Pre AP Chemistry -4.1A Precipitation Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.1A Precipitation Reactions- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.1A Precipitation Reactions- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
A solution of compound Z gives a light blue precipitate with aqueous ammonia. The precipitate dissolves in an excess of ammonia.
A flame test is done on compound Z.
What is the colour of the flame?
- blue-green
- lilac
- red
- yellow
▶️ Answer/Explanation
Ans: A (blue-green)
The observations indicate that compound Z contains copper (Cu²⁺) ions:
- Light blue precipitate with ammonia: This is copper(II) hydroxide (\(Cu(OH)_2\)), which forms when aqueous ammonia reacts with Cu²⁺ ions.
- Precipitate dissolves in excess ammonia: \(Cu(OH)_2\) reacts further to form the deep blue complex ion \([Cu(NH_3)_4]^{2+}\).
- Flame test: Copper compounds produce a characteristic blue-green flame.
Other options can be eliminated because:
- Lilac (B) is for potassium (K⁺).
- Red (C) is for lithium (Li⁺) or strontium (Sr²⁺).
- Yellow (D) is for sodium (Na⁺).
Which rows identify two aqueous salts which react together to produce a precipitate?

A) 1 and 4
B) 1 and 3
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: B
1. Precipitation Reaction Analysis:
• Option 1 (BaCl2) + Option 3 (Na2SO4):
Ba2+ + SO42- → BaSO4↓ (white precipitate)
• Solubility rules: All sodium and chloride salts are soluble, but BaSO4 is insoluble 2. Why Other Combinations Fail:
• 1+4: Forms soluble NaCl and KNO3
• 2+3: Forms soluble Na2CO3 and KCl
• 2+4: Forms soluble K2CO3 and NaCl
3. Key Chemical Principle:
Precipitation occurs when product solubility is < 0.01 mol/L (BaSO4 Ksp = 1.1×10-10)
Compound X is dissolved in water and two separate samples of the solution are tested. The results of the tests are shown.
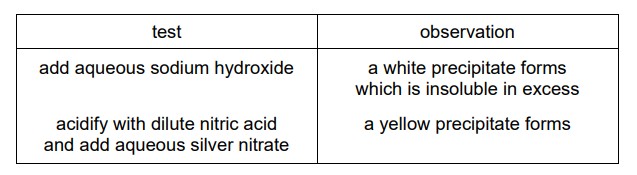
What is compound X?
A calcium chloride
B calcium iodide
C zinc chloride
D zinc iodide
▶️ Answer/Explanation
Ans: B
1. Test 1 Analysis (NaOH):
White precipitate indicates Ca2+ (forms Ca(OH)2) or Zn2+ (forms Zn(OH)2).
2. Test 2 Analysis (HNO3 + AgNO3):
Yellow precipitate confirms I– ions (forms AgI).
3. Compound Identification:
– A: Would give white AgCl precipitate (not yellow)
– B: Matches both tests (Ca2+ + I–)
– C: Would give white AgCl precipitate
– D: Would give white Zn(OH)2 but we need Ca2+ confirmation
Only calcium iodide explains both test results.
Which two compounds give a white precipitate when their aqueous solutions are mixed?
A) Silver nitrate and sodium chloride
B) Silver nitrate and sodium iodide
C) Sodium hydroxide and copper(II) sulfate
D) Sodium hydroxide and iron(II) chloride
▶️ Answer/Explanation
Ans: A
1. Precipitation Reaction:
AgNO3(aq) + NaCl(aq) → AgCl↓(white) + NaNO3(aq)
• Silver chloride (AgCl) has very low solubility (Ksp = 1.8×10-10)
• Forms immediately as a curdy white precipitate 2. Why Other Options Are Incorrect:
• B: Forms yellow AgI precipitate
• C: Forms blue Cu(OH)2 precipitate
• D: Forms green Fe(OH)2 precipitate
3. Key Characteristics:
• AgCl darkens in sunlight (photochemical decomposition)
• Used in photographic films and chloride ion tests
• The only white precipitate among the options
The results of two tests on solid X are shown.

