Pre AP Chemistry -4.2C Representations of Redox Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.2C Representations of Redox Reactions- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.2C Representations of Redox Reactions- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Pentane is an alkane and pentene is an alkene.
What is observed when bromine water is added to a sample of each compound?
▶️ Answer/Explanation
Ans: C
With Pentane (alkane):
- No reaction occurs with bromine water
- The orange-brown color of bromine water remains unchanged
- Alkanes are saturated hydrocarbons and do not react with bromine under these conditions
With Pentene (alkene):
- The bromine water rapidly decolorizes (turns colorless)
- This occurs due to electrophilic addition across the C=C double bond:
\[ C_5H_{10} + Br_2 \rightarrow C_5H_{10}Br_2 \]
- The orange Br2 is consumed as it adds to the double bond
Key Observations:
- Pentane: Bromine water stays orange
- Pentene: Bromine water turns colorless
This matches option C in the table, where pentane shows no change and pentene shows decolorization.
Carbon reacts with carbon dioxide as shown.
\(CO_2 + C → 2CO\)
Which statement about this reaction is correct?
A) Carbon dioxide and carbon are both oxidised.
B) Carbon dioxide and carbon are both reduced.
C) Carbon dioxide is reduced and carbon is oxidised.
D) Carbon dioxide is oxidised and carbon is reduced.
▶️ Answer/Explanation
Ans: C
1. Oxidation State Analysis:
• In CO₂: C = +4 (each O = -2)
• In C: Oxidation state = 0 (elemental form)
• In CO: C = +2 (reduced from +4)
2. Redox Changes:
• CO₂ (+4 → +2): Reduction (gain of electrons)
• C (0 → +2): Oxidation (loss of electrons)
3. Why Option C:
• The reaction represents a disproportionation where:
– CO₂ is the oxidizing agent (gets reduced)
– C is the reducing agent (gets oxidized)
• Other options incorrectly pair the redox processes
Thus, C is the correct interpretation.
A violent reaction occurs when a mixture of chromium(III) oxide and aluminium is ignited with a magnesium fuse as shown.

Which substance is oxidised in the reaction?
A aluminium
B aluminium oxide
C chromium
D chromium(III) oxide
▶️ Answer/Explanation
Ans: A
This is an example of the thermite reaction. The balanced equation is:
Cr2O3 + 2Al → 2Cr + Al2O3
Oxidation state analysis:
- Aluminium: Changes from 0 (in Al) to +3 (in Al2O3) → oxidized (loses electrons)
- Chromium: Changes from +3 (in Cr2O3) to 0 (in Cr) → reduced (gains electrons)
Key points:
- Aluminium is the reducing agent (gets oxidized)
- Chromium(III) oxide is the oxidizing agent (gets reduced)
- Aluminium oxide and chromium are products, not reactants
Therefore, only aluminium (option A) is oxidized in this reaction.
In a blast furnace, iron is extracted when iron(III) oxide reacts with carbon monoxide. The equation is shown.
\(Fe_{2}O_{3}+3CO\rightarrow 2Fe+3CO_{2}\)
Which substance is oxidised and which is reduced?
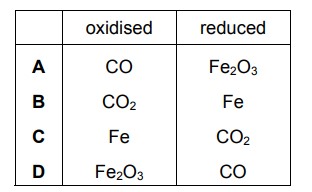
▶️ Answer/Explanation
Ans: A
Oxidation state analysis:
- Iron(III) oxide (Fe₂O₃):
- Fe: +3 → 0 (reduction, gain of electrons)
- O: -2 (no change)
- Carbon monoxide (CO):
- C: +2 → +4 (oxidation, loss of electrons)
- O: -2 (no change)
Therefore:
– CO is oxidized (carbon loses electrons)
– Fe₂O₃ is reduced (iron gains electrons)
This matches option A in the image.
Mercury(II) oxide, HgO, decomposes when heated.
The equation is shown.
2HgO → 2Hg + O2
Why is this a reduction reaction?
- The products weigh less than the reactants.
- There are fewer reactants than products.
- There is a gain of oxygen.
