Pre AP Chemistry -4.1B Representations of Precipitation Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.1B Representations of Precipitation Reactions- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.1B Representations of Precipitation Reactions- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which symbol equation represents the reaction between aqueous sodium hydroxide and dilute sulfuric acid?
A) Na₂OH + H₂SO₄ → 2NaSO₄ + H₂O
B) Na(OH)₂ + H₂SO₄ → Na₂SO₄ + 2H₂O
C) 2NaOH + H₂SO₄ → 2NaSO₄ + 2H₂O
D) 2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
▶️ Answer/Explanation
Ans: D
This is a neutralization reaction between an acid (H₂SO₄) and a base (NaOH).
The correct balanced equation is:
\[ 2NaOH + H_2SO_4 \rightarrow Na_2SO_4 + 2H_2O \]
Key points:
- Sodium hydroxide formula is NaOH (not Na₂OH or Na(OH)₂)
- The sulfate ion is SO₄²⁻, so sodium sulfate is Na₂SO₄ (not NaSO₄)
- The equation must balance for atoms and charge
Option D is the only one that meets all these requirements.
A student carries out an experiment to prepare pure magnesium sulfate crystals.
The diagram shows the first stage of the preparation.

He adds magnesium carbonate until no more reacts.
Which process should he use for the next stage?
A crystallisation
B evaporation
C filtration
D neutralisation
▶️ Answer/Explanation
Ans: C (Filtration)
The correct sequence of steps is:
- Reaction stage:
MgCO3(s) + H2SO4(aq) → MgSO4(aq) + H2O(l) + CO2(g)
- Next stage (immediate step):
- Must first filter to remove excess unreacted MgCO3
- This separates the solid from the MgSO4 solution
- Final stage:
- After filtration, then perform evaporation and crystallization
- But filtration must come first
Why other options are incorrect at this stage:
- A (Crystallization): Done after filtration
- B (Evaporation): Done after filtration
- D (Neutralization): Not required – reaction is already complete
Therefore, the immediate next step after the reaction is filtration (C) to remove the excess solid magnesium carbonate before proceeding to crystallization.
When pink crystals of cobalt(II) chloride are heated, steam is given off and the colour of the solid changes to blue.
$\mathrm{CoCl}_2 \cdot 6 \mathrm{H}_2 \mathrm{O} \rightleftharpoons \mathrm{CoCl}_2+6 \mathrm{H}_2 \mathrm{O}$
What happens when water is added to the blue solid?

▶️ Answer/Explanation
Ans: B
When water is added to anhydrous cobalt(II) chloride (blue solid):
- Color Change:
- Blue anhydrous CoCl2 → Pink hydrated [Co(H2O)6]Cl2
- This is a characteristic test for water
- Energy Change:
- The hydration process is exothermic (heat is released)
- Temperature of the solution increases
- Chemical Process:
- CoCl2 + 6H2O → [Co(H2O)6]Cl2
- Hexaaquacobalt(II) chloride forms (pink complex)
Thus, the correct observations are:
• Color changes to pink
• Temperature increases
This matches option B in the answer choices.
X is a pink solid.
Y is a blue solid.
When X is heated, water is produced and the solid turns blue.
When water is added to Y, the solid turns pink.
What are X and Y?
▶️ Answer/Explanation
Ans: B
Identification of Substances:
X: Cobalt(II) chloride hexahydrate (CoCl2·6H2O)
- Pink solid in hydrated form
- When heated: CoCl2·6H2O → CoCl2 + 6H2O
- Loses water to form blue anhydrous cobalt(II) chloride
Y: Anhydrous cobalt(II) chloride (CoCl2)
- Blue solid in anhydrous form
- When water added: CoCl2 + 6H2O → CoCl2·6H2O
- Absorbs water to form pink hydrated cobalt(II) chloride
Chemical Explanation:
- The color change is due to formation of different coordination complexes:
- Pink: [Co(H2O)6]2+ complex
- Blue: CoCl42- complex in anhydrous form
- This reversible reaction is used as a moisture indicator
Why Other Options Are Incorrect:
- A: Copper compounds don’t show this exact color change pattern
- C: Iron compounds have different colors (green/yellow)
- D: Zinc compounds are typically white
Therefore, the correct identification is B (CoCl2·6H2O and CoCl2), as these are the only substances that exhibit the described reversible color changes with water.
Which two compounds give a white precipitate when their aqueous solutions are mixed?
