Pre AP Chemistry -4.1C Molarity and Solution Concentration- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.1C Molarity and Solution Concentration- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.1C Molarity and Solution Concentration- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
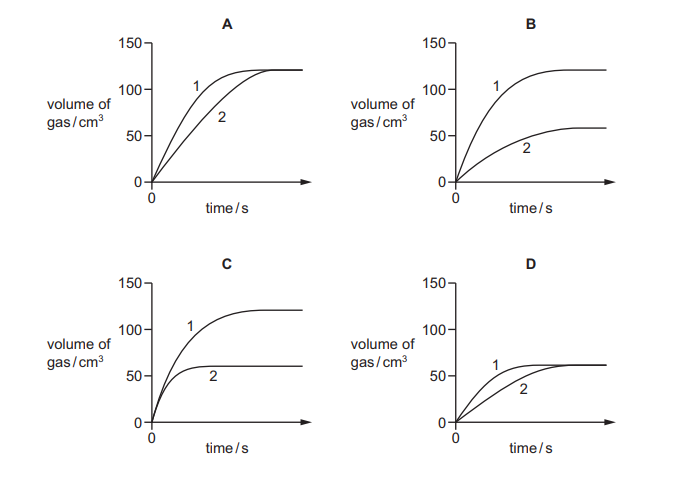
Excess calcium carbonate is added to 50 cm³ of dilute hydrochloric acid of different concentrations in two separate experiments. The volume of gas produced in experiment 1 and in experiment 2 is measured every 30 seconds. The results are shown.

Which diagram represents the data collected?
▶️ Answer/Explanation
Ans: B
Key Observations:
- Excess Calcium Carbonate means HCl is the limiting reactant in both experiments.
- Different HCl Concentrations will produce different reaction rates but the same total gas volume (since same HCl volume is used).
- Graph Analysis:
- Both curves should plateau at the same maximum volume (equal moles of gas produced).
- Experiment with higher HCl concentration (faster reaction) will reach plateau sooner (steeper initial slope).
Why Option B is Correct:
- Shows two curves reaching the same final volume.
- One curve rises faster (higher concentration) while the other rises slower (lower concentration).
- Matches the expected behavior for this chemical reaction.
Thus, B accurately represents the experimental data.
The concentration and volume of an aqueous alkali are known. Which additional information is required to calculate the number of moles of acid needed to neutralise the aqueous alkali?
A) the concentration of the acid
B) the equation for the acid–alkali reaction
C) the formula of the acid
D) the volume of the acid required for neutralisation
▶️ Answer/Explanation
Ans: B
1. The moles of alkali can be calculated using its concentration and volume (\(n = cV\)).
2. To find the moles of acid needed, the stoichiometric ratio from the balanced equation is required (e.g., 1:1 for HCl + NaOH → NaCl + H2O).
3. Options A, C, and D are irrelevant because:
– The acid’s concentration (A) or volume (D) only helps find the acid’s moles after knowing the required ratio.
– The formula (C) alone doesn’t provide the reaction ratio.
Thus, the equation (B) is essential to determine the mole ratio for neutralisation.
The equation for the reaction between aqueous lead(II) nitrate and aqueous sodium chloride is shown.
Pb(NO3)2(aq) + 2NaCl(aq) → PbCl2(s) + 2NaNO3(aq)
If 100 cm3 of aqueous lead(II) nitrate of concentration 0.1 mol / dm3 is reacted with an excess of aqueous sodium chloride, which mass of lead(II) chloride is obtained?
A 1.16 g B 2.42 g C 2.78 g D 3.31 g
▶️ Answer/Explanation
Ans: C
Step 1: Calculate moles of Pb(NO3)2
Volume = 100 cm3 = 0.1 dm3
Moles = Concentration × Volume = 0.1 mol/dm3 × 0.1 dm3 = 0.01 mol
Step 2: Determine moles of PbCl2 formed
From the balanced equation: 1 mol Pb(NO3)2 produces 1 mol PbCl2
∴ Moles of PbCl2 = 0.01 mol
Step 3: Calculate mass of PbCl2
Molar mass of PbCl2 = 207 + (2 × 35.5) = 278 g/mol
Mass = Moles × Molar mass = 0.01 mol × 278 g/mol = 2.78 g
Therefore, the correct answer is C (2.78 g).
A tablet contains 0.080 g of ascorbic acid (\(M_{r}\) = 176). What is the concentration of ascorbic acid when one tablet is dissolved in 200 cm3 of water?
