Pre AP Chemistry -4.3A Strength of Acids and Bases- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.3A Strength of Acids and Bases- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.3A Strength of Acids and Bases- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
What is not a typical characteristic of acids?
- They react with alkalis producing water.
- They react with all metals producing hydrogen.
- They react with carbonates producing carbon dioxide.
- They turn blue litmus paper red.
▶️ Answer/Explanation
Ans: B
Analysis of acid characteristics:
- Reaction with alkalis (A):
- True: Acids + bases → salt + water (neutralization)
- Reaction with metals (B):
- Not always true: Only reactive metals (above H in reactivity series) react
- Noble metals (Au, Pt) don’t react with most acids
- This is the incorrect statement
- Reaction with carbonates (C):
- True: Acid + carbonate → salt + water + CO2
- Litmus test (D):
- True: Acids turn blue litmus red
Key points:
- Option B is the exception – acids don’t react with all metals
- This makes it the correct answer as it’s not a universal characteristic
Therefore, Option B is not a typical characteristic of acids.
Which statements are properties of an acid?
1 reacts with ammonium sulfate to form ammonia
2 turns red litmus blue
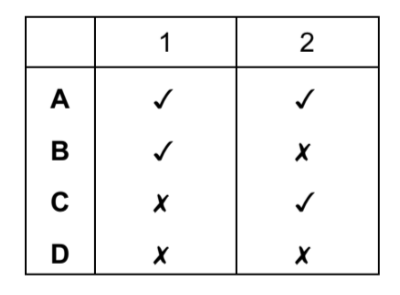
▶️ Answer/Explanation
Ans: D
Analysis of the statements:
| Statement | Property of | Explanation |
|---|---|---|
| 1 | Base | Only bases react with ammonium salts to release NH3 |
| 2 | Base | Bases turn red litmus blue (acids turn blue litmus red) |
Key chemical concepts:
- Ammonia formation:
- Base + NH4+ → NH3 + H2O
- Acids cannot produce ammonia from ammonium salts
- Litmus test:
- Acids: turn blue litmus red
- Bases: turn red litmus blue
- The statements describe base properties, not acid properties
Therefore, neither statement describes acid properties, making D the correct answer.
Which property is not characteristic of a base?
- It reacts with a carbonate to form carbon dioxide.
- It reacts with an acid to form a salt.
- It reacts with an ammonium salt to form ammonia.
- It turns universal indicator paper blue.
▶️ Answer/Explanation
Ans: A
Analysis of base characteristics:
- Reaction with carbonates (A):
- This is actually a property of acids, not bases
- Acid + Carbonate → Salt + Water + CO2
- Bases do not typically react with carbonates to produce CO2
- Reaction with acids (B):
- True: Base + Acid → Salt + Water (neutralization)
- Reaction with ammonium salts (C):
- True: Base + NH4+ → NH3 + H2O + salt
- Indicator test (D):
- True: Bases turn indicators blue/purple
Key points:
- Option A describes an acid’s behavior, not a base’s
- This makes it the correct answer as it’s not characteristic of bases
Therefore, Option A is not a characteristic property of bases.
Which statement about dilute hydrochloric acid is correct?
A) It is a strong acid as it fully dissociates.
B) It is a strong acid as it partially dissociates.
C) It is a weak acid as it fully dissociates.
D) It is a weak acid as it partially dissociates.
▶️ Answer/Explanation
Ans: A
Hydrochloric acid is a strong acid, meaning it completely (fully) dissociates in aqueous solution to form H⁺ and Cl⁻ ions. Therefore, option A is correct. The terms “strong” and “weak” refer to the degree of dissociation, not concentration – strong acids fully dissociate regardless of concentration (so B and D are wrong). Option C is contradictory as weak acids don’t fully dissociate.
Which substance turns methyl orange red?
