Pre AP Chemistry -3.1B Ideal Gas Law and Gas Particles- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -Link- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -Link- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
\(10 \, cm^{3}\) of ethane is burned in \(45 \, cm^{3}\) of oxygen at a pressure of 101 kPa and a temperature of 200 °C. Complete combustion takes place.
What is the total volume of gas present when the reaction is complete, measured under the same conditions?
A \(30 \, cm^{3}\) B \(50 \, cm^{3}\) C \(55 \, cm^{3}\) D \(60 \, cm^{3}\)
▶️ Answer/Explanation
Step 1: Write the balanced combustion equation
\(2C_{2}H_{6} + 7O_{2} \rightarrow 4CO_{2} + 6H_{2}O\)
Gas volumes are proportional to mole ratios under the same conditions.
Step 2: Determine oxygen required
From the equation:
\(2 \, C_{2}H_{6} : 7 \, O_{2}\)
\(1 \, C_{2}H_{6}\) requires \(3.5 \, O_{2}\)
For \(10 \, cm^{3}\) ethane:
Oxygen required = \(10 \times 3.5 = 35 \, cm^{3}\)
Step 3: Determine excess oxygen
Initial oxygen = \(45 \, cm^{3}\)
Oxygen remaining = \(45 – 35 = 10 \, cm^{3}\)
Step 4: Calculate products formed
From the equation:
\(2C_{2}H_{6} \rightarrow 4CO_{2} + 6H_{2}O\)
1 volume ethane produces:
\(2 \, CO_{2} + 3 \, H_{2}O\)
For \(10 \, cm^{3}\) ethane:
CO₂ = \(20 \, cm^{3}\)
H₂O = \(30 \, cm^{3}\)
At \(200^\circ C\), water remains as steam, so it counts as gas.
Step 5: Total final gas volume
Total gas = CO₂ + H₂O + remaining O₂
\(= 20 + 30 + 10\)
\(= 60 \, cm^{3}\)
Ans: D
1.8 g of water, heated to 227 °C in a sealed container, turns to steam with a pressure of 200 kPa.
What is the approximate volume of the container?
A \(9 \times 10^{-4} \, m^{3}\) B \(2 \times 10^{3} \, m^{3}\) C \(2 \, m^{3}\) D \(8 \times 10^{7} \, m^{3}\)
▶️ Answer/Explanation
Step 1: Convert mass to moles
Molar mass of water \(= 18 \, g\,mol^{-1}\)
\( n = \frac{1.8}{18} = 0.10 \, mol \)
Step 2: Convert temperature to Kelvin
\( T = 227 + 273 = 500 \, K \)
Step 3: Use the ideal gas equation
\( PV = nRT \)
\(P = 200 \, kPa = 2.0 \times 10^{5} \, Pa\)
\(R = 8.31 \, J\,mol^{-1}K^{-1}\)
\( V = \frac{nRT}{P} \)
\( V = \frac{(0.10)(8.31)(500)}{2.0 \times 10^{5}} \)
\( V = \frac{415.5}{2.0 \times 10^{5}} \)
\( V \approx 2.1 \times 10^{-3} \, m^{3} \)
The closest approximate value among the options is:
Ans: C
A fluorescent light tube has an internal volume of 400 \(cm^3\) and an internal pressure of 200 kPa. It is filled with 0.03 moles of an ideal gas.
What is the temperature of the gas inside the fluorescent light tube?
A \(3.21 \times 10^{-1}\) K
B \(3.21 \times 10^{2}\) K
C \(3.21 \times 10^{5}\) K
D \(3.21 \times 10^{8}\) K
▶️ Answer/Explanation
Step 1: Convert volume to \(m^3\)
\(400 \, cm^3 = 400 \times 10^{-6} \, m^3\)
\(V = 4.00 \times 10^{-4} \, m^3\)
Step 2: Use the ideal gas equation
\(pV = nRT\)
\(T = \dfrac{pV}{nR}\)
Step 3: Substitute values
\(p = 200 \, kPa = 2.00 \times 10^5 \, Pa\)
\(V = 4.00 \times 10^{-4} \, m^3\)
\(n = 0.03 \, mol\)
\(R = 8.31 \, J\,mol^{-1}K^{-1}\)
\( T = \dfrac{(2.00 \times 10^5)(4.00 \times 10^{-4})}{(0.03)(8.31)} \)
\( T = \dfrac{80}{0.249} \)
\( T \approx 3.21 \times 10^{2} \, K \)
Ans: B
The gas laws can be summarised in the ideal gas equation.