A) Silver nitrate and sodium chloride
B) Silver nitrate and sodium iodide
C) Sodium hydroxide and copper(II) sulfate
D) Sodium hydroxide and iron(II) chloride
▶️ Answer/Explanation
Ans: A
1. Precipitation Reaction:
AgNO3(aq) + NaCl(aq) → AgCl↓(white) + NaNO3(aq)
• Silver chloride (AgCl) has very low solubility (Ksp = 1.8×10-10)
• Forms immediately as a curdy white precipitate 2. Why Other Options Are Incorrect:
• B: Forms yellow AgI precipitate
• C: Forms blue Cu(OH)2 precipitate
• D: Forms green Fe(OH)2 precipitate
3. Key Characteristics:
• AgCl darkens in sunlight (photochemical decomposition)
• Used in photographic films and chloride ion tests
• The only white precipitate among the options
A method used to make copper(II) sulfate crystals is shown.
- Place dilute sulfuric acid in a beaker.
- Warm the acid.
- Add copper(II) oxide until it is in excess.
- Filter the mixture.
- Evaporate the filtrate until crystals start to form.
- Leave the filtrate to cool.
What are the purposes of step 3 and step 4?

▶️ Answer/Explanation
Ans: B
Analysis of the key steps in copper(II) sulfate preparation:
| Step | Purpose | Chemical Explanation |
|---|---|---|
| 3 (Add excess CuO) | Ensure complete acid neutralization | CuO + H2SO4 → CuSO4 + H2O Excess solid guarantees all H2SO4 reacts |
| 4 (Filtration) | Remove unreacted CuO | Separates soluble CuSO4 solution from insoluble excess CuO |
Key chemical principles:
- The reaction: CuO(s) + H2SO4(aq) → CuSO4(aq) + H2O(l)
- Excess CuO ensures:
- Maximum yield of CuSO4
- No leftover acid that could interfere with crystallization
- Filtration provides:
- Pure CuSO4 solution
- Removal of impurities and excess reactant
Therefore, option B correctly identifies that step 3 ensures complete reaction of the acid, and step 4 removes excess solid, allowing for pure crystal formation in subsequent steps.
Copper(II) sulfate crystals are blue. They are made by adding an excess of copper(II) oxide to sulfuric acid.
The mixture is heated and stirred.
It is then filtered and the filtrate is allowed to evaporate, leaving blue crystals.
Why is filtration necessary?
- to remove soluble properties
- to remove sulfuric acid
- to remove the blue crystals
- to remove unreacted copper(II) oxide
▶️ Answer/Explanation
Ans: D (to remove unreacted copper(II) oxide)
Process analysis:
- Reaction:
CuO(s) + H2SO4(aq) → CuSO4(aq) + H2O(l)
- Copper(II) oxide is insoluble (solid reactant)
- Copper(II) sulfate forms soluble blue solution
- Filtration purpose:
- Separates soluble CuSO4 solution from insoluble CuO excess
- Ensures only pure copper sulfate solution remains for crystallization
- Why other options are incorrect:
- A: Filtration doesn’t remove soluble properties
- B: Sulfuric acid reacts completely (excess CuO ensures this)
- C: Blue crystals form after evaporation, not before filtration
The filtration step is crucial for removing the solid excess reactant (CuO) while retaining the desired product in solution.
Information about the solubility in water of four oxides is shown.
Which oxide, when added to water, gives a solution with a pH less than pH7?

▶️ Answer/Explanation
Ans: A
1. Acidic Oxide Identification:
Only NO2 forms acidic solution (pH < 7) via the reaction:
2NO2 + H2O → HNO3 + HNO2
2. Other Oxides:
– CaO: Forms Ca(OH)2 (basic, pH > 7)
– MgO: Forms Mg(OH)2 (basic, pH > 7)
– SiO2: Insoluble (neutral, pH ≈ 7)
3. Key Characteristic:
NO2 is the only oxide listed that produces strong acids in water.
The apparatus shown is used to prepare aqueous copper(II) sulfate.
