Pre AP Chemistry -4.2A Oxidation Numbers and Electron Transfer- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.2A Oxidation Numbers and Electron Transfer- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.2A Oxidation Numbers and Electron Transfer- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which statements about a redox reaction are correct?
- Oxidation is the gain of oxygen.
- Both oxidation and reduction take place in a redox reaction.
- Reduction is the gain of oxygen.
A) 1 and 2
B) 1 only
C) 2 and 3
D) 3 only
▶️ Answer/Explanation
Ans: A
Let’s evaluate each statement:
1. Correct – Oxidation can be defined as gain of oxygen (though it’s more accurately loss of electrons).
2. Correct – Redox reactions always involve both oxidation and reduction occurring simultaneously.
3. Incorrect – Reduction is actually the loss of oxygen (or gain of electrons/hydrogen).
Therefore, only statements 1 and 2 are correct, making option A the right answer.
Zinc is formed when zinc oxide is heated with carbon.
\[ \text{zinc oxide + carbon} \rightarrow \text{zinc + carbon monoxide} \]
Which substance is oxidised in this reaction?
A) carbon
B) carbon monoxide
C) zinc
D) zinc oxide
▶️ Answer/Explanation
Ans: A
Oxidation involves loss of electrons or gain of oxygen. Let’s analyze the reaction:
\[ \text{ZnO + C} \rightarrow \text{Zn + CO} \]
- Zinc oxide (ZnO) is reduced to Zn (loses oxygen)
- Carbon (C) is oxidized to CO (gains oxygen)
The oxidation numbers change as follows:
- Carbon goes from 0 (in C) to +2 (in CO) – oxidation
- Zinc remains +2 throughout (in ZnO and Zn, though elemental Zn is 0)
- Oxygen remains -2 throughout
Therefore, carbon is the substance that is oxidized in this reaction (Option A).
The equation for the reaction of magnesium with copper(II) oxide is shown.
\[ \text{Mg} + \text{CuO} \rightarrow \text{MgO} + \text{Cu} \]
Which word describes this reaction?
A) combustion
B) decomposition
C) neutralisation
D) redox
▶️ Answer/Explanation
Ans: D
This is a redox (reduction-oxidation) reaction because:
- Magnesium is oxidized (loses electrons, going from Mg to Mg²⁺ in MgO)
- Copper is reduced (gains electrons, going from Cu²⁺ in CuO to Cu)
It’s not:
- Combustion (no oxygen or burning involved)
- Decomposition (one compound isn’t breaking down into simpler substances)
- Neutralisation (no acid-base reaction occurring)
The transfer of oxygen from copper to magnesium and the change in oxidation states clearly indicate a redox reaction.
The equation for the reaction between hydrogen and oxygen is shown.
\[ 2H_2(g) + O_2(g) \rightarrow 2H_2O(g) \]
Which statement explains why this is a redox reaction?
A) Both oxidation and reduction take place.
B) Heat energy is released to the surroundings.
C) Hydrogen is a reactant.
D) The reaction can be reversed.
▶️ Answer/Explanation
Ans: A
A redox reaction involves both reduction and oxidation occurring simultaneously:
1. Oxidation: Hydrogen is oxidized as its oxidation state increases from 0 in H2 to +1 in H2O (loss of electrons).
2. Reduction: Oxygen is reduced as its oxidation state decreases from 0 in O2 to -2 in H2O (gain of electrons).
Option B describes an exothermic reaction (which this is, but doesn’t define it as redox). Option C is true but irrelevant to redox. Option D describes reversibility, not redox nature. Therefore, option A is correct as it identifies the simultaneous oxidation and reduction.
In a blast furnace, iron is extracted when iron(III) oxide reacts with carbon monoxide. The equation is shown.
\(Fe_2O_3 + 3CO → 2Fe + 3CO_2\)
Which substance is oxidised and which is reduced in this reaction?

▶️ Answer/Explanation
Ans: A
1. Redox Analysis:
• Oxidation: CO → CO₂ (carbon gains oxygen, oxidation state +2 → +4)
• Reduction: Fe₂O₃ → Fe (iron loses oxygen, oxidation state +3 → 0)
2. Half-Reactions:
Reduction: \(Fe_2O_3 + 6e^- → 2Fe + 3O^{2-}\)
Oxidation: \(3CO + 3O^{2-} → 3CO_2 + 6e^-\)
3. Why Option A:
• CO is the reducing agent (gets oxidized)
• Fe₂O₃ is the oxidizing agent (gets reduced)
• Other options misassign the redox roles
Thus, A correctly identifies the redox changes.
Which of the reactions show the underlined substance being reduced?
