Pre AP Chemistry -4.3C Acid–Base Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -Link- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -Link- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
An excess of substance Z is added to some spilt acid. The solution produced as a result is neutral. What is Z?
A) Aqueous ammonia
B) Aqueous sodium hydroxide
C) Calcium carbonate
D) Water
▶️ Answer/Explanation
Ans: C
1. Neutralization Mechanism:
CaCO3 + 2H+ → Ca2+ + H2O + CO2↑
The carbonate neutralizes acid completely, leaving neutral Ca2+ ions in solution 2. Why Calcium Carbonate is Ideal:
• Self-limiting reaction (stops when acid is neutralized)
• Non-corrosive solid (safe for spill cleanup)
• Produces visible CO2 bubbles to monitor progress
• Forms neutral calcium salts (pH ≈ 7) 3. Problems with Other Options:
• A/B: Strong bases cause overshoot (pH > 7)
• D: Merely dilutes acid without neutralization
4. Practical Application:
Calcium carbonate is commonly used in acid spill kits for laboratories and industries
Potassium hydroxide is a base. Which statement describes a reaction of potassium hydroxide?
A Chlorine is formed when it is heated with ammonium chloride.
B It turns Universal Indicator green.
C It reacts with an acid to produce a salt and water.
D It turns methyl orange red.
▶️ Answer/Explanation
Ans: C
Analysis of each option for potassium hydroxide (KOH), a strong base:
- Option A (Incorrect):
- Heating KOH with NH4Cl produces ammonia (NH3), not chlorine
- Reaction: NH4Cl + KOH → NH3 + H2O + KCl
- Option B (Incorrect):
- Universal Indicator turns purple (not green) in strong bases like KOH
- Green indicates neutral pH (7), while KOH solutions are strongly basic (pH > 12)
- Option C (Correct):
- Characteristic neutralization reaction of bases
- Example: KOH + HCl → KCl + H2O
- Forms potassium salt + water
- Option D (Incorrect):
- Methyl orange turns yellow in bases (red in acids)
- KOH would make methyl orange yellow, not red
Therefore, only option C correctly describes a fundamental property of potassium hydroxide as a base.
Dilute nitric acid is added to a solid, F.
A gas, G, is produced which is denser than air and extinguishes a burning splint.
What are F and G?
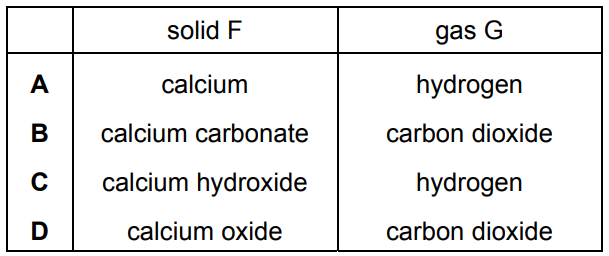
▶️ Answer/Explanation
Ans: B (F = calcium carbonate, G = carbon dioxide)
Key observations and their interpretation:
- Gas properties:
- Denser than air (CO₂ has higher molecular weight than N₂/O₂)
- Extinguishes flame (CO₂ is non-combustible and displaces oxygen)
- Chemical reaction:
CaCO₃(s) + 2HNO₃(aq) → Ca(NO₃)₂(aq) + CO₂(g) + H₂O(l)
- Only carbonate reactions produce CO₂ gas with acids
- This eliminates options with sulfur dioxide (A) and hydrogen (D)
- Image analysis:
- Option B shows the correct combination (CaCO₃ → CO₂)
- Other options show incorrect gas production
Therefore, solid F must be calcium carbonate and gas G must be carbon dioxide.
A solution of compound Z gives a light blue precipitate with aqueous ammonia. The precipitate dissolves in an excess of ammonia.
A flame test is done on compound Z.
What is the colour of the flame?
- blue-green
- lilac
- red
- yellow
▶️ Answer/Explanation
Ans: A
Analysis of the chemical tests:
| Test | Observation | Interpretation |
|---|---|---|
| Ammonia test | Light blue precipitate dissolves in excess | Characteristic of copper(II) ions |
| Flame test | Color produced | Copper gives blue-green flame |
Key points:
- Ammonia test results:
- Cu2+ + 2NH3 + 2H2O → Cu(OH)2↓ (light blue) + 2NH4+
- Cu(OH)2 + 4NH3 → [Cu(NH3)4]2+ (deep blue solution)
- Flame test colors:
- Copper: blue-green (option A)
- Potassium: lilac (option B)
- Lithium/Strontium: red (option C)
- Sodium: yellow (option D)
The light blue precipitate that dissolves in excess ammonia is characteristic of copper(II) ions, which produce a blue-green flame color.