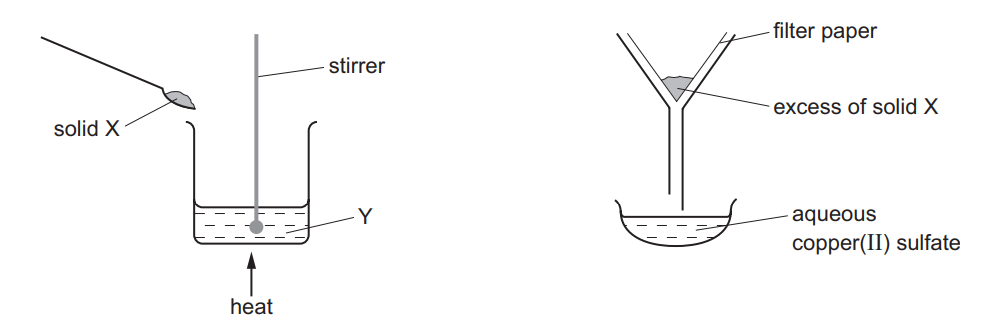
What are X and Y?
▶️ Answer/Explanation
Ans: C (X = copper(II) oxide, Y = sulfuric acid)
The preparation of copper(II) sulfate involves:
- Reaction:
CuO(s) + H2SO4(aq) → CuSO4(aq) + H2O(l)
- Copper(II) oxide (black solid) reacts with sulfuric acid
- Produces blue copper(II) sulfate solution
- Apparatus setup:
- X must be the solid reactant (CuO)
- Y must be the liquid reactant (H2SO4)
- The filter funnel separates unreacted CuO from the solution
Why other options are incorrect:
- A/B: Copper metal won’t react with sulfuric acid at room temperature
- D: Copper carbonate would react but produce CO2 gas (not shown in apparatus)
Therefore, the correct combination is C (CuO and H2SO4).
Aqueous sodium hydroxide is added to solid Q in a test-tube. A gas is produced which turns damp red litmus blue. What is Q?
- aluminium
- ammonia
- ammonium chloride
- sodium nitrate
▶️ Answer/Explanation
Ans: C
The key observations and reactions:
- Gas Identification:
- Turns damp red litmus blue → basic gas (ammonia, NH3)
- Reaction Analysis:
- NaOH + NH4Cl → NaCl + H2O + NH3↑
- This is the only reaction among the options that produces ammonia gas
- Eliminating Other Options:
- A (Aluminium): Produces hydrogen gas (H2) with NaOH
- B (Ammonia): Already a gas, not a solid reactant
- D (NaNO3): No reaction with NaOH
The chemical equation for the correct reaction:
NH4Cl + NaOH → NaCl + NH3 + H2O
Thus, the only solid that fits all observations is ammonium chloride (C).
When pink crystals of cobalt(II) chloride are heated, steam is given off and the colour of the solid changes to blue.
$\mathrm{CoCl}_2 \cdot 6 \mathrm{H}_2 \mathrm{O} \rightleftharpoons \mathrm{CoCl}_2+6 \mathrm{H}_2 \mathrm{O}$
What happens when water is added to the blue solid?

▶️ Answer/Explanation
Ans: B
Chemical Process Analysis:
- Reversible Reaction:
- Hydrated form (pink): $\mathrm{CoCl}_2 \cdot 6\mathrm{H}_2\mathrm{O}$
- Anhydrous form (blue): $\mathrm{CoCl}_2$
- Adding Water to Anhydrous Form:
- Blue $\mathrm{CoCl}_2$ rehydrates to form pink $\mathrm{CoCl}_2 \cdot 6\mathrm{H}_2\mathrm{O}$
- Process is exothermic (heat released)
- Solution turns pink as cobalt ions hydrate
- Why Option B is Correct:
- Color change: blue solid → pink solution
- Temperature increases (exothermic hydration)
- Both observations match the description
The complete transformation involves:
- Rehydration of cobalt ions: $\mathrm{Co}^{2+}$ becomes surrounded by water molecules
- Formation of octahedral $\mathrm{[Co(H_2O)_6]}^{2+}$ complex ions (pink color)
- Release of hydration energy (temperature rise)
A method used to make copper(II) sulfate crystals is shown.
1 Place dilute sulfuric acid in a beaker.
2 Warm the acid.
3 Add copper(II) oxide until it is in excess.
4 Filter the mixture.
5 Evaporate the filtrate until crystals start to form.
6 Leave the filtrate to cool.
What are the purposes of step 3 and step 4?

