Pre AP Chemistry -4.2B Activity Series and Redox Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.2B Activity Series and Redox Reactions- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.2B Activity Series and Redox Reactions- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Four metals, P, Q, R and S, are added separately to water and to dilute hydrochloric acid.
The table shows the results.
| observation with water | observation with dilute hydrochloric acid | |
|---|---|---|
| P | no reaction | fizzes slowly |
| Q | fizzes rapidly | fizzes rapidly |
| R | no reaction | no reaction |
| S | fizzes slowly | fizzes rapidly |
Which conclusion can be made from these observations?
A) P is the least reactive of the four metals.
B) Q is more reactive than S.
C) Q is less reactive than P.
D) R is the most reactive of the four metals.
▶️ Answer/Explanation
Ans: B
From the observations, we can deduce the reactivity order:
1. Q reacts rapidly with both water and acid, indicating it’s the most reactive.
2. S reacts slowly with water but rapidly with acid, making it less reactive than Q but more reactive than P.
3. P only reacts with acid (and slowly), making it less reactive than both Q and S.
4. R doesn’t react with either, making it the least reactive.
Therefore, the correct conclusion is B: Q is more reactive than S. The other options are incorrect because:
A) P isn’t the least reactive (R is)
C) Q is more reactive than P
D) R is actually the least reactive
Four metals, W, X, Y and Z, are tested with either cold water, steam or both.
The observations are shown.
| metal | observations |
|---|---|
| W | reacts slowly with cold water |
| X | reacts rapidly with cold water |
| Y | does not react with cold water but reacts with steam |
| Z | does not react with cold water or steam |
What is the order of reactivity of the metals from the least reactive to the most reactive?

▶️ Answer/Explanation
Ans: D
The reactivity series of metals with water/steam is as follows (from most reactive to least):
- Metals that react rapidly with cold water (e.g., Group 1 metals like sodium)
- Metals that react slowly with cold water (e.g., calcium)
- Metals that react only with steam (e.g., magnesium, aluminum, zinc, iron)
- Metals that don’t react with water or steam (e.g., copper, silver, gold)
From the observations:
- X (reacts rapidly with cold water) is most reactive
- W (reacts slowly with cold water) is next
- Y (reacts only with steam) is less reactive than W
- Z (no reaction) is least reactive
Therefore, the correct order from least to most reactive is: Z → Y → W → X, which matches option D.
Silver metal is separately tested with cold water, with steam and with dilute hydrochloric acid.
Which row identifies the tests that show the chemical reactivity of silver?
| cold water | steam | dilute hydrochloric acid | |
|---|---|---|---|
| A | ✓ | ✓ | ✓ |
| B | ✗ | ✓ | ✓ |
| C | ✗ | ✗ | ✓ |
| D | ✗ | ✗ | ✗ |
| key: ✓ = reaction, ✗ = no reaction | |||
▶️ Answer/Explanation
Ans: D
Silver is a relatively unreactive metal in the reactivity series. It doesn’t react with:
1. Cold water – silver is below hydrogen in the reactivity series
2. Steam – same reason as cold water
3. Dilute hydrochloric acid – silver is below hydrogen so can’t displace it from acids
Therefore, silver shows no reaction in all three tests, which corresponds to option D.
Four different metals are separately mixed with an equal volume of dilute hydrochloric acid. The table shows the rate of effervescence for each metal.

What is the order of reactivity of the four metals starting with the most reactive?
A) iron → magnesium → calcium → copper
B) magnesium → calcium → copper → iron
C) copper → iron → magnesium → calcium
D) calcium → magnesium → iron → copper
▶️ Answer/Explanation
Ans: D
The rate of effervescence (gas production) indicates reactivity with hydrochloric acid. Higher reactivity means faster reaction and more vigorous effervescence. From the table:
1. Calcium shows the fastest effervescence (most reactive).
2. Magnesium is next in reactivity.
3. Iron reacts slower than magnesium.
4. Copper shows no effervescence (least reactive).
Thus, the correct order is D (calcium → magnesium → iron → copper).
Magnesium reacts with dilute hydrochloric acid. Which gas is given off in this reaction?