Which property is shown by the alkali sodium hydroxide?
- It has a pH less than pH 7.
- It produces a gas when it is warmed with ammonium chloride.
- It turns blue litmus red.
- It turns Universal Indicator green.
▶️ Answer/Explanation
Ans: B
1. Alkali properties:
Sodium hydroxide (NaOH) is a strong alkali with pH > 7 (eliminates A and C).
2. Reaction with NH4Cl:
NaOH + NH4Cl → NaCl + H2O + NH3↑ (ammonia gas produced – correct).
3. Indicator behavior:
– Turns red litmus blue (not blue to red – C wrong)
– Turns UI purple (not green – D wrong)
Only B correctly describes an alkali property of NaOH.
What is not a typical characteristic of acids?
- They react with alkalis producing water.
- They react with all metals producing hydrogen.
- They react with carbonates producing carbon dioxide.
- They turn blue litmus paper red.
▶️ Answer/Explanation
Ans: B
Analysis of acid characteristics:
- Reaction with alkalis (A):
- True: Acids + bases → salt + water (neutralization)
- Reaction with metals (B):
- Not always true: Only reactive metals (above H in reactivity series) react
- Noble metals (Au, Pt) don’t react with most acids
- This is the incorrect statement
- Reaction with carbonates (C):
- True: Acid + carbonate → salt + water + CO2
- Litmus test (D):
- True: Acids turn blue litmus red
Key points:
- Option B is the exception – acids don’t react with all metals
- This makes it the correct answer as it’s not a universal characteristic
Therefore, Option B is not a typical characteristic of acids.
Magnesium, phosphorus and chlorine are elements in the same period of the Periodic Table. Which row describes the type of oxide formed by each of these elements?
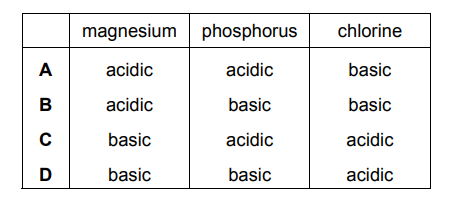
▶️ Answer/Explanation
Ans: C
Analysis of oxides formed by period 3 elements:
- Magnesium Oxide (MgO):
- Formed by a metal
- Basic oxide (reacts with acids)
- pH > 7 in water
- Phosphorus Oxides (P4O6, P4O10):
- Formed by a non-metal
- Acidic oxides (form phosphoric acids in water)
- pH < 7
- Chlorine Oxides (Cl2O, ClO2, Cl2O7):
- Formed by a non-metal
- Strongly acidic oxides
- Form chloric acids in water
Thus, the correct classification is:
• Magnesium – basic
• Phosphorus – acidic
• Chlorine – acidic
This matches option C in the answer choices.
Which reaction is a neutralisation reaction?
A AgNO3 + HCl → AgCl + HNO3
B CaCO3 → CaO + CO2
C 4Na + O2 → 2Na2O
D 2NaOH + H2SO4 → Na2SO4 + 2H2O
▶️ Answer/Explanation
Ans: D
Neutralization Reaction Characteristics:
- Option D (Neutralization):
- Base (NaOH) + Acid (H2SO4) → Salt (Na2SO4) + Water (H2O)
- H+ + OH– → H2O (characteristic of neutralization)
- pH moves toward 7 (neutral)
- Other Options:
- A: Precipitation reaction (forms AgCl solid)
- B: Thermal decomposition
- C: Oxidation reaction
Key features of neutralization:
- Must involve an acid and a base as reactants
- Always produces salt and water
- Results in a more neutral pH
Only Option D shows all these characteristics, making it the correct neutralization reaction.
A student is given an unknown solution.
Which two tests provide evidence that the solution is copper(II) sulfate?
1 adding dilute hydrochloric acid
2 adding aqueous sodium hydroxide
3 adding dilute nitric acid, then silver nitrate solution
4 adding dilute nitric acid, then barium nitrate solution
A 1 and 3 B 1 and 4 C 2 and 3 D 2 and 4
▶️ Answer/Explanation
Ans: D
To confirm a solution contains copper(II) sulfate (CuSO4), we need two tests:
Test 2: Sodium Hydroxide
Identifies Cu2+ ions:
\[ Cu^{2+} + 2OH^- \rightarrow Cu(OH)_2 \downarrow \]
Observation: Pale blue gelatinous precipitate
Test 4: Barium Nitrate
Identifies SO42- ions:
\[ Ba^{2+} + SO_4^{2-} \rightarrow BaSO_4 \downarrow \]
Observation: White precipitate (after acidification)
Why other tests don’t work:
- Test 1 (HCl): No reaction with CuSO4
- Test 3 (AgNO3): Tests for chloride ions (not relevant)
Conclusion: Only tests 2 and 4 together confirm both copper(II) and sulfate ions are present, making D the correct answer.
