Pre AP Chemistry -4.3D Representations of Acid–Base Reactions- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.3D Representations of Acid–Base Reactions- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.3D Representations of Acid–Base Reactions- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
The name ‘chlorate’ is used for an anion consisting of chlorine and oxygen only. In a molecule of ICl, the iodine atom has oxidation number x and the chlorine atom has oxidation number y. When ICl is added to \(H_2O\), iodine is reduced.
\(4ICl + 2H_2O \to 4HCl + O_2 + 2I_2\)
Which statement about the value of x or y is correct?
▶️ Answer/Explanation
Ans: A
Step 1: Determine oxidation numbers in ICl
- In \(ICl\), chlorine is more electronegative, so \(y = -1\).
- Since the molecule is neutral, iodine must have \(x = +1\).
Step 2: Analyze chlorate formation
- Cold NaOH: Forms \(ClO^–\) (hypochlorite), where Cl has an oxidation number of \(+1\).
- Hot NaOH: Forms \(ClO_3^–\) (chlorate), where Cl has an oxidation number of \(+5\).
Step 3: Compare with ICl
Since \(x = +1\) in \(ICl\), it matches the oxidation number of Cl in \(ClO^–\) (formed in cold NaOH). Thus, option A is correct.
Equations for some reactions of hydrogen peroxide are given.
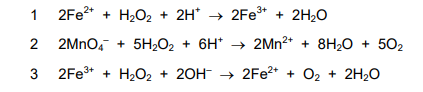
In which reactions is hydrogen peroxide acting as a reducing agent?
▶️ Answer/Explanation
Ans: C
Hydrogen peroxide acts as a reducing agent when it gets oxidized (loses electrons). In reaction 2, the oxygen oxidation state increases from -1 in \( \text{H}_2\text{O}_2 \) to 0 in \( \text{O}_2 \). In reaction 3, it increases from -1 to +2 in \( \text{PbO}_2 \). Reaction 1 shows reduction (acting as oxidizing agent). Thus, the correct answer is C (2 and 3).
Phosphorus reacts with concentrated sulfuric acid to produce phosphoric acid, sulfur dioxide and water.
aH₂SO₄ + bP → cH₃PO₄ + dSO₂ + eH₂O
a, b, c, d and e are all whole numbers. The equation can be balanced by using oxidation numbers. What is the value of the sum a + b + c + d + e?
A. 10
B. 14
C. 15
D. 16
▶️ Answer/Explanation
Ans: D
To balance the equation using oxidation numbers:
- Oxidation State Changes:
- Phosphorus (\(P\)) is oxidized from \(0\) to \(+5\) in \(H_3PO_4\).
- Sulfur (\(S\)) is reduced from \(+6\) in \(H_2SO_4\) to \(+4\) in \(SO_2\).
- Balancing Electrons:
- Each \(P\) loses \(5\) electrons.
- Each \(S\) gains \(2\) electrons.
- For charge balance, \(5\) electrons lost by \(P\) must equal those gained by \(S\). Thus, \(5P\) require \(5 \times 2 = 10\) electrons, meaning \(5S\) atoms are reduced.
- Balanced Equation: \[ 5H_2SO_4 + 2P \rightarrow 2H_3PO_4 + 5SO_2 + 2H_2O \] Here, \(a = 5\), \(b = 2\), \(c = 2\), \(d = 5\), and \(e = 2\).
- Sum Calculation: \[ a + b + c + d + e = 5 + 2 + 2 + 5 + 2 = 16 \]
Thus, the correct answer is D (16).
Nitrogen dioxide reacts with water.
\(2NO_2 + H_2O → HNO_2 + HNO_3\)
Which statement about this reaction is correct?
▶️ Answer/Explanation
Ans: B
In the reaction \(2NO_2 + H_2O → HNO_2 + HNO_3\), nitrogen changes its oxidation state from \(+4\) in \(NO_2\) to \(+3\) in \(HNO_2\) and \(+5\) in \(HNO_3\). This simultaneous oxidation and reduction of nitrogen is called disproportionation, making option B correct. Option A is incorrect because oxygen does not gain electrons. Option C is wrong as hydrogen’s oxidation state remains \(+1\). Option D is incorrect because water is not an oxidizing agent here.
