Pre AP Chemistry -2.3D Representations of Ionic and Covalent Compounds- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -2.3D Representations of Ionic and Covalent Compounds- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -2.3D Representations of Ionic and Covalent Compounds- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
The electronic structures of two atoms, $\mathrm{P}$ and $\mathrm{Q}$, are shown.
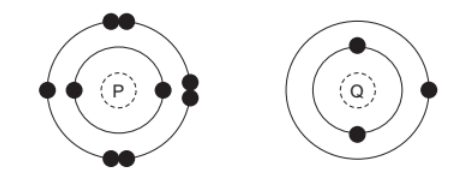
P and Q combine together to form a compound.
What is the type of bonding in the compound and what is the formula of the compound?

▶️ Answer/Explanation
Ans: A
1. Electron Analysis: – P: 7 valence electrons (needs 1 to complete octet) – Q: 1 valence electron (can donate 1 electron)
2. Bond Formation: – Q donates its valence electron to P – Forms $\mathrm{Q}^+$ cation and $\mathrm{P}^-$ anion – Ionic bond results from this electron transfer
3. Compound Formula: – Charges balance directly (1:1 ratio) – Simplest formula is PQ
4. Why Other Options are Incorrect: – B: Wrong formula (P2Q doesn’t balance charges) – C: Covalent bonding doesn’t apply here – D: Wrong bonding type and formula
Which row describes the type of bonding present in substances 1 and 2?
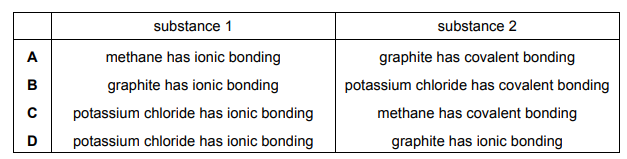
▶️ Answer/Explanation
Ans: C
Analysis of bonding types:
- Substance 1 (Methane, CH4):
- Covalent bonding between carbon and hydrogen atoms (electron sharing).
- Typical of molecular compounds.
- Substance 2 (Graphite):
- Covalent bonding within each carbon layer (strong C-C bonds).
- Delocalized electrons between layers give conductivity.
- Potassium Chloride (KCl):
- Ionic bonding (electron transfer from K to Cl).
- Not relevant to this question but shown for comparison.
Thus, Row C correctly identifies both substances as having covalent bonding.
Which diagram correctly shows the ions present in the compound potassium fluoride?
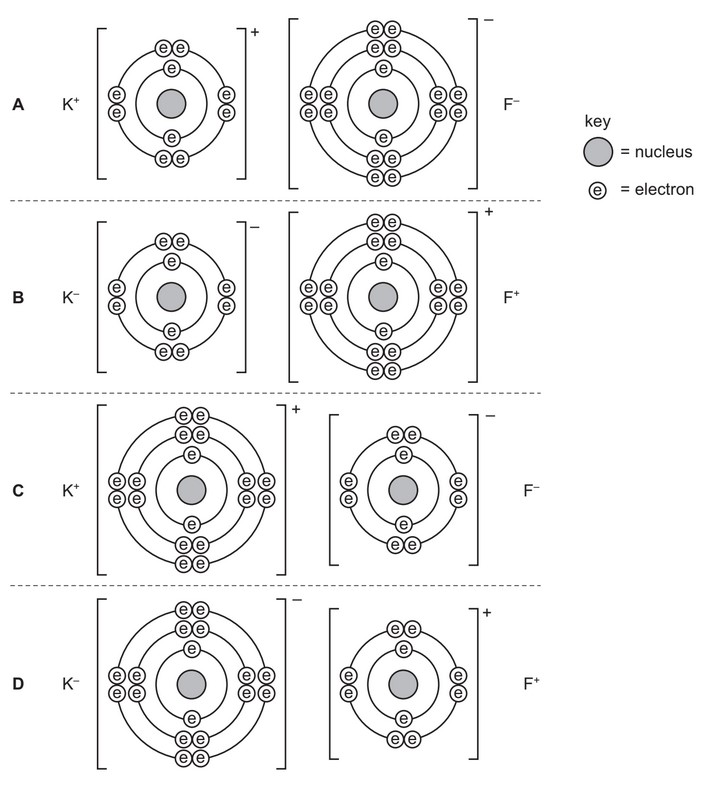
▶️ Answer/Explanation
Ans: C
1. Ion Formation Process:
- Potassium (K) donates its 4s¹ electron → K⁺ ion (loses 1 electron)
- Fluorine (F) gains 1 electron → F⁻ ion (completes its octet)
2. Diagram Analysis:
- Option C correctly shows:
- K⁺ with 18 electrons (original 19 – 1 donated)
- F⁻ with 10 electrons (original 9 + 1 gained)
- Proper charge distribution (+1 and -1)
3. Why Other Options Are Incorrect:
- A: Shows incorrect electron counts (neutral atoms)
- B: Shows incorrect charges (reversed polarity)
- D: Shows incorrect electron transfer (multiple electrons)
Chemical Insight: The 1:1 ratio of K⁺ to F⁻ ions in potassium fluoride forms a cubic crystal lattice structure, similar to NaCl, with strong electrostatic attractions giving it a high melting point (858°C).
4. Conclusion:
Diagram C accurately represents the ionic bonding in KF, showing the correct electron transfer and resulting ion charges.
Element X has six electrons in its outer shell.
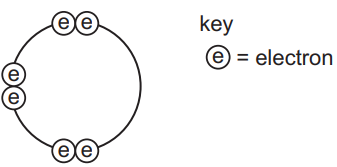
How could the element react?
- by gaining two electrons to form a positive ion
- by losing six electrons to form a negative ion
- by sharing two electrons with two electrons from another element to form two covalent bonds
- by sharing two electrons with two electrons from another element to form four covalent bonds
▶️ Answer/Explanation
Ans: C
Analysis of element X with 6 valence electrons (Group 16 element like oxygen):
- Option A:
- Gaining electrons would form a negative ion, not positive.
- Incorrect charge direction.
- Option B:
- Losing 6 electrons is energetically unfavorable.
- Would form a positive ion, not negative.
- Double incorrect.
- Option C:
- Forms two covalent bonds by sharing its 2 unpaired electrons.
- Example: H₂O (oxygen shares 2 electrons with 2 hydrogen atoms).
- Correct and most likely behavior.
- Option D:
- Would require promoting electrons to form 4 bonds (unlikely for Group 16).
- Typical maximum is 2 covalent bonds for these elements.
Key concept: Group 16 elements typically complete their octet by forming two covalent bonds (e.g., in H₂O or CO₂).
The electronic structures of atoms P and Q are shown.
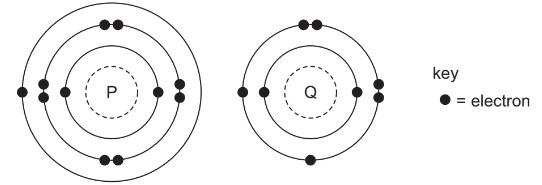
P and Q react to form an ionic compound.
What is the formula of this compound?
A. PQ2 B. P2Q C. P2Q6 D. P6Q2
▶️ Answer/Explanation
Ans: B
1. Valence Electron Analysis:
– P: 2,8,1 configuration (Group 1 metal – e.g., Na)
– Q: 2,8,6 configuration (Group 16 nonmetal – e.g., O)
2. Ion Formation:
– Each P atom loses 1 electron to form P+
– Each Q atom gains 2 electrons to form Q2-
3. Compound Formation:
– Charge balance requires 2 P+ ions for every 1 Q2- ion
– Results in the formula P2Q (e.g., Na2O)
4. Why Other Options Are Incorrect:
– PQ2: Would create charge imbalance (+1 vs -4)
– P2Q6/P6Q2: Don’t satisfy octet rule or charge balance
The correct ionic formula is P2Q (Option B).
A model of a molecule is shown.