A) aqueous ammonia
B) dilute hydrochloric acid
C) aqueous sodium hydroxide
D) distilled water
▶️ Answer/Explanation
Ans: B
1. Methyl orange is a pH indicator that turns red in acidic conditions (pH < 3.1).
2. Dilute hydrochloric acid is strongly acidic (pH ~1-2), so it turns methyl orange red.
3. Aqueous ammonia and sodium hydroxide are alkaline and would turn methyl orange yellow.
4. Distilled water is neutral (pH 7) and would give an intermediate orange color with methyl orange.
Which row shows the difference between a weak acid and a strong acid?
| weak acid | strong acid | |
|---|---|---|
| A | fully dissociated | partially dissociated |
| B | concentrated | dilute |
| C | dilute | concentrated |
| D | partially dissociated | fully dissociated |
▶️ Answer/Explanation
Ans: D
1. The key difference between weak and strong acids is their degree of dissociation in water.
2. Strong acids completely dissociate into ions in water (e.g., HCl → H+ + Cl–).
3. Weak acids only partially dissociate, establishing an equilibrium between dissociated and undissociated molecules (e.g., CH3COOH ⇌ H+ + CH3COO–).
4. Options B and C confuse strength (degree of dissociation) with concentration (amount of acid per unit volume).
5. Option A reverses the definitions.
Which colours are seen when litmus and methyl orange are added to separate samples of aqueous sodium hydroxide?
| litmus | methyl orange | |
|---|---|---|
| A | blue | orange |
| B | blue | yellow |
| C | purple | orange |
| D | purple | yellow |
▶️ Answer/Explanation
Ans: B
Sodium hydroxide is a strong alkali. Litmus turns blue in alkaline solutions (pH > 8). Methyl orange turns yellow in alkaline solutions (pH > 4.4). Purple is the neutral color for litmus, which would be incorrect for NaOH. Orange for methyl orange would indicate an acidic solution, which is also incorrect for NaOH.
Two acids, P and Q, with the same concentration and volume are reacted separately with the same mass of magnesium ribbon. The reactions produce the same total volume of hydrogen gas but acid Q reacts much more slowly than acid P. Which explanation for the difference between P and Q is correct?
- Acid P has a higher pH than acid Q.
- Acid P has a lower concentration of hydrogen ions.
- Acid Q is partially dissociated and acid P is fully dissociated.
- Acid Q is a proton acceptor.
▶️ Answer/Explanation
Ans: C
Since both acids produce the same volume of hydrogen gas, they must have the same total number of \(\text{H}^+\) ions available for reaction. However, acid Q reacts more slowly because it is partially dissociated (weak acid), while acid P is fully dissociated (strong acid). This means acid Q releases \(\text{H}^+\) ions at a slower rate, explaining the difference in reaction speed. Options A, B, and D are incorrect because they either contradict the given conditions or misrepresent the nature of acids.
What is the colour of thymolphthalein in dilute hydrochloric acid?
A) blue
B) red
C) yellow
D) colourless
▶️ Answer/Explanation
Ans: D
Thymolphthalein is a pH indicator that is:
1. Colourless in acidic solutions (pH < 8.3), such as dilute hydrochloric acid.
2. Turns blue in basic solutions (pH > 9.3).
Since hydrochloric acid is strongly acidic, thymolphthalein remains colourless.
Hydrogen chloride gas [\(M_r\): HCl, 36.5] is released at P in the apparatus shown. The universal indicator paper turns red after 38 s.
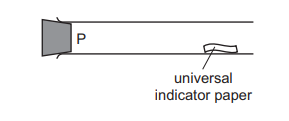
The experiment is repeated using sulfur dioxide gas \([M_r: SO_2, 64]\). What is the result for sulfur dioxide gas?
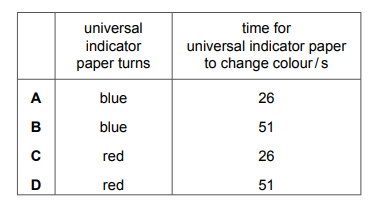
▶️ Answer/Explanation
Ans: D
The time taken for the gas to reach the indicator paper depends on its rate of diffusion, which is inversely proportional to the square root of its molar mass (Graham’s Law). For HCl (\(M_r = 36.5\)), the time was 38 s. For \(SO_2\) (\(M_r = 64\)), the rate of diffusion is slower because it is heavier. The time taken for \(SO_2\) will be longer than 38 s, as it diffuses more slowly compared to HCl. Thus, the correct result for \(SO_2\) corresponds to option D.
