Pre AP Chemistry -4.3B pH and Hydrogen Ion Concentration- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.3B pH and Hydrogen Ion Concentration- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.3B pH and Hydrogen Ion Concentration- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Which substance is the most acidic?

▶️ Answer/Explanation
Ans: B
pH Scale Analysis
| Substance | pH Value | Acidity Level |
|---|---|---|
| A. Water | 7.0 | Neutral |
| B. Lemon juice | 2.3 | Most acidic |
| C. Milk | 6.5 | Slightly acidic |
| D. Baking soda solution | 8.4 | Basic |
Key points:
- Acidity increases as pH decreases (pH < 7)
- Lemon juice contains citric acid (pH ≈ 2-3)
- Other substances are either:
- Neutral (water, pH 7)
- Mildly acidic (milk, pH ≈ 6.5)
- Basic (baking soda, pH > 7)
Conclusion: Lemon juice (Option B) has the lowest pH value (2.3), making it the most acidic substance among the options.
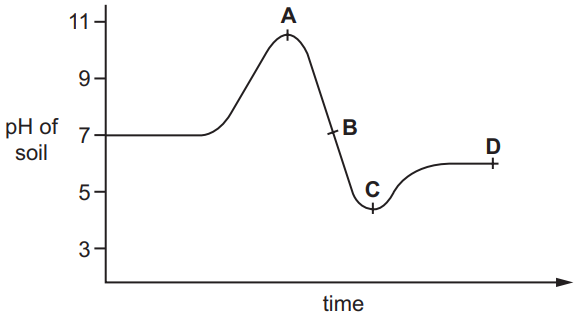
The graph shows how the pH of soil in a field changes over time.
At which point was the soil neutral?
▶️ Answer/Explanation
Ans: B
Understanding Neutral pH:
- Neutral pH is exactly 7 on the pH scale
- Below 7 is acidic, above 7 is alkaline
- The graph shows pH values at different time points
Graph Analysis:
- Point A: pH ≈ 5.5 (acidic)
- Point B: pH = 7 (neutral)
- Point C: pH ≈ 8.5 (alkaline)
- Point D: pH ≈ 6.5 (slightly acidic)
Key Observations:
- Only Point B touches the pH 7 line
- The soil becomes more alkaline after B
- Before B, the soil was acidic
Why Other Points Are Incorrect:
- A: Too acidic
- C: Too alkaline
- D: Still slightly acidic
Therefore, the soil was neutral only at Point B when the pH was exactly 7.
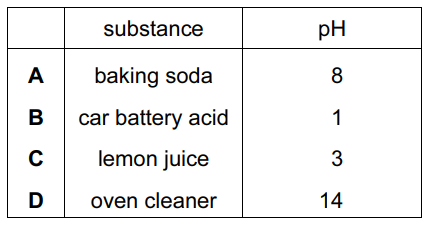
Ant stings hurt because of the methanoic acid produced by the ant.
Which substance could, most safely, be used to neutralize the acid?
▶️ Answer/Explanation
Ans: A
Key Considerations for Safe Neutralization:
- Methanoic acid (HCOOH) is a weak organic acid in ant venom
- Requires a mild base for safe neutralization on skin
- Should be non-toxic and non-irritating
Analysis of Options:
| Option | Substance | pH | Safety Evaluation |
|---|---|---|---|
| A | Sodium hydrogencarbonate (baking soda) | ~8.4 | Ideal choice:
|
| B | Sodium hydroxide | ~14 | Dangerous:
|
| C | Ammonium chloride | ~4.5-6 | Ineffective:
|
| D | Sodium chloride | ~7 | Neutral:
|
Chemical Reaction (Option A):
HCOOH + NaHCO3 → HCOONa + H2O + CO2
(Methanoic acid + Baking soda → Sodium methanoate + Water + Carbon dioxide)
Why Option A is Best:
- Creates a gentle fizz (CO2) that helps rinse the area
- Forms harmless sodium methanoate salt
- pH is close enough to neutral to prevent skin damage
- Commonly recommended by medical professionals
Therefore, A (sodium hydrogencarbonate) is the safest and most effective choice for neutralizing ant stings.
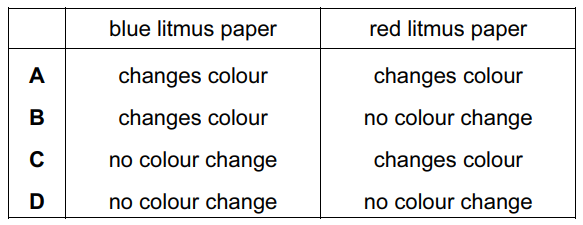
The diagram shows an experiment.