A) carbon dioxide
B) chlorine
C) hydrogen
D) oxygen
▶️ Answer/Explanation
Ans: C
The reaction between magnesium (Mg) and dilute hydrochloric acid (HCl) produces magnesium chloride (MgCl₂) and hydrogen gas (H₂). The balanced chemical equation is:
\[ \text{Mg} + 2\text{HCl} \rightarrow \text{MgCl}_2 + \text{H}_2 \uparrow \]
Since hydrogen gas (\(\text{H}_2\)) is the product, the correct answer is C. Carbon dioxide, chlorine, and oxygen are not formed in this reaction.
Three metals, L, M and N, are added separately to dilute hydrochloric acid and cold water. The results are shown.
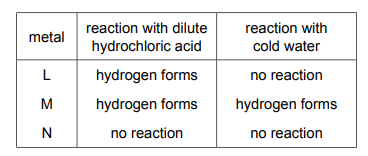
What is the order of reactivity of the metals? 
▶️ Answer/Explanation
Ans: C
From the table:
1. Metal L reacts vigorously with both HCl and cold water, indicating high reactivity.
2. Metal M reacts slowly with HCl but not with cold water, placing it below L in reactivity.
3. Metal N shows no reaction with either HCl or cold water, making it the least reactive.
Thus, the correct order of reactivity is L > M > N, which corresponds to option C.
The table shows the results of separately heating four metals with oxides of different metals. The four metals are iron, copper, magnesium and Y. The results are shown.
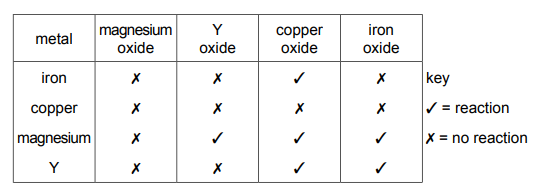
What is the order of reactivity of the metals, least reactive first? 
▶️ Answer/Explanation
Ans: A
From the table: 1. Copper doesn’t displace any metal (least reactive). 2. Iron displaces only copper. 3. Metal Y displaces both iron and copper. 4. Magnesium displaces all three (most reactive). The correct order from least to most reactive is: Copper → Iron → Y → Magnesium.
A metal reacts vigorously with cold water. Which statement about the metal is correct?
A. It is above hydrogen in the reactivity series.
B. It is below magnesium in the reactivity series.
C. Its oxide can be reduced with carbon.
D. It does not react with dilute acids.
▶️ Answer/Explanation
Ans: A
A metal that reacts vigorously with cold water must be highly reactive. In the reactivity series, such metals (e.g., sodium, potassium) are placed above hydrogen, as they can displace hydrogen from water. Metals below hydrogen (e.g., copper, silver) do not react with water. Since the metal reacts with water, it must also react with dilute acids, ruling out option D. The reactivity with water alone does not provide information about its position relative to magnesium or reducibility of its oxide.
A reactivity series is shown.
sodium
calcium
magnesium
carbon
zinc
iron
hydrogen
copper
Which statement is correct?
A All the metals above carbon are extracted by electrolysis.
B Iron can only be extracted by electrolysis.
C Calcium can be extracted by heating calcium oxide with carbon.
D Copper can only be extracted by passing hydrogen over heated copper(II) oxide.
▶️ Answer/Explanation
Ans: A
In the reactivity series, metals above carbon (sodium, calcium, magnesium) are highly reactive and cannot be reduced by carbon. Thus, they are extracted via electrolysis of their molten compounds. Iron (B) is extracted by reduction with carbon, not electrolysis. Calcium (C) cannot be extracted using carbon due to its high reactivity. Copper (D) can be extracted by heating its oxide with carbon or by reduction with hydrogen, not exclusively the latter.
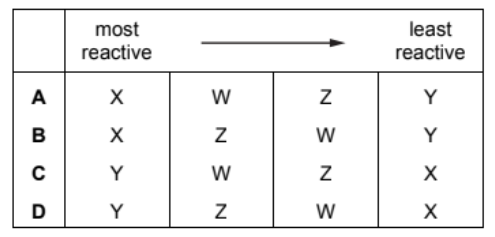
The properties of four metals, $\mathrm{W}, \mathrm{X}, \mathrm{Y}$ and $\mathrm{Z}$, are shown.
W It does not react with cold water but reacts with steam.
X It does not react with water or dilute acid but the oxide of X is reduced by carbon.
Y The oxide of Y is not reduced by carbon but Y reacts vigorously with cold water.
Z It does not react with water or steam but reacts with dilute acid.
What is the order of reactivity of the elements starting with the most reactive?