Which statement describes the properties of hydrochloric acid?
- Carbon dioxide is produced when limestone reacts with hydrochloric acid.
- Hydrogen is produced when sodium hydroxide reacts with hydrochloric acid.
- Methyl orange turns yellow in strong hydrochloric acid.
- Red litmus paper turns blue when dipped into hydrochloric acid.
▶️ Answer/Explanation
Ans: A
Hydrochloric acid (HCl) reacts with calcium carbonate (limestone) to produce carbon dioxide, water, and calcium chloride (statement A is correct). Sodium hydroxide neutralizes HCl, producing salt and water, not hydrogen (B is wrong). Methyl orange turns red in strong acids like HCl (C is wrong). HCl is acidic, so red litmus remains red (D is wrong). Thus, only A is correct.
Aqueous ethanoic acid is a weak acid. Aqueous sodium hydroxide is a strong base. Aqueous ethanoic acid is neutralised by aqueous sodium hydroxide. Which statements are correct?
- Aqueous ethanoic acid accepts protons from hydroxide ions.
- The aqueous ethanoic acid used is fully dissociated into ions.
- The aqueous sodium hydroxide used is fully dissociated into ions.
- The reaction produces a salt and water.
A) 1 and 2
B) 1 and 3
C) 2 and 4
D) 3 and 4
▶️ Answer/Explanation
Ans: D
Explanation:
1. Ethanoic acid (weak acid) donates protons to hydroxide ions, not accepts (statement 1 is incorrect).
2. Weak acids like ethanoic acid are only partially dissociated (statement 2 is incorrect).
3. Strong bases like NaOH are fully dissociated in solution (statement 3 is correct).
4. Neutralisation of an acid and base always produces a salt (sodium ethanoate) and water (statement 4 is correct).
Thus, only statements 3 and 4 are correct.
Water is added to anhydrous copper(II) sulfate.
What happens during the reaction?
- The copper(II) sulfate turns blue and the solution formed gets colder.
- The copper(II) sulfate turns blue and the solution formed gets hotter.
- The copper(II) sulfate turns white and the solution formed gets colder.
- The copper(II) sulfate turns white and the solution formed gets hotter.
▶️ Answer/Explanation
Ans: B
When water is added to anhydrous copper(II) sulfate (white), it forms hydrated copper(II) sulfate (blue). This is an exothermic reaction, releasing heat and making the solution hotter. Thus:
1. The color changes from white to blue (eliminating options C and D).
2. The reaction is exothermic, so the solution gets hotter (eliminating option A).
Therefore, the correct answer is B.
Which statement about acids is correct?
- A strong acid has a higher pH than a weak acid of the same concentration.
- A strong acid is a proton acceptor.
- A weak acid is a proton donor.
- A weak acid is fully ionized in aqueous solution.
▶️ Answer/Explanation
Ans: C
1. A strong acid has a lower pH than a weak acid (A is incorrect).
2. Acids are proton donors, not acceptors (B is incorrect).
3. A weak acid is a proton donor (C is correct).
4. A weak acid is partially ionized, not fully (D is incorrect).
Thus, only statement C is correct.
Methyl orange is added to dilute hydrochloric acid and to aqueous sodium hydroxide. What is the colour of the methyl orange in each solution?

▶️ Answer/Explanation
Ans: B
Methyl orange is an acid-base indicator that changes color depending on the pH of the solution:
1. In dilute hydrochloric acid (acidic medium), methyl orange turns red.
2. In aqueous sodium hydroxide (alkaline medium), methyl orange turns yellow.
From the given table in the image, option B correctly matches these observations.