A. 9.1 × \(10^{-5}\) mol / \(dm^{3}\)
B. 4.5 × \(10^{-4}\) mol / \(dm^{3}\)
C. 9.1 × \(10^{-2}\) mol / \(dm^{3}\)
D. 2.3 × \(10^{-3}\) mol / \(dm^{3}\)
▶️ Answer/Explanation
Ans: D
1. Calculate moles of ascorbic acid:
\( \text{Moles} = \frac{\text{Mass}}{M_r} = \frac{0.080\, \text{g}}{176\, \text{g/mol}} = 4.55 \times 10^{-4}\, \text{mol} \)
2. Convert volume to dm3:
\( 200\, \text{cm}^3 = 0.200\, \text{dm}^3 \)
3. Calculate concentration:
\( \text{Concentration} = \frac{\text{Moles}}{\text{Volume}} = \frac{4.55 \times 10^{-4}\, \text{mol}}{0.200\, \text{dm}^3} = 2.28 \times 10^{-3}\, \text{mol/dm}^3 \)
4. Round to match options:
The closest option is D (\( 2.3 \times 10^{-3}\, \text{mol/dm}^3 \)).
The concentration of a hydrochloric acid solution is 0.5 mol/dm3.
How many moles of hydrochloric acid are present in 25 cm3 of this solution?
A. 0.0125
B. 0.0200
C. 12.5
D. 20.0
▶️ Answer/Explanation
Ans: A
1. Convert volume to dm3:
25 cm3 = \(\frac{25}{1000} = 0.025 \text{ dm}^3\).
2. Calculate moles of HCl:
Moles = Concentration × Volume = \(0.5 \text{ mol/dm}^3 \times 0.025 \text{ dm}^3 = 0.0125 \text{ mol}\).
Thus, the correct answer is A (0.0125).
What mass (g) of hydrogen chloride (HCl) is needed to make up 500 cm3 of a solution of concentration 0.2 mol/dm3? (Ar: H = 1, Cl = 35.5)
A. 7.30
B. 1.83
C. 14.6
D. 3.65
▶️ Answer/Explanation
Step 1: Convert volume to dm3
500 cm3 = 500 ÷ 1000 = 0.5 dm3
Step 2: Calculate moles
Moles = Concentration × Volume
= 0.2 × 0.5 = 0.1 mol
Step 3: Calculate molar mass of HCl
= 1 + 35.5 = 36.5 g/mol
Step 4: Calculate mass
Mass = Moles × Molar mass
= 0.1 × 36.5 = 3.65 g
Ans: D (3.65 g)
What is the molarity of the solution formed by dissolving 80 g of sodium hydroxide (NaOH) in 500 cm3 of water? (Ar: Na = 23, O = 16, H = 1)
A. 5.30
B. 4.83
C. 4.0
D. 3.65
▶️ Answer/Explanation
Step 1: Calculate molar mass of NaOH
= 23 + 16 + 1 = 40 g/mol
Step 2: Calculate moles
Moles = Mass ÷ Molar mass
= 80 ÷ 40 = 2 mol
Step 3: Convert volume to dm3
500 cm3 = 500 ÷ 1000 = 0.5 dm3
Step 4: Calculate molarity
Molarity = Moles ÷ Volume
= 2 ÷ 0.5 = 4.0 mol/dm3
Answer: 4.0 M
What is the molarity of the solution formed by dissolving 49 g of sulphuric acid (H2SO4) in 250 cm3 of water? (Ar: H = 1, S = 32, O = 16)
A. 1.30
B. 3.83
C. 2.0
D. 3.65
▶️ Answer/Explanation
Step 1: Calculate molar mass of H2SO4
= (2 × 1) + 32 + (4 × 16)
= 2 + 32 + 64 = 98 g/mol
Step 2: Calculate moles
Moles = Mass ÷ Molar mass
= 49 ÷ 98 = 0.5 mol
Step 3: Convert volume to dm3
250 cm3 = 250 ÷ 1000 = 0.25 dm3
Step 4: Calculate molarity
Molarity = Moles ÷ Volume
= 0.5 ÷ 0.25 = 2.0 mol/dm3
Answer: 2.0 M
How many grams of NaNO3 (molar mass = 85.0 g/mol) are needed to prepare 350 mL of a 0.477 M solution?
A. 11.5 g
B. 16.6 g
C. 14.20 g
D. 10.9 g
▶️ Answer/Explanation
Step 1: Convert volume to dm3
350 mL = 350 ÷ 1000 = 0.350 dm3
Step 2: Calculate moles
Moles = Molarity × Volume
= 0.477 × 0.350 = 0.167 mol (approx)
Step 3: Calculate mass
Mass = Moles × Molar mass
= 0.167 × 85.0 = 14.2 g (approx)
Answer: 14.2 g
How many grams of NaBr (molar mass = 102.9 g/mol) are needed to prepare 700 mL of a 0.230 M NaBr solution?