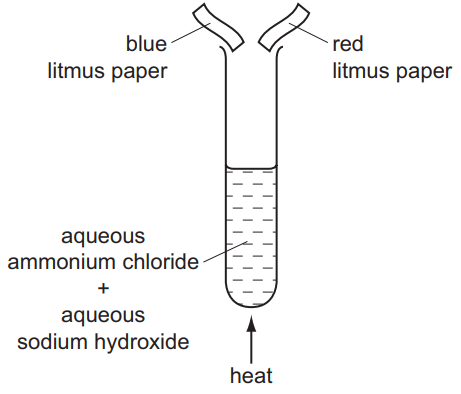
What happens to the pieces of litmus paper?
▶️ Answer/Explanation
Ans: C
1. Chemical Reaction:
NH4Cl + NaOH → NaCl + NH3↑ + H2O
Ammonia gas (NH3) is produced and travels up the delivery tube 2. Litmus Behavior:
• NH3 dissolves in water on damp litmus papers:
NH3 + H2O ⇌ NH4+ + OH–
• OH– ions make solution alkaline (pH ≈ 11) 3. Color Changes:
• Red litmus: Turns blue (alkaline indicator)
• Blue litmus: Remains blue (already in correct form)
• Dry papers show no change (no water for NH3 dissolution) 4. Why Not Other Options:
• A/B: Incorrectly show blue→red change (acidic condition)
• D: Shows no change for red litmus (would require neutral gas)
Two indicators, bromophenol blue and Congo red, show the following colours in acidic solutions and in alkaline solutions.
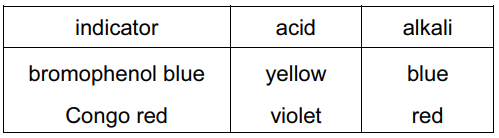
A few drops of each indicator are added to separate samples of a solution of pH 2.
What are the colours of the indicators in this solution?

▶️ Answer/Explanation
Ans: D
Analysis of indicator behavior at pH 2 (strongly acidic):
| Indicator | Acidic Color | Alkaline Color | Color at pH 2 |
|---|---|---|---|
| Bromophenol blue | Yellow | Blue | Yellow (acidic form) |
| Congo red | Violet | Red | Violet (acidic form) |
Key observations:
- At pH 2, both indicators show their acidic colors
- Bromophenol blue transitions between pH 3.0-4.6 (yellow ↔ blue)
- Congo red transitions between pH 3.0-5.2 (violet ↔ red)
- pH 2 is well below both transition ranges
Therefore, the correct combination is yellow (bromophenol blue) and violet (Congo red), matching option D.
Lead iodide is insoluble in water.
Lead iodide is made by adding aqueous lead nitrate to aqueous potassium iodide.
Which pieces of apparatus are needed to obtain solid lead iodide from 20cm³ of aqueous lead nitrate?
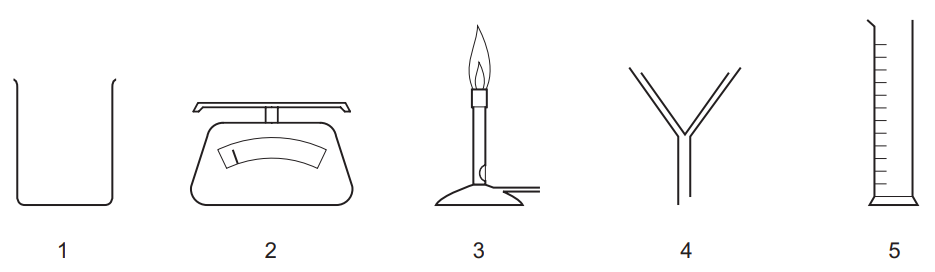
A 1, 2 and 4 B 1, 3 and 5 C 1, 4 and 5 D 2, 4 and 5
▶️ Answer/Explanation
Ans: B (1, 3 and 5)
Required apparatus for the precipitation and isolation of lead iodide:
- Beaker (1): For mixing the solutions and precipitation reaction
- Bunsen burner (3): To gently heat and concentrate the solution if needed
- Measuring cylinder (5): To accurately measure 20cm³ of lead nitrate solution
Why other options are incorrect:
- A/C/D: Include unnecessary items (filter funnel/evaporating basin) or omit essential ones
- Filter paper (2) and evaporating basin (4) aren’t needed as lead iodide precipitates can be collected by simple decantation
The complete process requires: measuring → mixing → heating → decanting → drying.