Methyl orange is added to dilute hydrochloric acid and to aqueous sodium hydroxide. What is the colour of the methyl orange in each solution?
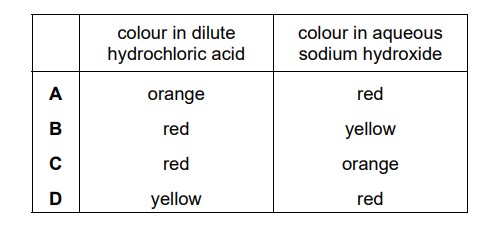
▶️ Answer/Explanation
Ans: B
Methyl orange is a pH indicator with the following color transitions:
- Red in strongly acidic solutions (pH < 3.1)
- Orange in weakly acidic solutions
- Yellow in basic solutions (pH > 4.4)
For the given solutions:
- Dilute hydrochloric acid (HCl):
- Strong acid with pH ≈ 1
- Methyl orange appears red
- Aqueous sodium hydroxide (NaOH):
- Strong base with pH ≈ 13-14
- Methyl orange appears yellow
The color change occurs because:
- In acid: The indicator exists in its protonated form (red)
- In base: It loses a proton to form the yellow conjugate base
Therefore, the correct color combination is:
- HCl solution: Red
- NaOH solution: Yellow
This corresponds to Option B in the answer choices.
Which row describes the colour changes when water is added to anhydrous cobalt(II) chloride and anhydrous copper(II) sulfate?
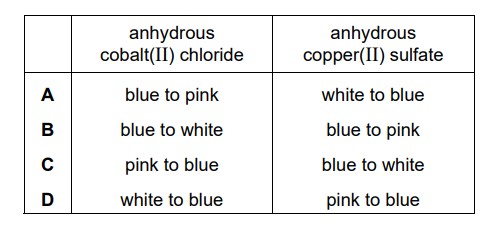
▶️ Answer/Explanation
Ans: A
Color Changes Explained:
1. Anhydrous Cobalt(II) Chloride (CoCl2):
- Initial color: Blue (anhydrous form)
- After adding water: Changes to Pink (forms hexahydrate CoCl2·6H2O)
- This is a reversible hydration reaction
2. Anhydrous Copper(II) Sulfate (CuSO4):
- Initial color: White/Colorless (anhydrous form)
- After adding water: Changes to Blue (forms pentahydrate CuSO4·5H2O)
- This change is also reversible
Why Other Options Are Incorrect:
- B: Incorrect cobalt chloride color change
- C: Incorrect copper sulfate color change
- D: Both color changes are wrong
Key Observations:
- Cobalt(II) chloride: Blue → Pink
- Copper(II) sulfate: White → Blue
- These changes are characteristic test for water
Therefore, the correct answer is A as it’s the only option that correctly describes both color changes.
What is a characteristic of acids?
A Acids turn methyl orange indicator yellow.
B Acids have a high pH value.
C Acids react with ammonium salts to give ammonia gas.
D Acids react with carbonates to produce salts.
▶️ Answer/Explanation
Ans: D
Analysis of each option:
| Option | Evaluation | Explanation |
|---|---|---|
| A | Incorrect | Acids turn methyl orange red (not yellow) |
| B | Incorrect | Acids have low pH values (pH < 7) |
| C | Incorrect | Bases (not acids) react with ammonium salts to produce NH3 |
| D | Correct | Acids react with carbonates to produce salt + water + CO2 |
Detailed Explanation for Option D:
- General reaction:
Acid + Carbonate → Salt + Water + Carbon Dioxide
2HCl + CaCO3 → CaCl2 + H2O + CO2
- This is a definitive test for acids (effervescence observed)
- Occurs because acids can donate H+ ions to react with CO32- ions
Why Other Options Are Incorrect:
- A: Methyl orange turns red in acids, yellow in bases
- B: Acids have pH < 7; bases have high pH
- C: NH3 production requires bases (e.g., NaOH + NH4Cl → NH3)
Therefore, the correct characteristic of acids is D – their reaction with carbonates to form salts.