What is X?
A) copper(II) chloride
B) copper(II) iodide
C) iron(II) chloride
D) iron(II) iodide
▶️ Answer/Explanation
Ans: D
Analysis of the test results:
| Test | Observation | Interpretation |
|---|---|---|
| NaOH test | Green precipitate | Characteristic of Fe2+ (forms Fe(OH)2) |
| Acidified AgNO3 test | Yellow precipitate | Characteristic of I– (forms AgI) |
Key chemical reactions:
- With NaOH:
FeI2 + 2NaOH → Fe(OH)2↓ (green) + 2NaI - With AgNO3:
FeI2 + 2AgNO3 → 2AgI↓ (yellow) + Fe(NO3)2
Eliminating other options:
- A/B (Copper compounds):
- Would give blue precipitate with NaOH (Cu(OH)2)
- AgCl is white, AgI is yellow (but B would match second test)
- C (FeCl2):
- Correct green precipitate with NaOH
- But would give white AgCl precipitate, not yellow
Therefore, X must be iron(II) iodide (D) as it’s the only compound that produces both:
- Green Fe(OH)2 precipitate with NaOH
- Yellow AgI precipitate with AgNO3
Which salt preparation uses a burette and a pipette?
A calcium nitrate from calcium carbonate and nitric acid
B copper(II) sulfate from copper(II) hydroxide and sulfuric acid
C potassium chloride from potassium hydroxide and hydrochloric acid
D zinc chloride from zinc and hydrochloric acid
▶️ Answer/Explanation
Ans: C
Key Concept: Burettes and pipettes are used for precise liquid volume measurements in titration reactions between two solutions.
Analysis of Options:
| Option | Reactants | Apparatus Needed | Reason |
|---|---|---|---|
| A | Solid + Acid | Beaker only | Solid carbonate added until no more reacts |
| B | Solid + Acid | Beaker only | Insoluble base added to excess acid |
| C | Alkali + Acid | Burette & Pipette | Titration required for neutralization |
| D | Metal + Acid | Beaker only | Metal added until no more reacts |
Detailed Explanation for Option C:
- Titration Setup:
- Pipette measures fixed volume of KOH solution
- Burette dispenses measured HCl volumes
- Indicator shows neutralization point
- Reaction:
KOH(aq) + HCl(aq) → KCl(aq) + H2O(l)
- Why Precision Needed:
- Both reactants are colorless solutions
- Exact 1:1 mole ratio required
- No visual indication without indicator
Why Other Options Don’t Require This Apparatus:
- A/B/D: All involve adding solid until reaction completes (visible endpoint)
- Only C requires precise liquid-liquid measurement
Therefore, C (potassium chloride preparation) is the correct answer as it’s the only option requiring titration apparatus.
The results of two tests on solid X are shown.

What is X?
A copper(II) chloride
B copper(II) iodide
C iron(II) chloride
D iron(II) iodide
▶️ Answer/Explanation
Ans: D
1. Test 1 Analysis (NaOH):
Green precipitate indicates Fe2+ (forms Fe(OH)2)
Blue precipitate would indicate Cu2+ (not observed)
2. Test 2 Analysis (HNO3 + AgNO3):
Yellow precipitate confirms I– ions (forms AgI)
White precipitate would indicate Cl– (not observed)
3. Compound Identification:
Only iron(II) iodide (FeI2) explains both:
– Green Fe(OH)2 precipitate with NaOH
– Yellow AgI precipitate with AgNO3
Which two compounds give a white precipitate when their aqueous solutions are mixed?
- silver nitrate and sodium chloride
- silver nitrate and sodium iodide
- sodium hydroxide and copper(II) sulfate
- sodium hydroxide and iron(II) chloride
▶️ Answer/Explanation
Ans: A
Analysis of precipitation reactions:
| Option | Reaction | Precipitate | Color |
|---|---|---|---|
| A | AgNO3 + NaCl → AgCl + NaNO3 | Silver chloride | White |
| B | AgNO3 + NaI → AgI + NaNO3 | Silver iodide | Yellow |
| C | 2NaOH + CuSO4 → Cu(OH)2 + Na2SO4 | Copper(II) hydroxide | Blue |
| D | 2NaOH + FeCl2 → Fe(OH)2 + 2NaCl | Iron(II) hydroxide | Green |
Key observations:
• Only option A produces a white precipitate (AgCl)
• Other options produce colored precipitates (yellow, blue, green)
• Silver chloride precipitation is a standard test for chloride ions
Thus, the correct answer is A (silver nitrate and sodium chloride).
The equations for three reactions are shown.
1 Pb(NO₃)₂(aq) + 2KI(aq) → PbI₂(s) + 2KNO₃(aq)
2 2AgNO₃(aq) + CuI₂(aq) → Cu(NO₃)₂(aq) + 2AgI(s)
3 CuO(s) + H₂SO₄(aq) → CuSO₄(aq) + H₂O(l)
Which reactions are suitable for making a salt by precipitation?
A) 1 and 2 only
B) 1 and 3 only
C) 2 and 3 only
D) 1, 2 and 3
▶️ Answer/Explanation
Ans: A
Precipitation reactions involve the formation of an insoluble solid (precipitate) when two solutions are mixed. In reaction 1, PbI₂ is the precipitate. In reaction 2, AgI is the precipitate. Reaction 3 is an acid-base neutralization producing a soluble salt (CuSO₄) and water – no precipitate forms. Therefore, only reactions 1 and 2 are precipitation reactions (option A).
Salt S is dissolved in water and three tests are carried out on the solution.