▶️ Answer/Explanation
Ans: B
Detailed explanation of steps 3 and 4:
| Step | Purpose | Chemical Explanation |
|---|---|---|
| 3 (Add excess CuO) | Ensure complete reaction of acid | CuO + H2SO4 → CuSO4 + H2O Excess CuO guarantees all H2SO4 reacts |
| 4 (Filter) | Remove unreacted CuO | Separates soluble CuSO4 solution from insoluble excess CuO |
Key process details:
- Why excess CuO is added:
- Ensures maximum yield of CuSO4
- Prevents residual acid that would interfere with crystallization
- Excess solid is easily removable by filtration
- Why filtration is crucial:
- Produces pure CuSO4 solution for crystallization
- Removes impurities and unreacted starting material
- Prevents contamination of final crystals
The image shows option B correctly matching these purposes: “to make sure all the acid reacts” (step 3) and “to remove the excess copper(II) oxide” (step 4).
Four stages in the preparation of a salt from an acid and a solid metal oxide are listed.
1 Add excess solid.
2 Evaporate half the solution and leave to cool.
3 Filter to remove unwanted solid.
4 Heat the acid.
In which order should the stages be carried out?
A 1 → 3 → 4 → 2
B 2 → 1 → 3 → 4
C 4 → 1 → 3 → 2
D 4 → 2 → 1 → 3
▶️ Answer/Explanation
Ans: C (4 → 1 → 3 → 2)
The correct sequence for salt preparation is:
- Heat the acid (4):
- Increases reaction rate between acid and metal oxide
- Ensures complete reaction
- Add excess solid (1):
- Ensures all acid reacts completely
- Prevents acid contamination in final product
- Filter (3):
- Removes unreacted excess metal oxide
- Obtains pure salt solution
- Evaporate and cool (2):
- Concentrates solution by evaporating half
- Cooling allows salt crystals to form
Key reasons why other sequences are incorrect:
- A/B: Heating must come first to initiate reaction
- D: Evaporation cannot occur before reaction is complete
Therefore, the correct order is 4 → 1 → 3 → 2 (Option C).
Four steps to prepare a salt from an excess of a solid base and an acid are listed.
1 crystallisation
2 evaporation
3 filtration
4 neutralisation
In which order are the steps carried out?
A 2 → 3 → 4 → 1
B 3 → 1 → 2 → 4
C 4 → 2 → 1 → 3
D 4 → 3 → 2 → 1
▶️ Answer/Explanation
Ans: D
Step-by-Step Process:
- Neutralisation (Step 4):
- Mix acid with excess solid base to form salt solution
- Example: HCl + CuO → CuCl2 + H2O
- Filtration (Step 3):
- Filter to remove unreacted solid base (excess)
- Obtain pure salt solution as filtrate
- Evaporation (Step 2):
- Gently heat solution to concentrate it
- Stop when crystals form on glass rod (saturation point)
- Crystallisation (Step 1):
- Allow solution to cool slowly for crystal formation
- Filter to collect pure dry crystals
Why This Order is Correct:
- Must react first (neutralisation) before separating
- Need to filter before evaporating to remove impurities
- Evaporation must precede crystallisation for saturation
- Final crystals are obtained only after all other steps
Practical Tip: The excess solid base ensures all acid is neutralized and helps in easy separation by filtration.
The equations for three reactions are shown.
1 Pb(NO₃)₂(aq) + 2KI(aq) → PbI₂(s) + 2KNO₃(aq)
2 2AgNO₃(aq) + CuI₂(aq) → Cu(NO₃)₂(aq) + 2AgI(s)
3 CuO(s) + H₂SO₄(aq) → CuSO₄(aq) + H₂O(l)
Which reactions are suitable for making a salt by precipitation?
A) 1 and 2 only
B) 1 and 3 only
C) 2 and 3 only
D) 1, 2 and 3
▶️ Answer/Explanation
Ans: A
Precipitation reactions involve the formation of an insoluble solid (precipitate) when two solutions are mixed. In reaction 1, PbI₂ is the precipitate. In reaction 2, AgI is the precipitate. Reaction 3 is an acid-base neutralization producing a soluble salt (CuSO₄) and water – no precipitate forms. Therefore, only reactions 1 and 2 are precipitation reactions (option A).
Copper(II) sulfate is made when copper(II) carbonate reacts with dilute sulfuric acid.
\[ \text{CuCO}_3 + \text{H}_2\text{SO}_4 \rightarrow \text{CuSO}_4 + \text{H}_2\text{O} + \text{CO}_2 \]
Pure copper(II) sulfate crystals are obtained.