Ammonium chloride is heated with aqueous sodium hydroxide.
A gas is produced which turns damp universal indicator paper blue.
Which gas has been produced?
A) Ammonia
B) Hydrogen
C) Oxygen
D) Sulfur dioxide
▶️ Answer/Explanation
Ans: A
1. Chemical Reaction:
NH4Cl + NaOH → NaCl + H2O + NH3↑
This is a classic test for ammonium salts 2. Indicator Test:
• Blue color indicates alkaline conditions (pH > 7)
• NH3 dissolves in water to form NH4OH (ammonium hydroxide)
NH3 + H2O ⇌ NH4+ + OH– 3. Why Not Other Gases:
• B (H2): Neutral (no color change)
• C (O2): Neutral (no color change)
• D (SO2): Acidic (turns indicator red)
4. Practical Identification:
Ammonia also has a characteristic pungent smell, confirming its presence
Some reactions of element M are shown.
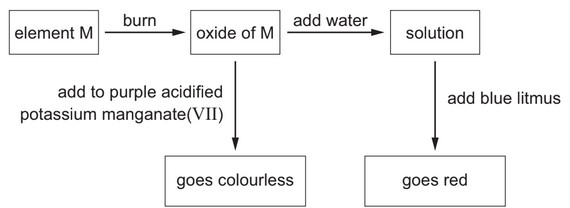
What is element M?
A carbon
B iron
C magnesium
D sulfur
▶️ Answer/Explanation
Ans: D
The reactions shown are characteristic of sulfur (S):
- With Oxygen:
- S(s) + O2(g) → SO2(g)
- Sulfur dioxide is a colorless, pungent gas
- With Acidified KMnO4:
- 5SO2 + 2MnO4– + 2H2O → 5SO42- + 4H+ + 2Mn2+
- Decolorizes purple permanganate solution
- With Water:
- SO2 + H2O ⇌ H2SO3 (sulfurous acid)
- Turns blue litmus red (acidic solution)
Why not other elements:
- Carbon: Forms CO2 which doesn’t decolorize KMnO4
- Iron/Magnesium: Don’t form gases that affect litmus paper this way
The complete reaction sequence matches sulfur’s chemical behavior perfectly.
Three separate experiments are carried out on an aqueous solution of S.
The results are shown.
1 Magnesium does not react with the solution.
2 A gas is given off when ammonium sulfate is heated with the solution.
3 Methyl orange turns yellow when added to the solution.
What is S?
A hydrochloric acid
B sodium hydroxide
C sodium chloride
D sulfur dioxide
▶️ Answer/Explanation
Ans: B (sodium hydroxide)
Analysis of each test result:
- Magnesium test:
- NaOH doesn’t react with Mg (explains observation)
- HCl would react vigorously with Mg
- Ammonium sulfate test:
- NaOH + (NH₄)₂SO₄ → NH₃(g) + Na₂SO₄ + H₂O
- Ammonia gas (NH₃) is produced
- Methyl orange test:
- Turns yellow in bases (pH > 4.4)
- NaOH is a strong base (pH ~14)
Only sodium hydroxide satisfies all three experimental results.
Hydrogen chloride gas reacts with water to produce an acidic solution. The equation for the reaction is shown.
\[ \mathrm{HCl} + \mathrm{H}_2 \mathrm{O} \rightarrow \mathrm{Cl}^- + \mathrm{H}_3 \mathrm{O}^+ \]
Which statement describes what happens during the reaction?
- The chloride ion is formed by accepting an electron from the water.
- The hydrogen chloride loses an electron to form the chloride ion.
- The water accepts a proton from the hydrogen chloride.