What is the identity of S?
A) copper(II) chloride
B) copper(II) sulfate
C) iron(II) chloride
D) iron(II) sulfate
▶️ Answer/Explanation
Ans: D
1. Blue precipitate with NaOH: Indicates the presence of Fe2+ (iron(II)) ions, which form a green precipitate that turns brown on standing. Copper(II) forms a blue precipitate, but the other tests rule it out.
2. No precipitate with BaCl2: Confirms the absence of sulfate ions (SO42-), eliminating options B and D.
3. White precipitate with AgNO3: Indicates chloride ions (Cl–), confirming the salt contains chloride rather than sulfate.
Combining these observations, the salt must be iron(II) chloride (Option C).
Correction: The initial answer was incorrectly marked as D. The correct identity based on the tests is C) iron(II) chloride.
The results of two tests on an aqueous solution of X are shown.

What is X?
- copper(II) chloride
- copper(II) iodide
- iron(II) chloride
- iron(II) iodide
▶️ Answer/Explanation
Ans: D
From the test results:
Test 1 (NaOH): A green precipitate forms, indicating the presence of Fe2+ (iron(II)) ions.
Test 2 (AgNO3): A yellow precipitate forms, confirming the presence of I− (iodide) ions.
Since copper(II) compounds typically form blue precipitates with NaOH and iron(II) chloride would give a white precipitate (AgCl) with AgNO3, the only possible compound is iron(II) iodide (FeI2).
Four stages used to prepare an insoluble salt are listed.
- drying
- filtration
- precipitation
- washing
In which order are the stages done?
- 2 → 1 → 3 → 4
- 3 → 2 → 4 → 1
- 3 → 4 → 1 → 2
- 4 → 3 → 2 → 1
▶️ Answer/Explanation
Ans: B
The correct sequence for preparing an insoluble salt is:
1. Precipitation: Reacting solutions to form the insoluble salt as a solid.
2. Filtration: Separating the solid precipitate from the liquid.
3. Washing: Removing soluble impurities with water.
4. Drying: Evaporating residual water to obtain the pure dry salt.
Thus, the order is 3 → 2 → 4 → 1 (Option B).
A student mixes silver nitrate and barium chloride to form a white precipitate of silver chloride.
The equation is shown.
\(2AgNO_{3}+BaCl _{2}\rightarrow 2AgCl +Ba\left ( NO_{3} \right )_{2}\)
Which row describes the solubility of the salts?

▶️ Answer/Explanation
Ans: C
1. Silver nitrate (AgNO3) is soluble in water (all nitrates are soluble).
2. Barium chloride (BaCl2) is soluble (most chlorides are soluble except AgCl, PbCl2, and Hg2Cl2).
3. Silver chloride (AgCl) is insoluble (forms a white precipitate).
4. Barium nitrate (Ba(NO3)2) is soluble (all nitrates are soluble).
Thus, the correct row is C (AgNO3 – soluble, BaCl2 – soluble, AgCl – insoluble, Ba(NO3)2 – soluble).
A student is given an unknown solution.
Which two tests provide evidence that the solution is copper(II) sulfate?
- adding dilute hydrochloric acid
- adding aqueous sodium hydroxide
- adding dilute nitric acid, then silver nitrate solution
- adding dilute nitric acid, then barium nitrate solution
A) 1 and 3
B) 1 and 4
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: D
To confirm the solution is copper(II) sulfate:
- Test 2 (NaOH): Forms a blue precipitate of copper(II) hydroxide, confirming the presence of Cu2+ ions.
- Test 4 (Ba(NO3)2): Forms a white precipitate of barium sulfate, confirming the presence of SO42− ions.
Tests 1 and 3 are irrelevant for identifying copper(II) sulfate, as HCl does not react with CuSO4, and AgNO3 tests for chloride ions (not sulfate).
Which substance reacts with dilute sulfuric acid to form a salt that can be removed from the resulting mixture by filtration?
A) aqueous barium chloride
B) aqueous sodium hydroxide
C) copper
D) copper(II) carbonate
▶️ Answer/Explanation
Ans: A
1. Aqueous barium chloride (A) reacts with dilute sulfuric acid to form barium sulfate (BaSO₄), which is insoluble and can be filtered out.
2. Sodium hydroxide (B) forms a soluble salt (Na₂SO₄).
3. Copper (C) does not react with dilute sulfuric acid.
4. Copper(II) carbonate (D) forms soluble copper sulfate (CuSO₄).
Thus, only BaSO₄ (from option A) precipitates and can be separated by filtration.
A salt is made by adding an excess of an insoluble metal oxide to an acid. How is the excess metal oxide removed from the mixture?
A) chromatography
B) crystallisation
C) distillation
D) filtration
▶️ Answer/Explanation
Ans: D
To remove the excess insoluble metal oxide:
1. Filtration is used because the metal oxide is a solid, while the desired salt remains dissolved in the liquid.
2. The solid oxide is trapped by the filter paper, while the filtrate contains the salt solution.
3. Chromatography (A) separates mixtures based on solubility, crystallisation (B) forms crystals from a solution, and distillation (C) purifies liquids—none of these separate solids from liquids effectively.