\(pV = nRT\)
0.960 g of oxygen gas is contained in a vessel of volume \(7.00 \times 10^{-3} \, m^{3}\) at a temperature of 30 °C.
Assume that the gas behaves as an ideal gas.
What is the pressure in the vessel?
A 1.07 kPa B 2.14 kPa C 10.8 kPa D 21.6 kPa
▶️ Answer/Explanation
Step 1: Calculate number of moles of oxygen
Molar mass of \(O_{2}\) = \(32 \, g\,mol^{-1}\)
\( n = \frac{0.960}{32} = 0.030 \, mol \)
Step 2: Convert temperature to Kelvin
\( T = 30 + 273 = 303 \, K \)
Step 3: Use the ideal gas equation
\( pV = nRT \)
\( p = \frac{nRT}{V} \)
\(R = 8.31 \, J\,mol^{-1}K^{-1}\)
\( p = \frac{(0.030)(8.31)(303)}{7.00 \times 10^{-3}} \)
\( p = \frac{75.5}{0.007} \)
\( p \approx 1.08 \times 10^{4} \, Pa \)
Convert to kPa:
\( p \approx 10.8 \, kPa \)
Ans: C
Use of the Data Booklet is relevant to this question. The gas laws can be summarised in the ideal gas equation below.
\(pV = nRT\)
0.96 g of oxygen gas is contained in a glass vessel of volume \(7.0 \times 10^{-3} m^{3}\) at a temperature of 30°C. Assume the gas behaves as an ideal gas.
What is the pressure in the vessel?
A 1.1 kPa B 2.1 kPa C 10.8 kPa D 21.6 kPa
▶️ Answer/Explanation
Step 1: Calculate number of moles
Molar mass of \(O_2 = 32 \, g\,mol^{-1}\)
\(n = \dfrac{0.96}{32} = 0.03 \, mol\)
Step 2: Convert temperature to Kelvin
\(T = 30 + 273 = 303 \, K\)
Step 3: Use ideal gas equation
\(pV = nRT\)
\(p = \dfrac{nRT}{V}\)
\(R = 8.31 \, J\,mol^{-1}K^{-1}\)
\( p = \dfrac{(0.03)(8.31)(303)}{7.0 \times 10^{-3}} \)
\( p = \dfrac{75.5}{0.007} \)
\( p \approx 1.08 \times 10^{4} \, Pa \)
Step 4: Convert to kPa
\(1.08 \times 10^{4} \, Pa = 10.8 \, kPa\)
Ans: C
The gas laws can be summarised in the ideal gas equation.
\(pV = nRT\)
where each symbol has its usual meaning.
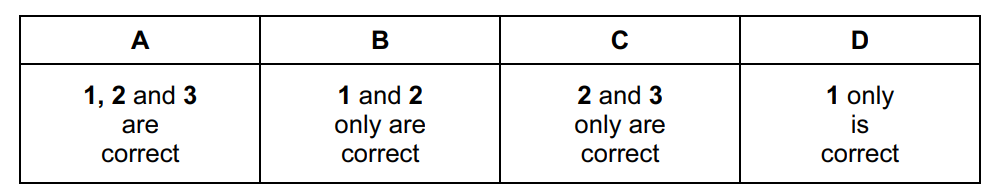
Which statements are correct?
- One mole of an ideal gas occupies the same volume under the same conditions of temperature and pressure.
- The density of an ideal gas at constant pressure is inversely proportional to the temperature, \(T\).
- The volume of a given mass of an ideal gas is doubled if its temperature is raised from 25 °C to 50 °C at constant pressure.