▶️ Answer/Explanation
Ans: C
The reactivity order is determined based on the reactions of the metals:
- Y is most reactive as it reacts vigorously with cold water.
- W is next, reacting only with steam (less reactive than Y).
- Z follows, reacting only with dilute acid (less reactive than W).
- X is least reactive, not reacting with water or acid, and its oxide is reduced by carbon.
Thus, the correct order is Y > W > Z > X.
The metal beryllium does not react with cold water.
It reacts with hydrochloric acid but cannot be extracted from its ore by using carbon.
Where is beryllium placed in the reactivity series?
A) magnesium
B) zinc
C) iron
D) copper
▶️ Answer/Explanation
Ans: A
Beryllium reacts with hydrochloric acid, indicating it is more reactive than metals like copper and iron. However, since it cannot be extracted using carbon, it must be placed above zinc but below magnesium in the reactivity series. Thus, the correct position is above zinc (A: magnesium).
Manganese, nickel and silver are all metals.
Samples of powdered manganese, nickel and silver were placed in separate test-tubes containing dilute hydrochloric acid.
The results are shown.
What is the order of reactivity of the metals, most reactive to least reactive?
A) manganese → nickel → silver
B) manganese → silver → nickel
C) silver → manganese → nickel
D) silver → nickel → manganese
▶️ Answer/Explanation
Ans: A
The reactivity of metals with dilute hydrochloric acid determines their order of reactivity. Manganese (Mn) reacts vigorously, indicating high reactivity. Nickel (Ni) reacts but less vigorously, placing it below manganese. Silver (Ag) does not react, making it the least reactive. Thus, the correct order is Manganese → Nickel → Silver.
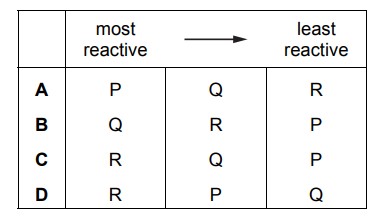
The reactions of three metals, P, Q and R, are shown.
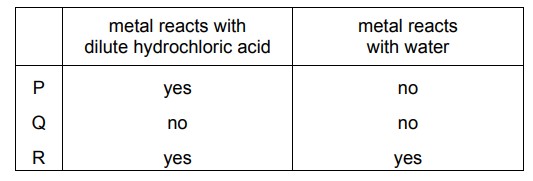
What is the order of reactivity of the metals?
▶️ Answer/Explanation
Ans: D
The reactivity order is determined by observing which metals react with water and/or acid. Metal R is most reactive as it reacts with both water and HCl. Metal Q is least reactive since it doesn’t react with either. Metal P shows intermediate reactivity by reacting only with HCl. Thus, the correct order is R > P > Q, which corresponds to option D.
Four different metals are reacted with an equal volume of dilute hydrochloric acid. The results of the reactions are shown.
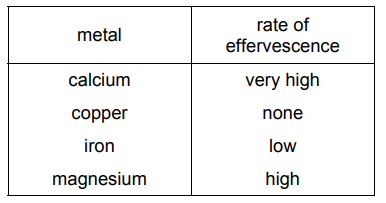
What is the order of reactivity of the four metals starting with the most reactive?
- iron → magnesium → calcium → copper
- magnesium → calcium → copper → iron
- copper → iron → magnesium → calcium
- calcium → magnesium → iron → copper
▶️ Answer/Explanation
Ans: D
The reactivity of metals with acids is determined by the amount of effervescence (gas bubbles) produced. From the image:
- Calcium (Ca) shows the most vigorous reaction → most reactive.
- Magnesium (Mg) reacts strongly but less than calcium.
- Iron (Fe) shows a slower reaction.
- Copper (Cu) does not react → least reactive.
Thus, the correct order is D: calcium → magnesium → iron → copper.
Below are some metals in decreasing order of reactivity.
magnesium
zinc
iron
copper
Titanium reacts with acid and cannot be extracted from its ore by heating with carbon.
Where should titanium be placed in this list?
- below copper
- between iron and copper
- between magnesium and zinc
- between zinc and iron
▶️ Answer/Explanation
Ans: B
Titanium reacts with acid, meaning it is more reactive than copper (which does not react with acid). However, it cannot be extracted using carbon reduction, unlike magnesium, zinc, and iron, which can. This places titanium below iron but above copper in reactivity.