- The water donates a proton to the hydrogen chloride.
▶️ Answer/Explanation
Ans: C
Key Observations:
- Reaction Mechanism: In the reaction, \(\mathrm{HCl}\) donates a proton (\(\mathrm{H}^+\)) to \(\mathrm{H}_2\mathrm{O}\), forming \(\mathrm{H}_3\mathrm{O}^+\) (hydronium ion) and \(\mathrm{Cl}^-\).
- Role of Water: Water acts as a proton acceptor (Brønsted-Lowry base), not a proton donor.
- Why Other Options Are Incorrect:
- A: \(\mathrm{Cl}^-\) forms by releasing a proton, not accepting an electron.
- B: \(\mathrm{HCl}\) does not lose an electron; it loses a proton.
- D: Water accepts a proton, it does not donate one in this reaction.
Thus, the correct description is C: The water accepts a proton from the hydrogen chloride.
In which equation is the underlined reactant acting as a base?
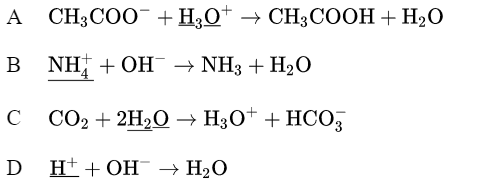
▶️ Answer/Explanation
Ans: C
A base is a substance that accepts protons (\(H^+\)). In option C, the underlined reactant \(NH_3\) accepts a proton from \(H_2O\) to form \(NH_4^+\), fulfilling the definition of a base. The other options involve proton donation (A, B) or no proton transfer (D), so they do not represent base behavior.
How many species are acting as bases in this reversible reaction?
\(HNO_{3}+H_{2}O\rightleftharpoons H_{3}O^{+}+NO_{3}^{-}\)
A. 3
B. 2
C. 1
D. 0
▶️ Answer/Explanation
Ans: B
In the reaction \(HNO_{3} + H_{2}O \rightleftharpoons H_{3}O^{+} + NO_{3}^{-}\):
1. \(H_{2}O\) acts as a base by accepting a proton (\(H^{+}\)) to form \(H_{3}O^{+}\).
2. \(NO_{3}^{-}\) acts as a base in the reverse reaction by accepting a proton to reform \(HNO_{3}\).
Thus, two species (\(H_{2}O\) and \(NO_{3}^{-}\)) function as bases.
Which statement is correct?
A) When anhydrous copper(II) sulfate is heated its colour changes to a deeper blue.
B) When hydrated copper(II) sulfate is heated its colour changes to a deeper blue.
C) When water is added to blue cobalt(II) chloride paper it turns pink.
D) When water is added to pink cobalt(II) chloride paper it turns blue.
▶️ Answer/Explanation
Ans: C
Analysis of each statement:
| Option | Chemical Process | Color Change | Validity |
|---|---|---|---|
| A | Anhydrous CuSO4 heated | White → White (no change) | False |
| B | Hydrated CuSO4 heated | Blue → White | False |
| C | Water added to CoCl2 paper | Blue → Pink | True |
| D | Water added to pink CoCl2 paper | Pink → Pink (no change) | False |
Detailed explanation for option C:
- Chemical reaction:
CoCl2 (anhydrous, blue) + 6H2O → CoCl2·6H2O (hydrated, pink) - Practical use:
- Cobalt chloride paper is blue when dry (anhydrous form)
- Turns pink when moist (hydrated form)
- Commonly used as humidity indicator
- Why other options are wrong:
- A/B: Copper sulfate turns white when heated, not blue
- D: Pink paper is already hydrated – water causes no further change
Therefore, C is the only correct statement.
A farmer’s soil is acidic.
Which substance should the farmer add to neutralise the soil?
A ammonium sulfate
B calcium oxide
C hydrochloric acid
D NPK fertiliser
▶️ Answer/Explanation
Ans: B
1. Neutralization requirements:
To neutralize acidic soil, a basic substance is needed.