Compound X is tested and the results are shown in the table.

Which ions are present in compound X?
- ammonium ions and carbonate ions
- ammonium ions and chloride ions
- calcium ions and carbonate ions
- calcium ions and chloride ions
▶️ Answer/Explanation
Ans: A
1. Test Analysis:
– Dilute HCl test: Effervescence and limewater turning milky confirms CO32- ions (carbonate).
– NaOH + heat test: Ammonia gas (turns damp red litmus blue) confirms NH4+ ions (ammonium).
2. Negative Results:
– No precipitate with NaOH → excludes Ca2+
– No precipitate with AgNO3 → excludes Cl–
3. Conclusion:
Only ammonium and carbonate ions explain all test results.
The table shows the pH of four aqueous solutions, W, X, Y and Z.
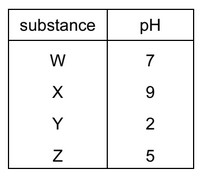
Universal Indicator is added to each solution.
Which row shows the colour of each solution after the indicator is added?

▶️ Answer/Explanation
Ans: B
Universal indicator colors correspond to pH values as follows:
| pH Range | Color |
|---|---|
| 0-3 (Strong acid) | Red |
| 4-6 (Weak acid) | Orange/Yellow |
| 7 (Neutral) | Green |
| 8-11 (Weak base) | Blue |
| 12-14 (Strong base) | Purple |
Analysis of each solution:
- W (pH 7): Neutral → Green
- X (pH 13): Strong base → Purple
- Y (pH 2): Strong acid → Red
- Z (pH 5): Weak acid → Orange
Only option B correctly matches all these color changes.
Ammonium chloride reacts with solution X to produce alkaline gas Y. The equation is shown.
ammonium chloride + solution X → alkaline gas Y
Which row identifies X and Y?
| X | Y | |
|---|---|---|
| A | hydrochloric acid | ammonia |
| B | hydrochloric acid | chlorine |
| C | sodium hydroxide | ammonia |
| D | sodium hydroxide | chlorine |
▶️ Answer/Explanation
Ans: C
Ammonium chloride reacts with sodium hydroxide (a base) to produce ammonia gas (NH₃), which is alkaline. This is a typical test for ammonium ions. The reaction is:
NH₄Cl + NaOH → NaCl + H₂O + NH₃
Hydrochloric acid (options A and B) would not produce an alkaline gas, and chlorine (options B and D) is not alkaline. Therefore, the correct combination is sodium hydroxide (X) and ammonia (Y).
Which indicators turn blue when added to aqueous ammonia?
- litmus
- thymolphthalein
- universal indicator
A) 1, 2 and 3
B) 1 and 2 only
C) 1 and 3 only
D) 2 and 3 only
▶️ Answer/Explanation
Ans: A
Aqueous ammonia is a weak alkali (pH about 11). Let’s examine each indicator:
1. Litmus turns blue in alkaline solutions (pH > 7).
2. Thymolphthalein is colorless in acidic and neutral solutions but turns blue in alkaline solutions (pH > 9.3).
3. Universal indicator turns blue/purple in alkaline solutions, depending on the pH.
Since all three indicators turn blue (or bluish colors) in alkaline solutions like aqueous ammonia, the correct answer is A (1, 2, and 3).
A student tests four solutions with universal indicator.
Which colour identifies the solution containing the greatest concentration of OH– ions?
A) red
B) yellow
C) green
D) blue
▶️ Answer/Explanation
Ans: D
Universal indicator shows different colors at different pH levels:
- Red (pH 1-3): Strongly acidic (very low OH– concentration)
- Yellow (pH 4-6): Weakly acidic (low OH– concentration)
- Green (pH 7): Neutral (equal H+ and OH– concentrations)
- Blue (pH 8-11): Alkaline (high OH– concentration)
The blue color indicates the highest pH and therefore the greatest concentration of OH– ions among the given options.
Compound M contains calcium. Two reactions of M are listed.
- M reacts with dilute hydrochloric acid to form a salt and water only.
- M reacts with aqueous ammonium chloride to form a gas that turns damp red litmus paper blue.
What is M?
A) CaOH
B) Ca(OH)₂
C) CaCO₃
D) Ca(CO₃)₂
▶️ Answer/Explanation
Ans: B
Let’s analyze the clues:
First reaction: Forms salt and water only – this is characteristic of a base (hydroxide) reacting with acid. Carbonates would produce CO₂ gas as well.