Which statements about strong acids are correct?
- They have a high concentration of \(OH^–\) ions.
- They have a pH value of 1.
- They completely ionise in water.
- They turn red litmus blue.
A) 1 and 3
B) 1 and 4
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: C
Strong acids are characterized by:
1. High concentration of \(H^+\) ions, not \(OH^–\) (statement 1 is wrong).
2. A low pH (typically 1 or below for strong acids, so statement 2 is correct).
3. Complete ionisation in water (statement 3 is correct).
4. Turning blue litmus red, not red litmus blue (statement 4 is wrong).
Thus, only statements 2 and 3 are correct.
P is a hydrated metal salt with a blue colour. When P is heated, water is given off, leaving solid Q.
R is a hydrated metal salt with a pink colour. When R is heated, water is given off, leaving solid S.
Which row gives the name of P and the colour of S?

▶️ Answer/Explanation
Ans: C
Key Observations:
- Hydrated Copper(II) Sulfate (P): Blue in colour (\(\text{CuSO}_4 \cdot 5\text{H}_2\text{O}\)). When heated, it loses water to form anhydrous \(\text{CuSO}_4\) (Q), which is white.
- Hydrated Cobalt(II) Chloride (R): Pink in colour (\(\text{CoCl}_2 \cdot 6\text{H}_2\text{O}\)). When heated, it loses water to form anhydrous \(\text{CoCl}_2\) (S), which is blue.
Thus, the correct combination is:
- P: Copper(II) sulfate (blue hydrated form).
- S: Anhydrous cobalt(II) chloride (blue).
This matches Option C in the table.
Element X forms an oxide, XO, that neutralises sulfuric acid.
Which row describes X and XO?
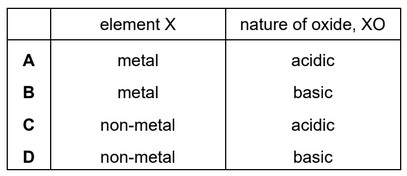
▶️ Answer/Explanation
Ans: B
1. Neutralisation of Sulfuric Acid: Since XO neutralizes \( \text{H}_2\text{SO}_4 \), it must be a basic oxide.
2. Nature of X: Basic oxides are typically formed by metals (e.g., \( \text{MgO}, \text{CaO} \)).
3. Electrical Conductivity: Metals conduct electricity in solid state due to delocalized electrons.
4. Conclusion: X is a metal (conducts electricity), and XO is a basic oxide (neutralizes acids).
Which row describes the properties of an acid?
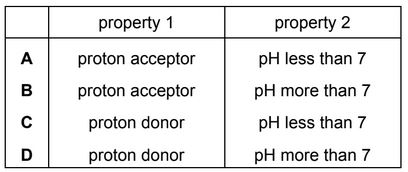
▶️ Answer/Explanation
Ans: C
Explanation:
Acids are proton (\( \text{H}^+ \)) donors (not acceptors), so the first column must be “proton donor.”
They turn blue litmus paper red (not the other way around), so the second column must be “red.”
Their pH is less than 7 (not greater than 7), so the third column must be “less than 7.”
Only Row C satisfies all these properties.
Ethanoic acid is a weak acid. Hydrochloric acid is a strong acid. Which statements are correct?
- Ethanoic acid molecules are partially dissociated into ions.
- 1.0 mol / \(dm^{3}\) ethanoic acid has a higher pH than 1.0 mol / \(dm^{3}\) hydrochloric acid.
- Ethanoic acid is always more dilute than hydrochloric acid.
- Ethanoic acid is a proton acceptor.
A. 1 and 2
B. 1 and 3
C. 2 and 4
D. 3 and 4
▶️ Answer/Explanation
Ans: A
Statement 1: Correct. Weak acids (like ethanoic acid) only partially dissociate into ions in solution.
Statement 2: Correct. Since ethanoic acid dissociates less, it produces fewer \(H^{+}\) ions, resulting in a higher pH than HCl at the same concentration.