Which reagent is in excess and how are the crystals obtained?
| reagent in excess | how the crystals are obtained | |
|---|---|---|
| A | copper(II) carbonate | filter and evaporate the solution to dryness |
| B | copper(II) carbonate | filter, evaporate the solution to crystallising point and then cool |
| C | dilute sulfuric acid | evaporate the solution to dryness |
| D | dilute sulfuric acid | evaporate the solution to crystallising point and then cool |
▶️ Answer/Explanation
Ans: B
Copper(II) carbonate is insoluble, so it’s typically added in excess to ensure all the acid reacts. The excess solid can then be removed by filtration. To obtain pure crystals, the solution should be evaporated to the crystallising point (not to dryness, which would produce an impure powder) and then cooled to allow crystals to form. This method gives larger, purer crystals than evaporating to dryness.
Which process is not used in the preparation of an insoluble salt?
A) filtration
B) washing
C) crystallisation
D) drying
▶️ Answer/Explanation
Ans: C
Insoluble salts are prepared by mixing two soluble salts to form a precipitate. The steps involved are:
1. Filtration to separate the insoluble salt from the solution.
2. Washing to remove impurities.
3. Drying to obtain the pure solid.
Crystallisation is used for soluble salts, not insoluble ones, making it the correct answer.
Information about some silver compounds is shown.

Which equation shows a reaction which cannot be used to make a silver salt?
A. \(AgNO_{3}(aq)+HC\imath (aq)\rightarrow AgC\imath (s)+HNO_{3}(aq)\)
B. \(Ag_{2}O(s)+2HNO_{3}(aq)\rightarrow 2AgNO_{3}(aq)+H_{2}O(l)\)
C. \(Ag_{2}CO_{3}(s)+2HNO_{3}(aq)\rightarrow 2AgNO_{3}(aq)+H_{2}O(l)+CO_{2}(g)\)
D. \(2Ag(s)+2HC\imath (aq)\rightarrow 2AgC\imath (s)+H_{2}(g)\)
▶️ Answer/Explanation
Ans: D
To determine which reaction cannot be used to make a silver salt, we analyze each option:
A: Silver nitrate reacts with HCl to form silver chloride (a silver salt).
B: Silver oxide reacts with nitric acid to produce silver nitrate (a silver salt).
C: Silver carbonate reacts with nitric acid to form silver nitrate (a silver salt).
D: Silver metal does not react with HCl under normal conditions, making this reaction invalid for producing silver chloride. Thus, D is the correct answer.
An acid is neutralized by adding an excess of an insoluble solid base.
A soluble salt is formed.
How is the pure salt obtained from the reaction mixture?
- crystallization → evaporation → filtration
- evaporation → crystallization → filtration
- filtration → crystallization → evaporation
- filtration → evaporation → crystallization
▶️ Answer/Explanation
Ans: D
To obtain the pure salt from the reaction mixture:
1. Filtration removes the excess insoluble base.
2. Evaporation concentrates the solution to near saturation.
3. Crystallization allows the pure salt to form as crystals upon cooling.
Thus, the correct sequence is filtration → evaporation → crystallization (Option D).
Copper(II) chloride crystals are made by adding solid copper(II) carbonate to dilute hydrochloric acid until no more dissolves.
Which process is used to obtain pure copper(II) chloride crystals from the mixture?
A) distillation of the mixture
B) evaporation of the mixture
C) filtration followed by drying of the residue
D) filtration followed by evaporation of the filtrate
▶️ Answer/Explanation
Ans: D
To obtain pure copper(II) chloride crystals:
1. First, filter the mixture to remove the undissolved copper(II) carbonate residue.
2. The filtrate contains dissolved copper(II) chloride in water.
3. Evaporate the filtrate to remove water, leaving behind pure copper(II) chloride crystals.
Distillation (A) is unnecessary, and evaporation alone (B) would leave impurities. Drying the residue (C) would yield the wrong product.
The following substances can be reacted together to prepare salts.
- copper(II) oxide and excess hydrochloric acid
- hydrochloric acid and excess sodium hydroxide
- hydrochloric acid and excess zinc carbonate
In which reactions can the excess reactant be separated from the solution by filtration?
A) 1 and 2
B) 1 and 3
C) 2 and 3
D) 3 only
▶️ Answer/Explanation
Ans: B
Filtration is used to separate an insoluble excess reactant from a solution.
1. Copper(II) oxide is insoluble, so excess can be filtered out.
2. Sodium hydroxide is soluble, so excess cannot be filtered.
3. Zinc carbonate is insoluble, so excess can be filtered out.
Thus, only reactions 1 and 3 allow filtration of excess reactant.