A. 11.5 g
B. 16.6 g
C. 7.00 g
D. 102.9 g
▶️ Answer/Explanation
Step 1: Convert volume to dm3
700 mL = 700 ÷ 1000 = 0.700 dm3
Step 2: Calculate moles
Moles = Molarity × Volume
= 0.230 × 0.700 = 0.161 mol
Step 3: Calculate mass
Mass = Moles × Molar mass
= 0.161 × 102.9 = 16.6 g (approx)
Ans: B (16.6 g)
What is the molarity of the solution formed by dissolving 80 g of sodium hydroxide (NaOH) in 500 cm3 of water? (Ar: Na = 23, O = 16, H = 1)
A. 2.0 mol/dm3
B. 4.0 mol/dm3
C. 0.25 mol/dm3
D. 1.0 mol/dm3
▶️ Answer/Explanation
Step 1: Calculate molar mass of NaOH
= 23 + 16 + 1 = 40 g/mol
Step 2: Calculate moles
Moles = 80 ÷ 40 = 2 mol
Step 3: Convert volume to dm3
500 cm3 = 0.5 dm3
Step 4: Calculate molarity
Molarity = 2 ÷ 0.5 = 4.0 mol/dm3
Ans: B (4.0 mol/dm3)
What is the molarity of the solution formed by dissolving 49 g of sulphuric acid (H2SO4) in 250 cm3 of water? (Ar: H = 1, S = 32, O = 16)
A. 0.50 mol/dm3
B. 1.0 mol/dm3
C. 2.0 mol/dm3
D. 4.0 mol/dm3
▶️ Answer/Explanation
Step 1: Calculate molar mass of H2SO4
= (2 × 1) + 32 + (4 × 16)
= 2 + 32 + 64 = 98 g/mol
Step 2: Calculate moles
Moles = 49 ÷ 98 = 0.5 mol
Step 3: Convert volume to dm3
250 cm3 = 0.25 dm3
Step 4: Calculate molarity
Molarity = 0.5 ÷ 0.25 = 2.0 mol/dm3
Ans: C (2.0 mol/dm3)
A volumetric pipette is used to measure 25.0 cm3 of 2.0 mol/dm3 aqueous sodium hydroxide into a conical flask.
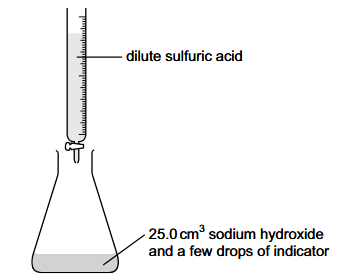
A burette is filled with dilute sulfuric acid.
The equation for the reaction is:
2NaOH + H2SO4 → Na2SO4 + 2H2O
The reaction requires 50.0 cm3 of dilute sulfuric acid to reach the end-point.
What is the concentration of the dilute sulfuric acid?
A. 0.50 mol/dm3
B. 1.0 mol/dm3
C. 2.0 mol/dm3
D. 4.0 mol/dm3
▶️ Answer/Explanation
Step 1: Calculate moles of NaOH
Volume = 25.0 cm3 = 0.025 dm3
Moles = 2.0 × 0.025 = 0.050 mol
Step 2: Use mole ratio
2NaOH : 1H2SO4
So moles of H2SO4 = 0.050 ÷ 2 = 0.025 mol
Step 3: Calculate concentration
Volume of acid = 50.0 cm3 = 0.050 dm3
Concentration = 0.025 ÷ 0.050 = 0.50 mol/dm3
Ans: A (0.50 mol/dm3)
The equation for the reaction between aqueous lead(II) nitrate and aqueous sodium chloride is:
Pb(NO3)2(aq) + 2NaCl(aq) → PbCl2(s) + 2NaNO3(aq)
If 100 cm3 of aqueous lead(II) nitrate of concentration 0.1 mol/dm3 reacts with excess sodium chloride, what mass of lead(II) chloride is obtained?
A. 1.16 g
B. 2.42 g
C. 2.78 g
D. 3.31 g
▶️ Answer/Explanation
Step 1: Calculate moles of Pb(NO3)2
100 cm3 = 0.100 dm3
Moles = 0.1 × 0.100 = 0.010 mol
Step 2: Mole ratio
1:1 ratio → moles of PbCl2 = 0.010 mol
Step 3: Molar mass of PbCl2
Pb = 207
Cl = 35.5 × 2 = 71
Total = 278 g/mol
Step 4: Calculate mass
Mass = 0.010 × 278 = 2.78 g
Ans: C (2.78 g)