- There is a loss of oxygen.
▶️ Answer/Explanation
Ans: D
Understanding the redox aspects of this decomposition:
- Redox analysis:
- Mercury: Changes from Hg²⁺ (in HgO) to Hg⁰ (elemental) – reduction
- Oxygen: Changes from O²⁻ (in HgO) to O⁰ (in O2) – oxidation
- Oxygen perspective:
- HgO loses oxygen atoms (forming O2 gas)
- This matches the traditional definition of reduction as “loss of oxygen”
- Why other options are incorrect:
- A: Mass conservation means total mass remains equal
- B: 2HgO (2 particles) → 2Hg + 1O2 (3 particles) but irrelevant to redox
- C: Opposite of what occurs (oxygen is lost, not gained)
Key redox concepts:
- REDUCTION can be defined as:
- Gain of electrons
- Decrease in oxidation state
- Loss of oxygen
- Here, mercury undergoes reduction by all three definitions
Therefore, D (loss of oxygen) correctly explains why this is a reduction reaction.
The reaction between magnesium and carbon dioxide is shown in the equation.
2Mg + CO2 → 2MgO + C
Which statement describes what happens in this reaction?
- Carbon is oxidised.
- Magnesium is reduced.
- Neither oxidation nor reduction happens.
- The carbon in carbon dioxide is reduced.
▶️ Answer/Explanation
Ans: D
1. Oxidation states:
– Mg: 0 → +2 (oxidation)
– C in CO2: +4 → 0 (reduction)
2. Analysis of options:
– A: Incorrect (carbon gains electrons)
– B: Incorrect (Mg loses electrons)
– C: Incorrect (redox occurs)
– D: Correct (carbon’s oxidation state decreases from +4 to 0)
Tin is formed when tin(II) oxide is heated with carbon.
What happens to the tin in the tin(II) oxide in this reaction?
A It is burnt.
B It is electrolysed.
C It is oxidised.
D It is reduced.
▶️ Answer/Explanation
Ans: D
The reaction between tin(II) oxide and carbon can be represented as:
SnO(s) + C(s) → Sn(s) + CO(g)
Key observations:
- Oxidation state changes:
- Tin changes from +2 (in SnO) to 0 (in Sn) → reduction
- Carbon changes from 0 (in C) to +2 (in CO) → oxidation
- Process identification:
- Reduction: Gain of electrons (tin’s oxidation number decreases)
- Carbon acts as the reducing agent
Therefore, the tin in tin(II) oxide is reduced (Option D).
Which equation shows reduction of an iron compound?
- \(4Fe + 3O_2 \rightarrow 2Fe_2O_3\)
- \(Fe + 2HCl \rightarrow FeCl_2 + H_2\)
- \(4FeO + O_2 \rightarrow 2Fe_2O_3\)
- \(Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2\)
▶️ Answer/Explanation
Ans: D
To identify reduction of an iron compound:
- Reduction involves a decrease in oxidation state (gain of electrons).
- Analyzing each option:
- A: Fe (0) → Fe³⁺ (oxidation)
- B: Fe (0) → Fe²⁺ (oxidation)
- C: Fe²⁺ → Fe³⁺ (oxidation)
- D: Fe³⁺ → Fe (0) (reduction)
Only option D shows iron’s oxidation state decreasing from +3 to 0, confirming reduction of the iron compound.
Which equation shows an oxidation reaction?
- C + O2 → CO2
- CaCO3 → CaO + CO2
- CaO + 2HCl → CaCl2 + H2O
- N2O4 → 2NO2
▶️ Answer/Explanation
Ans: A
Oxidation Analysis:
- Option A (C + O2 → CO2):
- Carbon’s oxidation state changes from 0 to +4
- Clear loss of electrons (oxidation)
- Oxygen is the oxidizing agent
- Other Options:
- B: Decomposition (no oxidation state changes)
- C: Neutralization (no redox)
- D: Decomposition (N remains +4 in both compounds)
Only Option A shows a clear oxidation reaction where carbon loses electrons to oxygen. The oxidation state change from 0 to +4 confirms this is an oxidation process.