▶️ Answer/Explanation
Step 1: Evaluate statement 1
From the ideal gas equation \(pV = nRT\).
For the same temperature and pressure:
\(V = \dfrac{nRT}{p}\)
If \(n = 1\) mole, the volume will always be the same under identical conditions.
Statement 1 → Correct
Step 2: Evaluate statement 2
Density \(d = \dfrac{mass}{volume}\).
At constant pressure:
\(V \propto T\)
Therefore:
\(d \propto \dfrac{1}{T}\)
Statement 2 → Correct
Step 3: Evaluate statement 3
Temperature must be converted to Kelvin.
25°C = 298 K
50°C = 323 K
The temperature does not double, so the volume does not double.
Statement 3 → Incorrect
Correct statements: 1 and 2
Ans: B
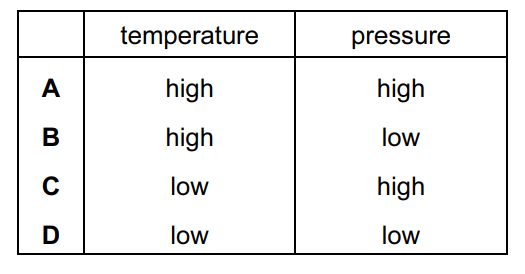
Under which set of conditions is a gas most likely to behave ideally?
▶️ Answer/Explanation
Ideal gas behaviour occurs when intermolecular forces and molecular volumes become negligible.
This happens when:
- Temperature is high → molecules move faster and intermolecular attractions become less significant.
- Pressure is low → molecules are far apart, so their own volume and intermolecular forces become negligible.
Therefore, the gas behaves most ideally under conditions of:
High temperature and low pressure.
From the table of options, this corresponds to Option B.
Ans: B
Use of the Data Booklet is relevant to this question.
The gas laws can be summarised in the ideal gas equation.
\(pV = nRT\)
0.56 g of ethene gas is contained in a vessel at a pressure of 102 kPa and a temperature of 30 °C.
What is the volume of the vessel?
A 49 cm3 B 494 cm3 C 48 900 cm3 D 494 000 cm3
▶️ Answer/Explanation
Step 1: Calculate number of moles of ethene
Molar mass of ethene \(C_2H_4 = 28 \, g\,mol^{-1}\)
\(n = \dfrac{0.56}{28} = 0.020 \, mol\)
Step 2: Convert temperature to Kelvin
\(T = 30 + 273 = 303 \, K\)
Step 3: Rearrange the ideal gas equation
\(pV = nRT\)
\(V = \dfrac{nRT}{p}\)
\(R = 8.31 \, J\,mol^{-1}K^{-1}\)
\(p = 102 \, kPa = 1.02 \times 10^{5} \, Pa\)
\( V = \dfrac{(0.020)(8.31)(303)}{1.02 \times 10^{5}} \)
\( V = \dfrac{50.3}{1.02 \times 10^{5}} \)
\( V \approx 4.93 \times 10^{-4} \, m^{3} \)
Step 4: Convert to \(cm^3\)
\(1 \, m^{3} = 10^{6} \, cm^{3}\)
\( V = 4.93 \times 10^{-4} \times 10^{6} \)
\( V \approx 494 \, cm^{3} \)
Ans: B
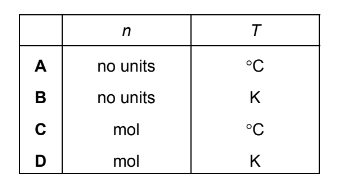
In the ideal gas equation, \(pV = nRT\), what are the units of \(n\) and \(T\)?
▶️ Answer/Explanation
Step 1: Recall the ideal gas equation
\(pV = nRT\)
Where:
- \(p\) = pressure
- \(V\) = volume
- \(n\) = number of moles
- \(R\) = gas constant
- \(T\) = absolute temperature
Step 2: Units required for the equation
- \(n\) must be measured in moles (mol).
- \(T\) must be measured in Kelvin (K), not °C.
This corresponds to option D.
Ans: D