Second reaction: The gas turns damp red litmus blue – this indicates ammonia (NH₃), which is basic. Calcium hydroxide reacts with ammonium chloride to produce ammonia:
\[ \text{Ca(OH)}_2 + 2\text{NH}_4\text{Cl} \rightarrow \text{CaCl}_2 + 2\text{NH}_3 + 2\text{H}_2\text{O} \]
CaOH doesn’t exist as a stable compound. The carbonate options would produce CO₂ with acid, not just salt and water. Therefore, M must be calcium hydroxide, Ca(OH)₂.
When a few drops of water are added to a solid, E, the colour changes from blue to pink. What is E?
A) anhydrous cobalt(II) chloride
B) anhydrous copper(II) sulfate
C) hydrated cobalt(II) chloride
D) hydrated copper(II) sulfate
▶️ Answer/Explanation
Ans: A
The key observations are:
- The solid changes color when water is added
- The color change is from blue to pink
This is characteristic of anhydrous cobalt(II) chloride (CoCl₂):
- Anhydrous form: Blue
- Hydrated form: Pink
Copper(II) sulfate changes from white (anhydrous) to blue (hydrated), so it doesn’t match. The hydrated forms wouldn’t show this color change when more water is added. Therefore, the correct answer is anhydrous cobalt(II) chloride.
An aqueous solution of Z turns universal indicator paper purple.
Which row identifies the colour of methyl orange and of thymolphthalein when they are added separately to an aqueous solution of Z?
| methyl orange | thymolphthalein | |
|---|---|---|
| A | yellow | blue |
| B | yellow | colourless |
| C | red | blue |
| D | red | colourless |
▶️ Answer/Explanation
Ans: A
Universal indicator turning purple indicates Z is alkaline (pH > 7).
Indicator color changes:
1. Methyl orange:
– Red in acid (pH < 3.1)
– Yellow in neutral/alkaline (pH ≥ 4.4)
→ In alkali: yellow
2. Thymolphthalein:
– Colorless in acid/neutral (pH < 9.3)
– Blue in alkali (pH ≥ 10.5)
→ In strong alkali: blue
Therefore, for an alkaline solution Z: methyl orange = yellow, thymolphthalein = blue (option A).
Universal indicator is added to an aqueous solution of oxide X. The indicator changes colour from green to red. What is X?
A) MgO
B) CaO
C) K2O
D) NO2
▶️ Answer/Explanation
Ans: D
1. pH Indicator Analysis:
• Green → Red indicates a change from neutral (pH 7) to acidic (pH 1-3)
• Only acidic oxides cause this color shift
2. Oxide Classification:
• MgO, CaO, K2O: Basic oxides (form alkaline solutions)
• NO2: Acidic oxide (forms HNO3 in water)
3. Chemical Behavior:
NO2 + H2O → HNO3 + NO
The nitric acid produced gives the acidic solution
4. Why Not Others:
• A/B/C would turn indicator blue/purple (basic)
• Only D produces the required acidic conditions
Thus, D (NO2) is the correct answer.
Aqueous sodium hydroxide is reacted with excess dilute hydrochloric acid. Which ion causes the resulting mixture to be acidic?
A \(Na^+\)
B \(H^+\)
C \(OH^-\)
D \(Cl^-\)
▶️ Answer/Explanation
Ans: B
This neutralization reaction involves:
NaOH(aq) + HCl(aq) → NaCl(aq) + H2O(l)
Key points about the resulting mixture:
- Excess HCl means there are leftover H+ ions after neutralization
- These H+ ions make the solution acidic (pH < 7)
- Other ions present:
- Na+ – neutral spectator ion
- Cl– – neutral spectator ion
- OH– – all reacted with H+
Why other options are incorrect:
- A: Na+ doesn’t affect pH
- C: OH– would make solution basic, but it’s all consumed
- D: Cl– is a neutral ion
Therefore, only the excess H+ ions (Option B) cause the acidic nature of the final solution.
Which row shows the colours of litmus and methyl orange with solutions of acids or bases?
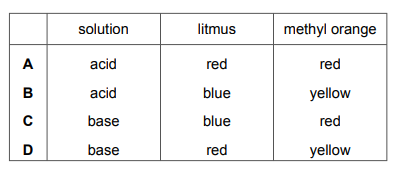
▶️ Answer/Explanation
Ans: A
Correct indicator colors:
| Indicator | Acid | Base |
|---|---|---|
| Litmus | Red | Blue |
| Methyl Orange | Red | Yellow |
Only row A correctly shows:
– Litmus: red in acid, blue in base
– Methyl orange: red in acid, yellow in base
The other rows have incorrect combinations of these indicator colors.