2. Option analysis:
– A: Ammonium sulfate → acidic (forms H+ ions)
– B: Calcium oxide → strongly basic (reacts with acids)
– C: Hydrochloric acid → would increase acidity
– D: NPK fertilizer → neutral/nutrient source
3. Best choice:
Calcium oxide (quicklime) effectively neutralizes soil acidity through the reaction:
CaO + 2H+ → Ca2+ + H2O
Three elements, X, Y and Z, are burned in oxygen.
The oxides formed are dissolved in water and the pH of the solutions measured.
The results are shown.
Which statements are correct?
1 Element X could be sulfur.
2 Element Y could be sodium.
3 Element Z is a non-metal.
4 No metal elements were used.
A 1 only B 1 and 2 C 2 and 3 D 3 and 4
▶️ Answer/Explanation
Ans: B (1 and 2)
Analysis of the pH results and statements:
- Element X (pH 2 – strongly acidic):
- Non-metal oxides are typically acidic (SO2, CO2, NO2)
- Sulfur forms SO2 which creates acidic solution → Statement 1 correct
- Element Y (pH 13 – strongly basic):
- Group 1 metals (Na, K) form strongly basic oxides
- Sodium forms Na2O which creates alkaline solution → Statement 2 correct
- Element Z (pH 9 – weakly basic):
- Only metals form basic oxides → Statement 3 incorrect
- Could be a transition metal (e.g., Mg, Ca)
- Statement 4:
- Y and Z are metals → Statement 4 incorrect
Therefore, only statements 1 and 2 are correct, making Option B the right answer.
Solid X is tested as shown.

What is X?
- copper(II) carbonate
- lithium carbonate
- potassium carbonate
- sodium sulfate
▶️ Answer/Explanation
Ans: B
The test results indicate:
- Flame test: Red flame → characteristic of lithium compounds
- HCl reaction:
- Effervescence occurs (CO2 production)
- CO2 turns limewater milky
- This confirms carbonate presence
- NaOH test: No reaction → rules out copper(II) compounds
Analysis of options:
- A: Copper(II) carbonate would react with NaOH and give green flame
- B: Lithium carbonate matches all observations (red flame, CO2 production, no reaction with NaOH)
- C: Potassium carbonate would give lilac flame
- D: Sodium sulfate wouldn’t produce CO2
Thus, lithium carbonate (B) is the only compound matching all test results.
Barium hydroxide is an alkali. It reacts with hydrochloric acid.
How does the pH of the hydrochloric acid change as an excess of aqueous barium hydroxide is added?
- The pH decreases from pH14 and becomes constant at pH7.
- The pH decreases from pH14 to about pH1.
- The pH increases from pH1 and becomes constant at pH7.
- The pH increases from pH1 to about pH14.
▶️ Answer/Explanation
Ans: D
pH Change Analysis:
- Initial Condition:
- Hydrochloric acid (strong acid) has pH ≈ 1
- Barium hydroxide (strong base) has pH ≈ 14
- Neutralization Process:
- Ba(OH)2 + 2HCl → BaCl2 + 2H2O
- pH increases as base is added to acid
- At equivalence point (neutral): pH = 7
- Excess Base:
- After neutralization, excess Ba(OH)2 makes solution basic
- pH continues rising toward 14
The complete pH change follows this pattern:
- Starts at pH 1 (strong acid)
- Rises through neutral (pH 7) at equivalence point
- Ends near pH 14 (excess strong base)
Only Option D correctly describes this entire progression.
In the experiment shown, a white precipitate forms in the limewater.
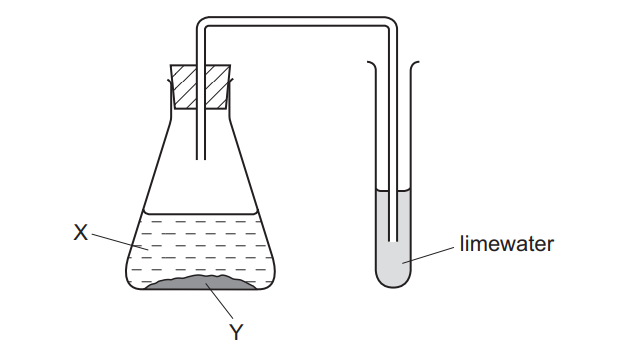
What are X and Y?