Statement 3: Incorrect. Concentration is independent of acid strength; ethanoic acid can be more or less concentrated than HCl.
Statement 4: Incorrect. Ethanoic acid is a proton donor (Brønsted acid), not a proton acceptor.
Thus, only 1 and 2 are correct.
Four different acids are dissolved in water.
Which beaker contains the most concentrated strong acid solution?

▶️ Answer/Explanation
Ans: A
To determine the most concentrated strong acid solution:
1. Strong acids fully dissociate in water (e.g., HCl, HNO3).
2. Concentration depends on the number of H+ ions per unit volume.
3. From the image, Beaker A has the highest number of H+ ions (red dots) per unit volume compared to others.
4. Since it’s a strong acid (fully dissociated) and has the highest ion density, A is the most concentrated.
Which statement describes a weak acid?
A) It is a proton acceptor and is fully ionised in aqueous solution.
B) It is a proton acceptor and is partially ionised in aqueous solution.
C) It is a proton donor and is fully ionised in aqueous solution.
D) It is a proton donor and is partially ionised in aqueous solution.
▶️ Answer/Explanation
Ans: D
1. Acids are proton donors, not acceptors (A and B are incorrect).
2. A weak acid is only partially ionized in solution (C is incorrect).
3. A weak acid donates protons (\(\text{H}^+\)) but does not fully dissociate (D is correct).
Thus, the correct description is D.
The graph shows how the pH of a solution changes as an acid is added to an alkali.
acid + alkali → salt + water
Which letter represents the area of the graph where both acid and salt are present?

▶️ Answer/Explanation
Ans: D
1. The graph represents a titration curve where an acid is added to an alkali.
2. Region D (after the equivalence point) indicates excess acid is present, while salt (from the neutralization reaction) is already formed.
3. Before the equivalence point (Region B), only alkali and salt are present. At the equivalence point (Region C), only salt and water exist.
4. Thus, Region D is where both acid and salt coexist.
Carbonic acid is a weak acid formed when carbon dioxide dissolves in water.
What is the pH of the solution?
A) 1
B) 5
C) 7
D) 9
▶️ Answer/Explanation
Ans: B
Carbonic acid (\(H_2CO_3\)) is a weak acid, meaning it only partially dissociates in water, producing a mildly acidic solution. The pH of weak acids typically ranges between 3 and 6. Since:
- pH = 1 (A) is too acidic (strong acid range).
- pH = 7 (C) is neutral (pure water).
- pH = 9 (D) is alkaline.
The most reasonable pH for a weak acid like carbonic acid is 5 (B).
Barium hydroxide is an alkali. It reacts with hydrochloric acid.
How does the pH of the hydrochloric acid change as an excess of aqueous barium hydroxide is added?
- The pH decreases from pH14 and becomes constant at pH7.
- The pH decreases from pH14 to about pH1.
- The pH increases from pH1 and becomes constant at pH7.
- The pH increases from pH1 to about pH14.
▶️ Answer/Explanation
Ans: D
Key Observations:
- Initial State (HCl Acid): Hydrochloric acid is strongly acidic with a pH of about 1.
- Neutralization Reaction: \[ \text{Ba(OH)}_2 + 2\text{HCl} \rightarrow \text{BaCl}_2 + 2\text{H}_2\text{O} \] As barium hydroxide is added, it neutralizes the acid, raising the pH towards 7.
- Excess Barium Hydroxide: After complete neutralization, additional \(\text{Ba(OH)}_2\) makes the solution strongly alkaline, raising the pH to about 13-14.
Thus, the correct trend is: pH increases from pH1 (acidic) to pH14 (strongly alkaline), matching Option D.
Ethanoic acid reacts with water to produce an acidic solution.
Which row describes the roles of ethanoic acid and water in this reaction?

▶️ Answer/Explanation
Ans: C
1. Ethanoic Acid (\( \text{CH}_3\text{COOH} \)): Acts as a Brønsted-Lowry acid by donating a proton (\( \text{H}^+ \)) to water.