Four different solutions are separately tested with blue litmus and with methyl orange. Each solution is known to be either acidic or alkaline. The results are shown.
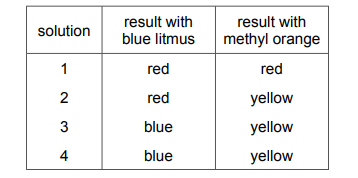
Which statement is correct?
A) Solutions 1 and 4 are acidic.
B) Solutions 1 and 2 are alkaline.
C) Solutions 3 and 4 are acidic.
D) Solutions 3 and 4 are alkaline.
▶️ Answer/Explanation
Ans: D
To analyze the results, we need to understand the indicator behaviors:
| Indicator | Acidic Solution | Alkaline Solution |
|---|---|---|
| Blue litmus | Turns red | Remains blue |
| Methyl orange | Red | Yellow |
Analyzing each solution:
- Solution 1:
- Blue litmus: Red → Acidic
- Methyl orange: Red → Acidic
- Conclusion: Acidic
- Solution 2:
- Blue litmus: Red → Acidic
- Methyl orange: Orange → Weakly acidic
- Conclusion: Acidic
- Solution 3:
- Blue litmus: Blue → Alkaline
- Methyl orange: Yellow → Alkaline
- Conclusion: Alkaline
- Solution 4:
- Blue litmus: Blue → Alkaline
- Methyl orange: Yellow → Alkaline
- Conclusion: Alkaline
Therefore, Solutions 3 and 4 are alkaline (Option D) is the correct statement.
Which statement about sulfur dioxide or calcium oxide is correct?
A Calcium oxide is an acid.
B Calcium oxide turns thymolphthalein yellow.
C Sulfur dioxide is a base.
D Sulfur dioxide turns thymolphthalein colourless.
▶️ Answer/Explanation
Ans: D
1. Chemical properties:
– Calcium oxide (CaO) is a basic oxide (not acid, so A is wrong)
– Sulfur dioxide (SO2) is an acidic oxide (not base, so C is wrong)
2. Indicator behavior:
– Thymolphthalein turns blue in bases, colorless in acids/neutral (B is wrong)
– SO2 forms acidic solution → turns thymolphthalein colorless (D is correct)
Dilute hydrochloric acid reacts with aqueous sodium hydroxide in a neutralisation reaction. Which two ions are involved in this neutralisation reaction?
![]()
▶️ Answer/Explanation
Ans: B (H+ and OH–)
The neutralization reaction between HCl and NaOH can be represented as:
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
Key points about the ionic equation:
- The actual neutralization occurs between:
- Hydrogen ions (H+) from the acid
- Hydroxide ions (OH–) from the base
- The net ionic equation is:
H+(aq) + OH–(aq) → H2O(l)
- Na+ and Cl– are spectator ions that don’t participate in the actual neutralization
Therefore, the correct pair of ions involved is H+ and OH– (Option B).
Compound X dissolves in water to form an aqueous solution. Methyl orange is added to aqueous compound X. The methyl orange turns red. What is compound X?
- sodium carbonate
- copper(II) oxide
- potassium oxide
- sulfur dioxide
▶️ Answer/Explanation
Ans: D
Methyl orange is an acid-base indicator that turns:
- Red in acidic solutions (pH < 3.1)
- Yellow in basic solutions (pH > 4.4)
Analysis of options:
- A (Na2CO3): Forms basic solution → methyl orange would turn yellow
- B (CuO): Insoluble in water → wouldn’t form aqueous solution
- C (K2O): Forms strongly basic KOH solution → methyl orange would turn yellow
- D (SO2): Forms acidic sulfurous acid (H2SO3) → methyl orange turns red
Thus, only sulfur dioxide (D) produces the observed color change.
Which list contains only chemical changes?
A melting, evaporating, dissolving
B rusting, freezing, cracking
C neutralisation, polymerisation, combustion
D boiling, condensing, distillation
▶️ Answer/Explanation
Ans: C
Chemical Change Analysis:
- Option C (All Chemical Changes):
- Neutralization: Acid + Base → Salt + Water (new substances formed)
- Polymerization: Monomers → Polymers (molecular structure changes)
- Combustion: Fuel + O2 → CO2 + H2O (irreversible reaction)
- Other Options:
- A: All physical changes (state changes only)
- B: Contains freezing (physical change)
- D: All physical changes (phase transitions)
Chemical changes always result in new substances with different properties. Only Option C contains processes that all meet this criterion:
- Irreversible under normal conditions
- Involve breaking/forming chemical bonds
- Result in fundamentally different products