The correct order is:
- Magnesium (most reactive)
- Zinc
- Iron
- Titanium
- Copper (least reactive)
Thus, titanium fits between iron and copper, corresponding to option B.
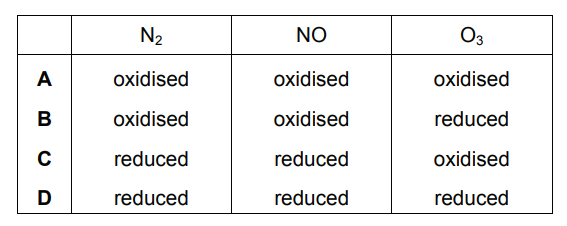
The reactions shown may occur in the air during a thunder-storm.
N2 + O2 → 2NO
2NO + O2 → 2NO2
NO + O3 → NO2 + O2
Which row shows what happens to the reactant molecules in each of these reactions?
▶️ Answer/Explanation
Ans: B
1. Reaction Analysis:
• N2 + O2 → 2NO:
– N (0 → +2): Oxidized
– O (0 → -2): Reduced
• 2NO + O2 → 2NO2:
– N (+2 → +4): Oxidized
– O (0 → -2): Reduced
• NO + O3 → NO2 + O2:
– N (+2 → +4): Oxidized
– O3 (0 → 0, but loses O atom): Reduced
2. Key Observations:
• Nitrogen is progressively oxidized in all reactions
• Oxygen-containing compounds are reduced
• O3 acts as an oxidizing agent (itself gets reduced)
Thus, B correctly identifies all redox changes.
Iron is extracted from iron oxide using carbon monoxide as shown.
iron oxide + carbon monoxide → iron + carbon dioxide
Which statement is correct?
A Carbon monoxide is oxidised to carbon dioxide.
B Carbon monoxide is reduced to carbon dioxide.
C Iron is oxidised to iron oxide.
D Iron oxide is oxidised to iron.
▶️ Answer/Explanation
Ans: A
This is the blast furnace reaction for iron extraction. Let’s analyze the redox changes:
- Carbon monoxide:
- Oxidation state of C in CO: +2
- Oxidation state of C in CO2: +4
- Change: +2 → +4 (oxidation, loses electrons)
- Iron oxide:
- Fe in Fe2O3: +3
- Fe in product: 0
- Change: +3 → 0 (reduction, gains electrons)
Key points:
- CO acts as the reducing agent (gets oxidized)
- Fe2O3 is the oxidizing agent (gets reduced)
- Option A is correct as it describes the oxidation of CO to CO2
The balanced equation is: Fe2O3 + 3CO → 2Fe + 3CO2
Aluminium reacts with iron(III) oxide as shown.
iron(III) oxide + aluminium → iron + aluminium oxide
Which statement about this reaction is correct?
A Aluminium is oxidised.
B Aluminium oxide is reduced.
C Iron(III) oxide is oxidised.
D Iron is oxidised.
▶️ Answer/Explanation
Ans: A
Balanced equation: Fe2O3 + 2Al → 2Fe + Al2O3
Oxidation state analysis:
- Aluminium: 0 → +3 (oxidation)
- Iron: +3 → 0 (reduction)
- Oxygen: -2 (no change)
Key points:
– Aluminium loses electrons (oxidized)
– Iron(III) gains electrons (reduced)
– This is a classic thermite reaction
Therefore, only statement A is correct.
The element vanadium, V, forms several oxides.
In which change is oxidation taking place?
A) \(VO_2 → V_2O_3\)
B) \(V_2O_5 → VO_2\)
C) \(V_2O_3 → VO\)
D) \(V_2O_3 → V_2O_5\)
▶️ Answer/Explanation
Ans: D
To determine where oxidation occurs, we analyze the vanadium oxidation states:
| Option | Change | Oxidation State Change | Process |
|---|---|---|---|
| A | \(VO_2 → V_2O_3\) | +4 → +3 | Reduction |
| B | \(V_2O_5 → VO_2\) | +5 → +4 | Reduction |
| C | \(V_2O_3 → VO\) | +3 → +2 | Reduction |
| D | \(V_2O_3 → V_2O_5\) | +3 → +5 | Oxidation |
Key points about option D:
- Vanadium’s oxidation state increases from +3 to +5
- This represents a loss of electrons (oxidation)
- The change requires an oxidizing agent
- More oxygen atoms are incorporated into the compound
Therefore, D (\(V_2O_3 → V_2O_5\)) is the correct answer as it’s the only option where vanadium undergoes oxidation.
