
What happens when a bond is formed between a green gaseous element and a soft metallic element?
A) The gas atoms gain an electron.
B) The gas atoms lose an electron.
C) The metal atoms gain an electron.
D) The two elements share a pair of electrons.
▶️ Answer/Explanation
Ans: A
Analysis of the bond formation:
1. Element Identification:
• Green gas = chlorine (Cl₂)
• Soft metal = sodium/potassium (Na/K)
2. Bonding Process:
• Metal atoms lose valence electron(s) → form cations
• Chlorine atoms gain electron(s) → form anions
3. Resulting Compound:
• Ionic bond forms (e.g., NaCl or KCl)
• Electrons transfer completely (not shared)
Element X is in Group I of the Periodic Table. X reacts with element Y to form an ionic compound.
Which equation shows the process that takes place when X forms ions?
- X + e– → X+
- X – e– → X–
- X + e– → X–
- X – e– → X+
▶️ Answer/Explanation
Ans: D
For Group I elements:
- They have 1 valence electron in their outer shell
- They lose this electron to form +1 cations
- The process is: X → X+ + e– (electron loss)
Analysis of options:
A: Incorrect (shows electron gain to form positive ion – impossible)
B: Incorrect (shows electron loss to form negative ion – impossible)
C: Incorrect (shows electron gain to form negative ion – this would happen with non-metals)
D: Correct (shows electron loss to form positive ion)
Key point: Group I metals always lose electrons to form positive ions in ionic bonding.
Sodium chloride is an ionic solid.
Which statement is not correct?
- Ions are formed when atoms lose or gain electrons.
- Ions in sodium chloride are strongly held together.
- Ions with the same charge attract each other.
- Sodium chloride solution can conduct electricity.
▶️ Answer/Explanation
Ans: C
Analysis of each statement:
- A: Correct – ions form through electron transfer (Na loses, Cl gains)
- B: Correct – strong electrostatic forces hold Na⁺ and Cl⁻ together
- C: Incorrect – like charges repel, opposite charges attract
- D: Correct – free-moving ions in solution conduct electricity
Key concepts about ionic compounds:
- Ionic bonding occurs between oppositely charged ions
- Electrostatic attraction follows Coulomb’s law: F ∝ (q₁q₂)/r²
- In solid NaCl, ions are fixed in lattice; in solution they’re mobile
Thus, C is the incorrect statement as ions with the same charge would actually repel each other.
Cesium chloride and rubidium bromide are halide compounds of Group I elements.
Cesium chloride has the formula ___ 1 ___ , a relative formula mass ___ 2 ___ that of rubidium bromide and bonds that are ___ 3 ___ .
Which words correctly complete gaps 1, 2 and 3?
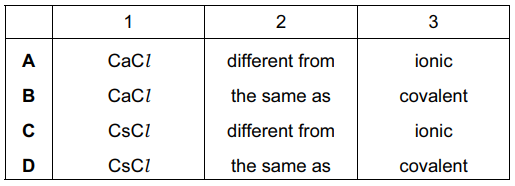
▶️ Answer/Explanation
Ans: C
Complete analysis of the gaps:
- Formula (Gap 1): CsCl (1:1 ratio of Cs+ and Cl– ions)
- Relative mass (Gap 2):
- CsCl: 132.9 (Cs) + 35.5 (Cl) = 168.4
- RbBr: 85.5 (Rb) + 79.9 (Br) = 165.4
- 168.4 > 165.4 → “greater than”
- Bond type (Gap 3): Both form ionic bonds (metal + non-metal electron transfer)
Key points about Group I halides:
- Always form 1:1 ionic compounds (MX)
- Bond strength increases with decreasing ion size
- Mass relationship depends on specific element combination
Rubidium is in Group I of the Periodic Table and bromine is in Group VII.
Rubidium reacts with bromine to form an ionic compound.
Which row shows the electron change taking place for rubidium and the correct formula of the rubidium ion?

▶️ Answer/Explanation
Ans: C
Key points about the reaction:
- Rubidium (Group I):
- Has 1 valence electron
- Loses 1 electron to form Rb+ ion
- Achieves stable noble gas configuration (Kr)
- Bromine (Group VII):
- Has 7 valence electrons
- Gains 1 electron to form Br– ion
- Achieves stable noble gas configuration (Kr)
- Ionic Compound Formation:
- Rb donates 1 electron to Br
- Forms RbBr with 1:1 ratio
Thus, Row C correctly shows:
– Electron change: loses 1 electron
– Ion formula: Rb+
The table shows the electronic structures of four atoms.
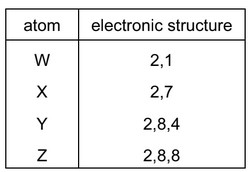
Which two atoms combine to form an ionic compound?
A. W and X B. W and Y C. X and Y D. X and Z
▶️ Answer/Explanation
Ans: A
1. Electronic Analysis:
– W: 2,8,1 configuration (alkali metal – likely Na)
– X: 2,8,7 configuration (halogen – likely Cl)
– Y: 2,8,4 configuration (Group 14 – nonmetal)
– Z: 2,8,8 configuration (noble gas – unreactive)
2. Ionic Bond Formation:
– W can donate its 1 valence electron to achieve stability
– X can accept 1 electron to complete its outer shell
– This electron transfer forms W+ and X– ions
3. Why Other Options Fail:
– W+Y: No complete electron transfer (Y needs 4 electrons)
– X+Y: Both nonmetals would form covalent bonds
– X+Z: Noble gases don’t form compounds
The only ionic pair is W and X (Option A), forming a compound like NaCl.