Elements W and X are metals.
Elements Y and Z are non-metals.
The oxides of W, X, Y and Z all form solutions when added to water.
Which statement is correct?
A The solution of the oxide of element W turns blue litmus red.
B The solution of the oxide of element X fizzes when sodium carbonate is added.
C The solution of the oxide of element Y has a pH greater than pH 7.
D The solution of the oxide of element Z fizzes when powdered magnesium is added.
▶️ Answer/Explanation
Ans: D
Analysis of Each Option:
Option A (Metal Oxide W):
- Metal oxides form basic solutions (pH > 7)
- Basic solutions turn red litmus blue, not blue to red
- This statement is incorrect
Option B (Metal Oxide X):
- Basic solutions don’t react with sodium carbonate
- Fizzing would require acid-carbonate reaction (CO₂ production)
- This statement is incorrect
Option C (Non-metal Oxide Y):
- Non-metal oxides form acidic solutions (pH < 7)
- pH greater than 7 would indicate basicity
- This statement is incorrect
Option D (Non-metal Oxide Z):
- Non-metal oxides form acidic solutions
- Magnesium reacts with acids: Mg + 2H⁺ → Mg²⁺ + H₂↑
- Hydrogen gas production causes fizzing
- This is the correct statement
Key Chemical Principles:
- Metal oxides + water → Basic solutions (e.g., Na₂O + H₂O → 2NaOH)
- Non-metal oxides + water → Acidic solutions (e.g., CO₂ + H₂O → H₂CO₃)
- Only acids react with:
- Metals (producing H₂ gas)
- Carbonates (producing CO₂ gas)
Therefore, the only correct statement is D, as it correctly describes the reaction between an acidic non-metal oxide solution and magnesium.
When blue copper(II) sulfate is heated, a white solid and water are formed.
The white solid turns blue and gives out heat when water is added to it.
Which terms describe the blue copper(II) sulfate and the reactions?

▶️ Answer/Explanation
Ans: C
Chemical Process Analysis:
- Initial Substance: Blue copper(II) sulfate is hydrated (CuSO4·5H2O)
- Heating Reaction:
CuSO4·5H2O (blue) → CuSO4 (white) + 5H2O
- Water molecules are lost (endothermic process)
- Color change indicates dehydration
- Rehydration:
CuSO4 + 5H2O → CuSO4·5H2O + heat
- Exothermic reaction (heat released)
- Original hydrated compound reforms
Key Classification:
| Term | Explanation | Match to Option C |
|---|---|---|
| Hydrated | Original blue compound contains water of crystallization | ✓ Both correctly identified |
| Reversible | Process can go forward (dehydration) and backward (rehydration) |
Why Other Options Are Incorrect:
- A: Incorrectly suggests anhydrous for the blue form
- B: Wrongly implies the white form is hydrated
- D: Misclassifies the reaction as irreversible
This demonstrates a classic reversible hydration/dehydration process, making C the correct choice.
Hydrochloric acid is added to magnesium metal and to sodium carbonate in separate tests.
Which row shows the observations?
| Magnesium metal | Sodium carbonate | |
|---|---|---|
| A | Effervescence | Effervescence |
| B | Effervescence | No reaction |
| C | No reaction | Effervescence |
| D | No reaction | No reaction |
▶️ Answer/Explanation
Ans: A
1. Magnesium Reaction:
Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)↑
• Vigorous effervescence (H2 gas bubbles)
• Exothermic (temperature increase)
• Metal dissolves completely 2. Sodium Carbonate Reaction:
Na2CO3(s) + 2HCl(aq) → 2NaCl(aq) + H2O(l) + CO2(g)↑
• Rapid effervescence (CO2 gas bubbles)
• Solution becomes clear as solid dissolves 3. Key Observations:
• Both reactions produce visible gas evolution
• No remaining solids when acid is in excess
• Distinctive bubble patterns (H2 rises quickly, CO2 forms slower streams)
Which row correctly matches the experiment and observations to the identity of the underlined substance?