Salt S is dissolved in water and three tests are carried out on the solution.

What is the identity of S?
A) copper(II) chloride
B) copper(II) sulfate
C) iron(II) chloride
D) iron(II) sulfate
▶️ Answer/Explanation
Ans: D
1. Blue precipitate with NaOH: Indicates the presence of Fe2+ (iron(II)) ions, which form a green precipitate that turns brown on standing. Copper(II) forms a blue precipitate, but the other tests rule it out.
2. No precipitate with BaCl2: Confirms the absence of sulfate ions (SO42-), eliminating options B and D.
3. White precipitate with AgNO3: Indicates chloride ions (Cl–), confirming the salt contains chloride rather than sulfate.
Combining these observations, the salt must be iron(II) chloride (Option C).
Correction: The initial answer was incorrectly marked as D. The correct identity based on the tests is C) iron(II) chloride.
The apparatus shown is used to prepare aqueous copper(II) sulfate.

What are X and Y?

▶️ Answer/Explanation
Ans: C
To prepare aqueous copper(II) sulfate:
1. X must be copper(II) oxide (a base) to react with sulfuric acid (Y).
2. Y is sulfuric acid, which reacts with copper(II) oxide to form copper(II) sulfate and water.
The reaction is: \(\text{CuO} + \text{H}_2\text{SO}_4 \rightarrow \text{CuSO}_4 + \text{H}_2\text{O}\).
Filtration is needed to remove excess insoluble copper(II) oxide, leaving aqueous copper(II) sulfate.
Sodium carbonate reacts with sulfuric acid to form carbon dioxide, water and a sodium salt.
An incomplete equation for the reaction is shown.
$ \mathrm{Na}_2 \mathrm{CO}_3+\mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{CO}_2+\mathrm{H}_2 \mathrm{O}+ $
What is the formula of the sodium salt?
A) $\mathrm{Na}_2\left(\mathrm{SO}_4\right)_2$
B) $\mathrm{Na}\left(\mathrm{SO}_4\right)_2$
C) $\mathrm{Na}_2 \mathrm{SO}_4$
D) $\mathrm{NaSO}_4$
▶️ Answer/Explanation
Ans: C
The reaction between sodium carbonate ($\mathrm{Na}_2 \mathrm{CO}_3$) and sulfuric acid ($\mathrm{H}_2 \mathrm{SO}_4$) is a neutralization reaction. The sodium salt formed is sodium sulfate ($\mathrm{Na}_2 \mathrm{SO}_4$), as the sodium ions ($\mathrm{Na}^+$) from sodium carbonate combine with the sulfate ions ($\mathrm{SO}_4^{2-}$) from sulfuric acid. The balanced equation is:
$\mathrm{Na}_2 \mathrm{CO}_3 + \mathrm{H}_2 \mathrm{SO}_4 \rightarrow \mathrm{CO}_2 + \mathrm{H}_2 \mathrm{O} + \mathrm{Na}_2 \mathrm{SO}_4$.
Option C is correct because $\mathrm{Na}_2 \mathrm{SO}_4$ maintains charge balance (2 $\mathrm{Na}^+$ ions for 1 $\mathrm{SO}_4^{2-}$ ion).
Lead(II) sulfate is an insoluble salt.
Which process is not used to prepare a pure sample of this salt?
- crystallization
- drying
- filtration
- precipitation
▶️ Answer/Explanation
Ans: A
Lead(II) sulfate is prepared by precipitation (mixing solutions of lead(II) nitrate and sodium sulfate). The solid is then separated by filtration and dried. Crystallization is not used because the salt is insoluble and does not form crystals from a solution. Thus, the correct answer is A (crystallization), as it is the process not involved in the preparation.
A method used to make copper(II) sulfate crystals is shown.
- Place dilute sulfuric acid in a beaker.
- Warm the acid.
- Add copper(II) oxide until it is in excess.
- Filter the mixture.
- Evaporate the filtrate until crystals start to form.
- Leave the filtrate to cool.
What are the purposes of step 3 and step 4?
▶️ Answer/Explanation
Ans: B
Step 3 (Adding excess CuO): Ensures all sulfuric acid reacts, maximizing copper(II) sulfate formation.
Step 4 (Filtration): Removes unreacted CuO (insoluble), leaving pure copper(II) sulfate solution.
The combination guarantees complete reaction and purification, making B the correct choice.
Which method is used to make the salt copper(II) sulfate?