1 \(CuO + H_2 → Cu + H_2O\)
2 \(2Mg + O_2 → 2MgO\)
3 \(MgO + 2HCl → MgCl_2 + H_2O\)
A 1 and 2 B 1 only C 2 and 3 D 3 only
▶️ Answer/Explanation
Ans: B
Analysis of reduction in each reaction (reduction = gain of electrons, decrease in oxidation state):
- Reaction 1:
- Cu in CuO: +2 → 0 (reduction)
- H in H₂: 0 → +1 (oxidation)
- CuO is being reduced
- Reaction 2:
- O in O₂: 0 → -2 (reduction)
- Mg: 0 → +2 (oxidation)
- O₂ is reduced, but not underlined in question
- Reaction 3:
- No change in oxidation states (ionic reaction)
- Mg remains +2, O remains -2
- No reduction occurs
Since only Reaction 1 shows the underlined substance (CuO) being reduced, the correct answer is B (1 only).
Which equation represents an oxidation reaction?
A \(CaCO_3 → CaO + CO_2\)
B \(4FeO + O_2 → 2Fe_2O_3\)
C \(2NO_2 → N_2O_4\)
D \(2P_2O_5 → P_4O_{10}\)
▶️ Answer/Explanation
Ans: B
Analysis of oxidation states:
- A: Decomposition (no oxidation state change)
- B: Fe oxidation state increases from +2 to +3 (oxidation)
- C: Dimerization (no oxidation state change)
- D: Molecular combination (no oxidation state change)
Only option B shows an increase in oxidation state (Fe²⁺ → Fe³⁺), making it the correct choice for an oxidation reaction.
Silver oxide reacts with magnesium to make silver and magnesium oxide.
![]()
Which substance is oxidised in this reaction?
A) magnesium
B) magnesium oxide
C) silver
D) silver oxide
▶️ Answer/Explanation
Ans: A
Analyzing the redox reaction:
- Reaction equation:
2Ag₂O + 2Mg → 4Ag + 2MgO - Oxidation process:
- Magnesium (Mg → Mg²⁺ + 2e⁻) loses electrons
- Oxidation state increases from 0 to +2
- This is the definition of oxidation
- Reduction process:
- Silver oxide (Ag⁺ + e⁻ → Ag) gains electrons
- Oxidation state decreases from +1 to 0
- Why other options are incorrect:
- B: MgO is a product, not oxidised further
- C: Silver is reduced, not oxidised
- D: Silver oxide is being reduced
Key redox concepts:
- OXIDATION = loss of electrons (OIL RIG: Oxidation Is Loss)
- Magnesium acts as the reducing agent
- Silver oxide acts as the oxidizing agent
Therefore, magnesium (A) is the substance that gets oxidised in this reaction.
Which row describes the changes that occur when metals burn in oxygen?
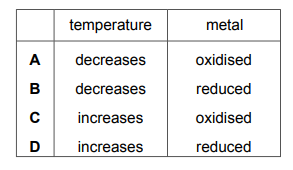
▶️ Answer/Explanation
Ans: C
When metals burn in oxygen:
1. They gain mass as oxygen atoms combine with metal atoms to form metal oxides.
2. The reaction is exothermic, releasing heat energy (shown by bright flames/sparks).
3. This matches row C’s description of “mass increases” and “energy is released”.
Other rows are incorrect as they either describe endothermic processes or decreasing mass.
The equations for two reactions are shown.
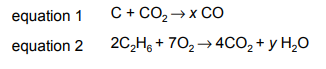
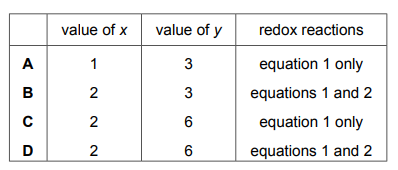
Which row shows the value of x, the value of y and the equations that are for redox reactions?
▶️ Answer/Explanation
Ans: D
Analysis of the reactions:
- First Reaction (NH4NO3 → N2O + 2H2O):
- Nitrogen changes from +5 (NO3–) to +1 (N2O)
- This is a redox reaction (x=1)
- Second Reaction (NH4Cl + NaOH → NH3 + NaCl + H2O):
- No change in oxidation states
- This is not a redox reaction (y=0)
Key points:
- Only the first reaction involves electron transfer (redox)
- Oxidation numbers confirm nitrogen is reduced in first reaction
- Second reaction is simple neutralization/displacement
Therefore, Option D (x=1, y=0, first equation only) is correct.
The equation for the reaction between magnesium and copper(II) oxide is shown.