2. Water (\( \text{H}_2\text{O} \)): Acts as a Brønsted-Lowry base by accepting the proton.
3. Resulting Solution: The reaction forms \( \text{CH}_3\text{COO}^- \) (acetate ion) and \( \text{H}_3\text{O}^+ \) (hydronium ion), making the solution acidic.
4. Conclusion: The correct roles are described in Row C (ethanoic acid as an acid, water as a base).
Aqueous sodium hydroxide is added to solid Q in a test-tube. A gas is produced which turns damp red litmus blue. What is Q?
- Aluminium
- Ammonia
- Ammonium chloride
- Sodium nitrate
▶️ Answer/Explanation
Ans: C
The gas that turns damp red litmus blue is ammonia (NH₃), which is basic. Now, let’s analyze the options:
- Ammonium chloride (C) reacts with NaOH to produce NH₃ gas: \[ \text{NH}_4\text{Cl} + \text{NaOH} \rightarrow \text{NaCl} + \text{H}_2\text{O} + \text{NH}_3 \uparrow \]
- Aluminium (A) reacts with NaOH to produce hydrogen gas (H₂), which is neutral.
- Ammonia (B) is already a gas, not a solid.
- Sodium nitrate (D) does not react with NaOH to produce any gas.
Thus, the correct answer is C (Ammonium chloride).
Potassium hydroxide is a base. Which statement describes a reaction of potassium hydroxide?
A) Chlorine is formed when it is heated with ammonium chloride.
B) It turns Universal Indicator green.
C) It reacts with an acid to produce a salt and water.
D) It turns methyl orange red.
▶️ Answer/Explanation
Ans: C
Explanation:
A Incorrect: Heating KOH with NH4Cl produces ammonia (NH3), not chlorine.
B Incorrect: Bases turn Universal Indicator blue/purple, not green (which is neutral).
C Correct: Bases like KOH neutralize acids to form a salt and water (e.g., \( \text{KOH} + \text{HCl} \rightarrow \text{KCl} + \text{H}_2\text{O} \)).
D Incorrect: Bases turn methyl orange yellow, not red (acids turn it red).
Thus, only C accurately describes a reaction of potassium hydroxide.
Which solution has the lowest pH?
- 0.1 mol / dm3 ammonia solution
- 0.1 mol / dm3 ethanoic acid
- 0.1 mol / dm3 lithium hydroxide
- 0.1 mol / dm3 nitric acid
▶️ Answer/Explanation
Ans: D
Analysis of each option:
A. Ammonia (NH3): A weak base (pH > 7).
B. Ethanoic acid (CH3COOH): A weak acid (pH ~2.9 at 0.1 M).
C. Lithium hydroxide (LiOH): A strong base (pH ~13 at 0.1 M).
D. Nitric acid (HNO3): A strong acid (pH ~1 at 0.1 M).
Nitric acid fully dissociates, producing the highest [H+], thus the lowest pH.
Which statement about sulfuric acid is correct?
- It is made by the Haber process.
- It is made in the atmosphere by the action of lightning.
- It reacts with ammonia to produce a fertiliser.
- It reacts with copper metal to produce hydrogen gas.
▶️ Answer/Explanation
Ans: C
Let’s analyze each option:
1. A is incorrect because the Haber process produces ammonia (NH3), not sulfuric acid.
2. B is incorrect because lightning produces nitrogen oxides (NOx), not sulfuric acid.
3. C is correct because sulfuric acid reacts with ammonia to form ammonium sulfate, a widely used fertiliser (2NH3 + H2SO4 → (NH4)2SO4).
4. D is incorrect because sulfuric acid does not react with copper to produce hydrogen gas (copper is below hydrogen in the reactivity series).
Thus, the correct answer is C.
Which row shows how the hydrogen ion concentration and pH of ethanoic acid compare to those of hydrochloric acid of the same concentration?