- dilute acid + alkali
- dilute acid + carbonate
- dilute acid + metal
- dilute acid + non-metal oxide
▶️ Answer/Explanation
Ans: B
To make copper(II) sulfate:
1. Copper(II) carbonate (a base) reacts with dilute sulfuric acid to form copper(II) sulfate, water, and carbon dioxide.
2. The reaction is: \[ \text{CuCO}_3 + \text{H}_2\text{SO}_4 \rightarrow \text{CuSO}_4 + \text{H}_2\text{O} + \text{CO}_2 \]
3. Copper does not react well with dilute acids (C), and non-metal oxides (D) do not form salts in this way. Alkalis (A) are not needed since carbonates suffice.
A student mixes silver nitrate and barium chloride to form a white precipitate of silver chloride.
The equation is shown.
\(2AgNO_{3}+BaCl _{2}\rightarrow 2AgCl +Ba\left ( NO_{3} \right )_{2}\)
Which row describes the solubility of the salts?

▶️ Answer/Explanation
Ans: C
1. Silver nitrate (AgNO3) is soluble in water (all nitrates are soluble).
2. Barium chloride (BaCl2) is soluble (most chlorides are soluble except AgCl, PbCl2, and Hg2Cl2).
3. Silver chloride (AgCl) is insoluble (forms a white precipitate).
4. Barium nitrate (Ba(NO3)2) is soluble (all nitrates are soluble).
Thus, the correct row is C (AgNO3 – soluble, BaCl2 – soluble, AgCl – insoluble, Ba(NO3)2 – soluble).
A pure sample of the insoluble salt barium carbonate can be made using the method given.
- step 1 Dissolve barium chloride in water.
- step 2 Separately dissolve sodium carbonate in water.
- step 3 Mix the two solutions together.
- step 4 Filter the mixture.
- step 5
- step 6 Dry the residue between two sheets of filter paper.
Which instruction is missing from step 5?
A) Heat the residue to dryness.
B) Heat the residue to the point of crystallisation.
C) Place the filtrate in an evaporating basin.
D) Wash the residue with water.
▶️ Answer/Explanation
Ans: D
After filtration (step 4), the residue (barium carbonate) will contain impurities such as soluble salts (e.g., NaCl). To obtain a pure sample, the residue must be washed with water (step 5) to remove these impurities before drying (step 6). Heating (options A and B) or evaporating the filtrate (option C) would not purify the insoluble barium carbonate. Therefore, the correct missing step is D) Wash the residue with water.
The diagram shows the steps in the preparation of a salt.
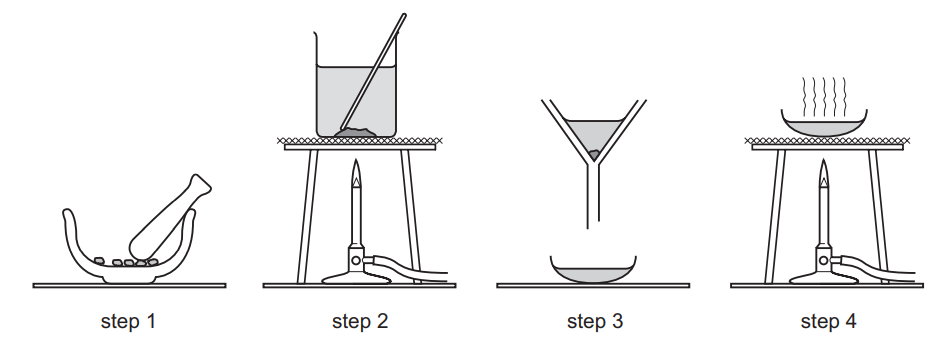
Which salt is prepared by this method?
- barium sulfate
- copper(II) sulfate
- potassium sulfate
- sodium sulfate
▶️ Answer/Explanation
Ans: B
The diagram depicts the preparation of a soluble salt (likely involving a metal oxide or carbonate reacting with an acid, followed by crystallization). Copper(II) sulfate is commonly prepared by reacting copper(II) oxide or carbonate with sulfuric acid, then crystallizing the solution. Barium sulfate is insoluble (prepared by precipitation), while potassium and sodium sulfates are typically made via neutralization of alkalis, not metal oxides/carbonates. Thus, the correct answer is B (copper(II) sulfate).
The apparatus shown is used to prepare aqueous copper(II) sulfate.