Calcium phosphate forms when calcium chloride and sodium phosphate solutions react together.
\[ xCaCl_{2} + yNa_{3}PO_{4} \rightarrow 2Ca_{3}(PO_{4})_{2} + 12NaCl \]
Which values of x and y balance the equation?
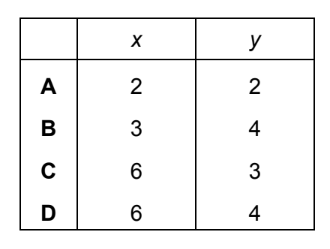
▶️ Answer/Explanation
Ans: D
Balancing the equation step-by-step:
- Balance calcium atoms:
- Products have: 2 × Ca₃(PO₄)₂ → 6 calcium atoms
- Each CaCl₂ provides 1 calcium → x = 6
- Balance sodium atoms:
- Products have: 12 NaCl → 12 sodium atoms
- Each Na₃PO₄ provides 3 sodiums → 3y = 12 → y = 4
- Verify phosphate balance:
- Products have: 2 × (PO₄)₂ → 4 phosphate groups
- Reactants have: y = 4 Na₃PO₄ → 4 phosphate groups
- Verify chlorine balance:
- Reactants: 6 CaCl₂ → 12 chlorine atoms
- Products: 12 NaCl → 12 chlorine atoms
The balanced equation is:
\[ 6CaCl_{2} + 4Na_{3}PO_{4} \rightarrow 2Ca_{3}(PO_{4})_{2} + 12NaCl \]
Therefore, the correct values are x = 6 and y = 4 (Option D).
In which equation does oxidation of the underlined substance occur?
- 2CuO + C → CO2 + 2Cu
- Fe2O3 + 3CO → 2Fe + 3CO2
- 2Mg + O2 → 2MgO
- MnO2 + 4HCl → MnCl2 + 2H2O + Cl2
▶️ Answer/Explanation
Ans: B
To identify oxidation, we examine changes in oxidation states of the underlined substances:
Option A: 2CuO + C → CO2 + 2Cu
- Cu changes from +2 (in CuO) to 0 (in Cu) → reduction
Option B: Fe2O3 + 3CO → 2Fe + 3CO2
- C in CO changes from +2 to +4 (in CO2) → oxidation
- This is the correct answer
Option C: 2Mg + O2 → 2MgO
- Mg changes from 0 to +2 → oxidation occurs, but Mg is not underlined
- Underlined MgO is the product, not the substance being oxidized
Option D: MnO2 + 4HCl → MnCl2 + 2H2O + Cl2
- Mn changes from +4 to +2 → reduction
Key Points:
- Oxidation involves increase in oxidation state
- Only Option B shows the underlined substance (CO) being oxidized
- The carbon in CO loses electrons (oxidation state +2 → +4)
Therefore, B is the correct answer as it’s the only option where the underlined substance undergoes oxidation.
In which reaction is the first substance in the equation oxidised?
A CaO + H2O → Ca(OH)2
B 4FeO + O2 → 2Fe2O3
C SnO2 + 2H2 → Sn + 2H2O
D ZnCO3 → ZnO + CO2
▶️ Answer/Explanation
Ans: B
Analysis of each option:
| Option | Oxidation State Change | Explanation |
|---|---|---|
| A | Ca: +2 → +2 | No oxidation state change (not oxidized) |
| B | Fe: +2 → +3 | Iron’s oxidation state increases (oxidized) |
| C | Sn: +4 → 0 | Tin is reduced (oxidation state decreases) |
| D | Zn: +2 → +2 | No oxidation state change (thermal decomposition) |
Key points for Option B:
- Iron’s oxidation state increases from +2 in FeO to +3 in Fe2O3
- This meets the definition of oxidation (loss of electrons/increase in oxidation state)
- The oxygen (O2) acts as the oxidizing agent
- Half-equation for oxidation: 4Fe2+ → 4Fe3+ + 4e–
Therefore, the correct answer is B, as it’s the only reaction where the first substance undergoes oxidation.