\(Mg + CuO \rightarrow MgO + Cu\)
Which substance is oxidised?
- Cu
- CuO
- Mg
- MgO
▶️ Answer/Explanation
Ans: C
To determine which substance is oxidized:
- Oxidation involves an increase in oxidation state.
- In the reaction:
- Mg goes from 0 (elemental) to +2 in MgO → oxidized
- Cu goes from +2 in CuO to 0 (elemental) → reduced
- Oxygen’s oxidation state remains -2 throughout.
Thus, Mg is the substance that gets oxidized (loses electrons).
In which change is the sulfur, $\mathrm{S}$, in sulfur(I) oxide, $\mathrm{S}_2 \mathrm{O}$, reduced?

▶️ Answer/Explanation
Ans: D
Oxidation State Analysis:
- Initial State (S2O):
- Oxygen always has -2 oxidation state
- 2S + (-2) = 0 ⇒ S = +1
- Option D (S2O → S):
- Elemental sulfur (S) has oxidation state 0
- Change: +1 → 0 (reduction)
- Other Options:
- A: +1 → +4 (oxidation)
- B: +1 → +6 (oxidation)
- C: +1 → +2 (oxidation)
Reduction is defined as a decrease in oxidation state. Only Option D shows sulfur’s oxidation state decreasing from +1 to 0.
The equation for burning propane in air is shown.
\[ \mathrm{C}_3 \mathrm{H}_8(\mathrm{g}) + \mathrm{xO}_2(\mathrm{g}) \rightarrow 3 \mathrm{CO}_2(\mathrm{g}) + \mathrm{yH}_2 \mathrm{O}(\mathrm{g}) \]
Which values of \(x\) and \(y\) balance the equation?

▶️ Answer/Explanation
Ans: C
Balancing the combustion equation step-by-step:
- Balance carbon atoms:
- Propane (C₃H₈) has 3 carbons → 3 CO₂ already shown
- Carbon is balanced (3 on both sides)
- Balance hydrogen atoms:
- 8 H in C₃H₈ → Need 4 H₂O (since 4 × 2H = 8H)
- Thus, \(y = 4\)
- Balance oxygen atoms:
- Products have: (3 CO₂ × 2O) + (4 H₂O × 1O) = 10 oxygen atoms
- Reactants need: \(x\) O₂ molecules providing 2 oxygen atoms each
- Thus, \(2x = 10\) → \(x = 5\)
The balanced equation is:
\[ \mathrm{C}_3\mathrm{H}_8(\mathrm{g}) + 5\mathrm{O}_2(\mathrm{g}) \rightarrow 3\mathrm{CO}_2(\mathrm{g}) + 4\mathrm{H}_2\mathrm{O}(\mathrm{g}) \]
Therefore, the correct values are \(x = 5\) and \(y = 4\) (Option C).
Carbon reacts with silver oxide to form carbon dioxide and silver.
Which substance is reduced?
A carbon
B carbon dioxide
C silver
D silver oxide
▶️ Answer/Explanation
Ans: D
To determine which substance is reduced, let’s analyze the oxidation states:
Reaction: \( C + Ag_2O \rightarrow CO_2 + 2Ag \)
Oxidation State Changes:
- Carbon (C): 0 → +4 (oxidation)
- Silver in Ag₂O: +1 → 0 (reduction)
- Oxygen: Remains -2 throughout
Key Points:
- Reduction involves gain of electrons or decrease in oxidation state
- Silver changes from +1 to 0, showing reduction
- This reduction occurs in silver oxide (Ag₂O)
Why Other Options Are Incorrect:
- A: Carbon is oxidized (loses electrons)
- B: CO₂ is a product, not reduced
- C: Silver is the reduced form, but the substance reduced is silver oxide
Therefore, D (silver oxide) is the correct answer as it contains the silver that gets reduced.
Pentane is an alkane and pentene is an alkene.
What is observed when bromine water is added to a sample of each compound?
▶️ Answer/Explanation
Ans: C
With Pentane (alkane):
- No reaction occurs with bromine water
- The orange-brown color of bromine water remains unchanged
- Alkanes are saturated hydrocarbons and do not react with bromine under these conditions
With Pentene (alkene):
- The bromine water rapidly decolorizes (turns colorless)
- This occurs due to electrophilic addition across the C=C double bond:
\[ C_5H_{10} + Br_2 \rightarrow C_5H_{10}Br_2 \]
- The orange Br2 is consumed as it adds to the double bond
Key Observations:
- Pentane: Bromine water stays orange
- Pentene: Bromine water turns colorless
This matches option C in the table, where pentane shows no change and pentene shows decolorization.