In which change is chromium(II) oxide, CrO, reduced?
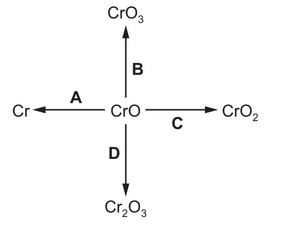
▶️ Answer/Explanation
Ans: A
1. Oxidation state analysis:
– In CrO: Cr has +2 oxidation state
– In Cr metal: oxidation state is 0
2. Reduction occurs when:
– Cr2+ gains electrons to become Cr0 (Option A)
3. Other options show:
– B: Cr2+ → Cr3+ (oxidation)
– C: Cr2+ → Cr6+ (oxidation)
– D: No change in Cr oxidation state
Which equation shows an oxidation reaction?
- C + O2 → CO2
- CaCO3 → CaO + CO2
- CaO + 2HCl → CaCl2 + H2O
- N2O4 → 2NO2
▶️ Answer/Explanation
Ans: A
Analysis of oxidation states:
- C + O2 → CO2:
- C: 0 → +4 (oxidation)
- O: 0 → -2 (reduction)
- This is a redox reaction with clear oxidation
- Other options:
- B: Decomposition (no oxidation state changes)
- C: Neutralization (no redox)
- D: Decomposition (N remains +4)
Key points:
- Only option A shows an increase in oxidation state (carbon from 0 to +4)
- This meets the definition of oxidation (loss of electrons)
- Other reactions involve no electron transfer
Therefore, Option A is the correct oxidation reaction.
The equations for two reactions P and Q are given.
P: 2NaNO2 + O2 → 2NaNO3
Q: 2HgO → 2Hg + O2
In which of these reactions does oxidation of the underlined substance occur?
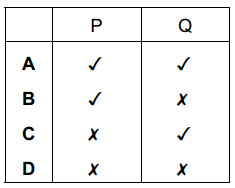
▶️ Answer/Explanation
Ans: B
To determine oxidation of the underlined substances:
- Reaction P (NaNO2):
- Nitrogen’s oxidation state in NaNO2: +3
- In NaNO3: +5
- Change: +3 → +5 (oxidation of nitrogen)
- Reaction Q (HgO):
- Mercury’s oxidation state in HgO: +2
- In Hg: 0
- Change: +2 → 0 (reduction of mercury)
Thus, only Reaction P shows oxidation of the underlined substance, corresponding to option B in the answer choices.
The equations represent redox reactions.
In which equation is the underlined substance acting as a reducing agent?
- 3CO + Fe2O3 → 2Fe + 3CO2
- CO2 + C → 2CO
- CuO + H2 → Cu + H2O
- CaO + H2O → Ca(OH)2
▶️ Answer/Explanation
Ans: A
Reducing Agent Analysis:
- Option A (3CO + Fe2O3 → 2Fe + 3CO2):
- CO is oxidized (C: +2 → +4)
- Fe2O3 is reduced (Fe: +3 → 0)
- CO acts as reducing agent by donating electrons
- Other Options:
- B: CO2 is reduced (C: +4 → +2), not acting as reducing agent
- C: CuO is reduced (Cu: +2 → 0), acting as oxidizing agent
- D: No redox occurs (acid-base reaction)
A reducing agent must get oxidized itself while causing reduction of another species. Only in Option A does the underlined substance (CO) show this behavior by:
- Increasing its oxidation state (+2 to +4)
- Causing reduction of iron oxide
The equations represent redox reactions.
In which equation is the underlined substance acting as a reducing agent?
- 3CO + Fe2O3 → 2Fe + 3CO2
- CO2 + C → 2CO
- CuO + H2 → Cu + H2O
- CaO + H2O → Ca(OH)2
▶️ Answer/Explanation
Ans: A
Analysis of each option for reducing agent behavior (substance that gets oxidized):
- 3CO + Fe2O3 → 2Fe + 3CO2
- CO (C2+) → CO2 (C4+): Oxidation (loses electrons)
- Fe2O3 (Fe3+) → Fe (0): Reduction
- CO is the reducing agent (correct answer)
- CO2 + C → 2CO
- CO2 gets reduced (C4+ → C2+)
- C acts as reducing agent (not underlined)
- CuO + H2 → Cu + H2O
- CuO gets reduced (Cu2+ → Cu0)
- H2 acts as reducing agent (not underlined)
- CaO + H2O → Ca(OH)2
- Not a redox reaction (no oxidation state changes)
Key characteristics of a reducing agent:
- Gets oxidized (loses electrons)
- Causes another substance to be reduced
- Increases its oxidation state
Only in Option A does the underlined substance (CO) meet all these criteria.