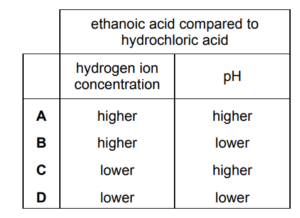
▶️ Answer/Explanation
Ans: C
1. Hydrochloric acid (HCl) is a strong acid and fully dissociates, producing a high \(\text{[H}^+]\) and low pH.
2. Ethanoic acid (CH₃COOH) is a weak acid and partially dissociates, resulting in a lower \(\text{[H}^+]\) and higher pH than HCl at the same concentration.
3. The table compares \(\text{[H}^+]\) and pH for both acids. Only row C correctly states that ethanoic acid has a lower \(\text{[H}^+]\) and higher pH than HCl.
Thus, the correct answer is C.
Which statements are properties of an acid?
- reacts with ammonium sulfate to form ammonia
- turns red litmus blue
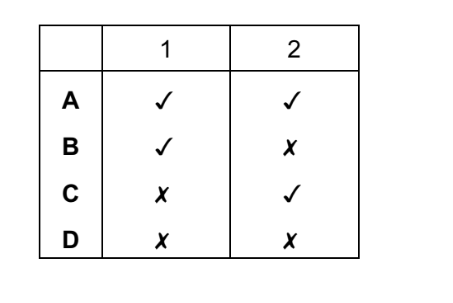
▶️ Answer/Explanation
Ans: D
1. Statement 1: Acids react with ammonium salts (like ammonium sulfate) to produce ammonia gas. This is a characteristic property of acids.
2. Statement 2: Acids turn blue litmus red, not red litmus blue (which is a property of bases). Thus, this statement is incorrect.
3. Conclusion: Only Statement 1 is correct. The correct option is D (1 only).
What is a property of concentrated sulfuric acid but not of dilute sulfuric acid?
A) It is a dehydrating agent.
B) It neutralises alkalis.
C) It produces a white precipitate with barium nitrate.
D) It reacts with metals to give a salt and hydrogen.
▶️ Answer/Explanation
Ans: A (It is a dehydrating agent)
Explanation:
- Option A: Concentrated sulfuric acid is a strong dehydrating agent, capable of removing water from substances like sugars or hydrated salts. Dilute sulfuric acid lacks this property.
- Option B: Both concentrated and dilute sulfuric acid neutralize alkalis (e.g., NaOH) to form salt and water.
- Option C: Both forms react with barium nitrate to produce a white precipitate of barium sulfate (\(BaSO_4\)).
- Option D: Dilute sulfuric acid reacts with reactive metals (e.g., Mg, Zn) to produce salt and hydrogen gas. Concentrated sulfuric acid reacts differently, often producing sulfur dioxide instead of hydrogen.
Thus, dehydration is unique to concentrated sulfuric acid.
Concentrated hydrochloric acid is a strong acid. What is meant by the terms ‘strong’ and ‘acid’?
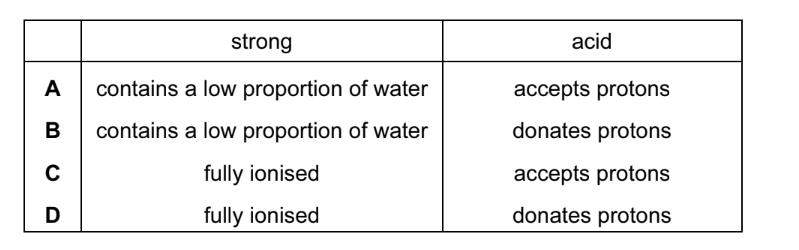
▶️ Answer/Explanation
Ans: D
To understand the terms:
- Acid: A substance that donates protons (H⁺ ions) in aqueous solution (Brønsted-Lowry definition).
- Strong acid: An acid that completely dissociates into its ions in aqueous solution (e.g., HCl → H⁺ + Cl⁻).
Key points:
- Strength refers to degree of dissociation, not concentration.
- Concentrated HCl is both strong (fully dissociates) and concentrated (high amount of solute).
Thus, the correct definitions are combined in D.