▶️ Answer/Explanation
Ans: D
The correct identification involves understanding the reversible hydration of copper(II) sulfate:
| Process | Observation | Chemical Explanation |
|---|---|---|
| Heating | Blue → White | CuSO₄·5H₂O → CuSO₄ + 5H₂O (dehydration) |
| Adding water | White → Blue | CuSO₄ + 5H₂O → CuSO₄·5H₂O (rehydration) |
Key points about the correct option D:
- The white anhydrous form (CuSO₄) turns blue when water is added, forming the pentahydrate (CuSO₄·5H₂O)
- This is a characteristic test for water
- The color change is due to formation of the [Cu(H₂O)₄]²⁺ complex ion
Why other options are incorrect:
- A: Cobalt chloride changes from blue to pink, not white to blue
- B: Calcium carbonate doesn’t show this reversible color change
- C: Sodium chloride doesn’t undergo this hydration reaction
Four different solutions, W, X, Y and Z, are tested with Universal Indicator.
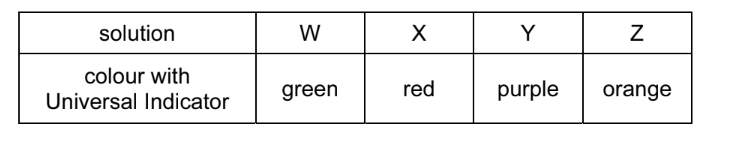
Which solutions are acidic?
A W and Z B X and Z C X only D Y only
▶️ Answer/Explanation
Ans: B (X and Z)
Universal Indicator color interpretation:
| Solution | Color | pH Range | Acidity |
|---|---|---|---|
| W | Green | 7 (Neutral) | – |
| X | Red | 1-3 (Strong acid) | Acidic |
| Y | Purple | 10-14 (Strong base) | – |
| Z | Orange | 4-6 (Weak acid) | Acidic |
Therefore, only solutions X (red) and Z (orange) show acidic colors, making option B correct.
Which statements are properties of an acid?
1 reacts with ammonium sulfate to form ammonia
2 turns red litmus blue
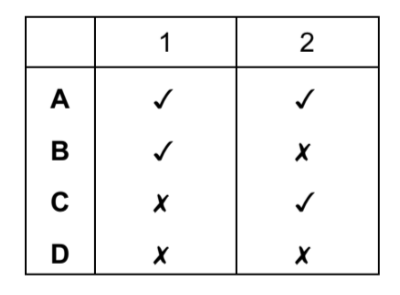
▶️ Answer/Explanation
Ans: D
Analysis of the statements:
| Statement | Property of | Explanation |
|---|---|---|
| 1 | Base | Only bases react with ammonium salts to release NH3 |
| 2 | Base | Bases turn red litmus blue (acids turn blue litmus red) |
Key chemical concepts:
- Ammonia formation:
- Base + NH4+ → NH3 + H2O
- Acids cannot produce ammonia from ammonium salts
- Litmus test:
- Acids: turn blue litmus red
- Bases: turn red litmus blue
- The statements describe base properties, not acid properties
Therefore, neither statement describes acid properties, making D the correct answer.
One of the instructions for an experiment reads as follows. Quickly add \(50cm^{3}\) of acid. What is the best piece of apparatus to use?
A a burette
B a conical flask
C a measuring cylinder
D a pipette
▶️ Answer/Explanation
Ans: C
1. Apparatus comparison:
– Burette (A): Precise but slow delivery
– Conical flask (B): Not for measuring/adding liquids
– Measuring cylinder (C): Quick volume measurement and pouring
– Pipette (D): Precise but for smaller volumes
2. Key requirement:
“Quickly add” favors the measuring cylinder for its speed and adequate accuracy for 50cm³.
3. Safety note:
When adding acid quickly, always pour down a glass rod to prevent splashing.
Which property is not characteristic of a base?
- It reacts with a carbonate to form carbon dioxide.
- It reacts with an acid to form a salt.
- It reacts with an ammonium salt to form ammonia.
- It turns universal indicator paper blue.
▶️ Answer/Explanation
Ans: A
Analysis of base characteristics:
- Reaction with carbonates (A):
- This is actually a property of acids, not bases
- Acid + Carbonate → Salt + Water + CO2
- Bases do not typically react with carbonates to produce CO2
- Reaction with acids (B):
- True: Base + Acid → Salt + Water (neutralization)
- Reaction with ammonium salts (C):
- True: Base + NH4+ → NH3 + H2O + salt
- Indicator test (D):
- True: Bases turn indicators blue/purple
Key points:
- Option A describes an acid’s behavior, not a base’s
- This makes it the correct answer as it’s not characteristic of bases
Therefore, Option A is not a characteristic property of bases.