Which row shows the formula of this molecule and describes the type of bonding between the atoms?

A 2BH3 / covalent
B 2BH3 / ionic
C B2H6 / covalent
D B2H6 / ionic
▶️ Answer / Explanation
The model represents diborane, \(B_2H_6\).
In this molecule:
• Two boron atoms are bonded to hydrogen atoms.
• Two hydrogen atoms form bridging bonds between the boron atoms.
• All bonds are covalent (electron sharing).
Therefore the molecule is \(B_2H_6\) and the bonding is covalent.
Answer: C
Which dot-and-cross diagram represents a molecule of ammonia?

▶️ Answer / Explanation
Ammonia (\(NH_3\)) has:
• Three N–H covalent bonds.
• One lone pair of electrons on the nitrogen atom.
• Each bond contains one electron from nitrogen and one from hydrogen.
Only diagram C correctly shows:
• Three shared electron pairs between N and H.
• One lone pair on nitrogen.
Answer: C
What is the dot-and-cross diagram for a water molecule?

▶️ Answer / Explanation
In a water molecule (\(H_2O\)):
• Oxygen has 6 valence electrons.
• Each hydrogen has 1 electron.
• Two O–H covalent bonds form by sharing one electron from H and one from O.
After bonding, oxygen still has two lone pairs of electrons.
The correct diagram must therefore show:
• Two shared electron pairs (O–H bonds).
• Two lone pairs on the oxygen atom.
Diagram A shows this correctly.
Answer: A
In which molecule are all the outer electrons of the atoms used in covalent bonds?
A. CH4
B. HCl
C. H2O
D. NH3
▶️ Answer / Explanation
CH4: Carbon has 4 valence electrons and forms four C–H bonds, using all its outer electrons in bonding. ✔
HCl: Chlorine has three lone pairs of electrons remaining. ✘
H2O: Oxygen has two lone pairs remaining. ✘
NH3: Nitrogen has one lone pair remaining. ✘
Only methane has no lone pairs on the central atom, meaning all outer electrons are used in bonding.
Answer: A
The diagram represents an ionic compound formed from three types of atom.

What is the chemical formula for this compound?
(A) \(Na_{2}S_{4}O\)
(B) \(NaO_{4}S_{2}\)
(C) \(Na_{2}SO_{4}\)
(D) \(S_{4}O_{2}Na\)
▶️ Answer / Explanation
The diagram shows:
• One sulfur atom at the center.
• Four oxygen atoms bonded around sulfur, forming a sulfate ion \(SO_4^{2-}\).
• Two sodium ions balancing the \(2-\) charge of the sulfate ion.
Therefore the compound contains:
2 sodium atoms
1 sulfur atom
4 oxygen atoms
This gives the formula:
\(Na_2SO_4\)
Answer: C