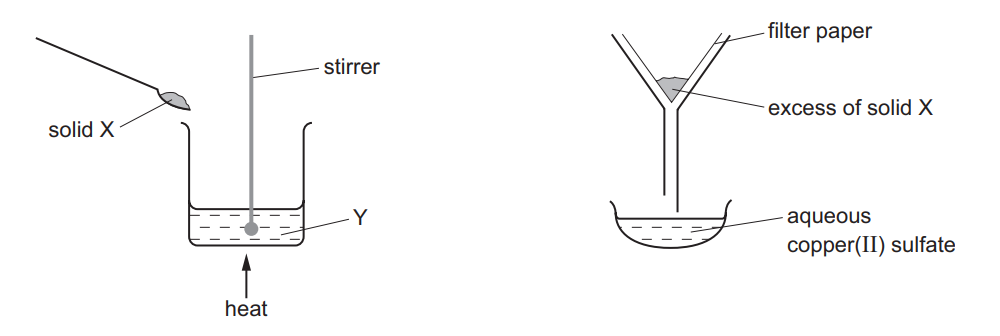
What are X and Y?
▶️ Answer/Explanation
Ans: C
X (Copper(II) oxide): Reacts with sulfuric acid to form copper(II) sulfate. Y (Filter funnel): Separates unreacted copper(II) oxide (insoluble) from the copper(II) sulfate solution. The process involves neutralization (acid + base) followed by filtration, making C the correct pairing.
Information about some silver compounds is shown in the table.
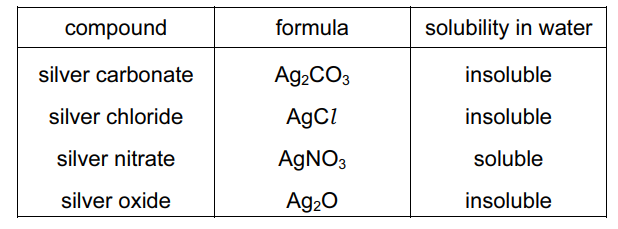
Which equation shows a reaction which cannot be used to make a silver salt?
A) $\mathrm{AgNO}_3(\mathrm{aq})+\mathrm{HCl}(\mathrm{aq}) \rightarrow \mathrm{AgCl}(\mathrm{s})+\mathrm{HNO}_3(\mathrm{aq})$
B) $\mathrm{Ag}_2 \mathrm{O}(\mathrm{s})+2 \mathrm{HNO}_3(\mathrm{aq}) \rightarrow 2 \mathrm{AgNO}_3(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{I})$
C) $\mathrm{Ag}_2 \mathrm{CO}_3(\mathrm{~s})+2 \mathrm{HNO}_3(\mathrm{aq}) \rightarrow 2 \mathrm{AgNO}_3(\mathrm{aq})+\mathrm{H}_2 \mathrm{O}(\mathrm{I})+\mathrm{CO}_2(\mathrm{~g})$
D) $2 \mathrm{Ag}(\mathrm{s})+2 \mathrm{HCl}(\mathrm{aq}) \rightarrow 2 \mathrm{AgCl}(\mathrm{s})+\mathrm{H}_2(\mathrm{~g})$
▶️ Answer/Explanation
Ans: D
To determine which reaction cannot produce a silver salt, we analyze each option:
A: Forms $\mathrm{AgCl}$ (silver chloride) via precipitation – valid.
B: Forms $\mathrm{AgNO_3}$ (silver nitrate) via acid-base reaction – valid.
C: Produces $\mathrm{AgNO_3}$ with $\mathrm{CO_2}$ release – valid.
D: Silver metal ($\mathrm{Ag}$) cannot displace hydrogen from $\mathrm{HCl}$ as it is below hydrogen in the reactivity series. This reaction does not occur under normal conditions, making it invalid for salt preparation.
Which substance reacts with dilute sulfuric acid to form a salt that can be removed from the resulting mixture by filtration?
A) aqueous barium chloride
B) aqueous sodium hydroxide
C) copper
D) copper(II) carbonate
▶️ Answer/Explanation
Ans: A
1. Aqueous barium chloride (A) reacts with dilute sulfuric acid to form barium sulfate (BaSO₄), which is insoluble and can be filtered out.
2. Sodium hydroxide (B) forms a soluble salt (Na₂SO₄).
3. Copper (C) does not react with dilute sulfuric acid.
4. Copper(II) carbonate (D) forms soluble copper sulfate (CuSO₄).
Thus, only BaSO₄ (from option A) precipitates and can be separated by filtration.