The equation shows the formation of anhydrous copper(II) sulfate from hydrated copper(II) sulfate.
\(CuSO_{4}.5H_{2}O\rightleftharpoons CuSO_{4}+5H_{2}O\)
Statements 1, 2 and 3 refer to this reaction.
- Hydrated copper(II) sulfate is reduced to anhydrous copper(II) sulfate.
- The (II) in the name copper(II) sulfate refers to the oxidation state of the metal.
- The reaction is reversible.
Which statements are correct?
A 1, 2 and 3 B 1 and 2 C 2 and 3 D 3 only
▶️ Answer/Explanation
Ans: C (2 and 3)
Let’s analyze each statement:
Statement 1: “Hydrated copper(II) sulfate is reduced to anhydrous copper(II) sulfate.”
- This is incorrect – the reaction is a dehydration (loss of water molecules), not a reduction
- The copper oxidation state remains +2 throughout
Statement 2: “The (II) in the name copper(II) sulfate refers to the oxidation state of the metal.”
- This is correct – the Roman numeral indicates Cu’s +2 oxidation state
Statement 3: “The reaction is reversible.”
- This is correct – the double arrow indicates reversibility
- Anhydrous form can reabsorb water to reform hydrated crystals
Key Points:
- Only statements 2 and 3 are correct
- Statement 1 is wrong because no redox occurs (Cu remains +2)
- The reaction involves water loss/gain, not electron transfer
Therefore, the correct answer is C (2 and 3).
Equations P and Q represent two reactions which occur inside a blast furnace.
P Fe2O3 + 3CO → 2Fe + 3CO2
Q CaCO3 → CaO + CO2
Which type of reactions are P and Q?

▶️ Answer/Explanation
Ans: B
Analysis of Reaction P:
- Iron is reduced: Fe3+ (in Fe2O3) → Fe0
- Carbon is oxidized: C2+ (in CO) → C4+ (in CO2)
- This simultaneous oxidation and reduction makes it a redox reaction
- Key industrial process for iron extraction
Analysis of Reaction Q:
- All elements maintain their oxidation states:
- Ca: +2 throughout
- C: +4 throughout
- O: -2 throughout
- This is simply a thermal decomposition reaction
- Common limestone decomposition in blast furnaces
Why Option B?
- P shows redox characteristics (electron transfer)
- Q shows non-redox decomposition
- Other options either misclassify P or Q
Summary Table
| Reaction | Type | Evidence |
|---|---|---|
| P | Redox | Fe reduced, C oxidized |
| Q | Thermal Decomposition | No oxidation state changes |
Therefore, the correct classification is B (redox and thermal decomposition).
Which change is an oxidation?
A FeO to Fe2O3
B Fe2O3 to FeO
C H2O2 to H2O
D H2O to H2
▶️ Answer/Explanation
Ans: A
Oxidation Analysis:
Oxidation is defined as an increase in oxidation state (loss of electrons). Let’s examine each option:
| Option | Oxidation State Change | Conclusion |
|---|---|---|
| A | Fe: +2 → +3 | Oxidation (state increases) |
| B | Fe: +3 → +2 | Reduction |
| C | O: -1 → -2 | Reduction |
| D | H: +1 → 0 | Reduction |
Detailed Explanation for Option A:
- In FeO: Fe has +2 oxidation state (Fe2+)
- In Fe2O3: Each Fe has +3 state (Fe3+)
- The change Fe2+ → Fe3+ involves:
- Loss of 1 electron per Fe ion
- Increase in oxidation number from +2 to +3
- Half-reaction: Fe2+ → Fe3+ + e–
Why Other Options Are Not Oxidation:
- B: Oxidation state decreases (+3 → +2) → reduction
- C: Oxygen’s state decreases (-1 → -2) → reduction
- D: Hydrogen’s state decreases (+1 → 0) → reduction
Thus, only Option A represents a true oxidation process.