▶️ Answer/Explanation
Ans: D
This experiment demonstrates the test for carbon dioxide gas:
- Reaction in Flask X:
- Dilute sulfuric acid reacts with zinc carbonate: \[ ZnCO_3 + H_2SO_4 \rightarrow ZnSO_4 + H_2O + CO_2 \]
- Produces carbon dioxide gas (CO2)
- Test in Flask Y:
- CO2 bubbles through limewater (calcium hydroxide solution): \[ Ca(OH)_2 + CO_2 \rightarrow CaCO_3 \downarrow + H_2O \]
- Forms white precipitate of calcium carbonate
Key observations:
- X must be a carbonate (zinc carbonate in Option D)
- Y must be an acid (sulfuric acid in Option D)
- Only this combination produces CO2 that turns limewater milky
Therefore, the correct identification is:
- X: Zinc carbonate
- Y: Sulfuric acid
This corresponds to Option D.
Four substances, P, Q, R and S, are tested as shown.
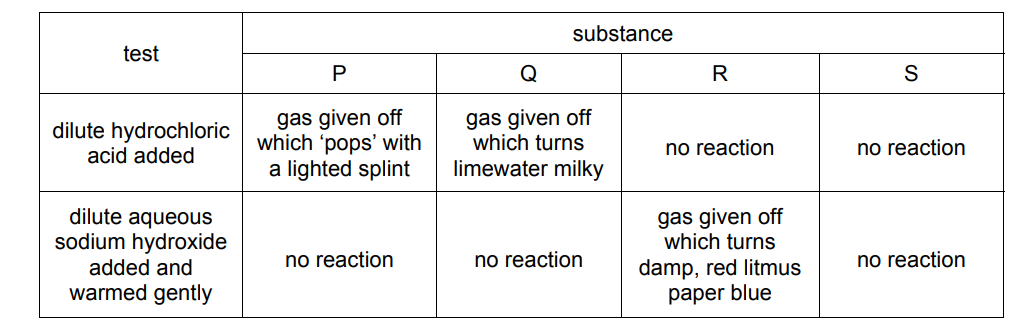
What are P, Q, R and S?
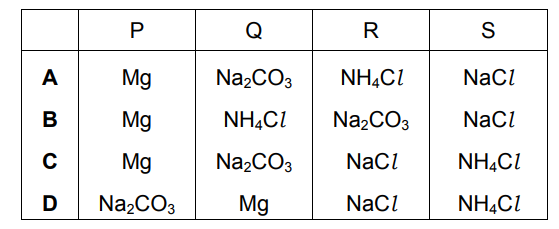
▶️ Answer/Explanation
Ans: A
Identification of Substances:
P: Magnesium (Mg)
- Reacts with HCl: Produces H₂ gas (pop sound)
- No reaction with NaOH: Mg is below Na in reactivity series
- Equation: Mg + 2HCl → MgCl₂ + H₂↑
Q: Sodium Carbonate (Na₂CO₃)
- Reacts with HCl: Produces CO₂ (turns limewater milky)
- No reaction with NaOH: Both are bases
- Equation: Na₂CO₃ + 2HCl → 2NaCl + H₂O + CO₂↑
R: Ammonium Chloride (NH₄Cl)
- Reacts with NaOH: Produces NH₃ gas (turns red litmus blue)
- No reaction with HCl: Already an acidic salt
- Equation: NH₄Cl + NaOH → NaCl + H₂O + NH₃↑
S: Sodium Chloride (NaCl)
- No reaction with HCl or NaOH: Chemically inert salt
Key Observations:
- Only magnesium produces hydrogen gas with HCl
- Only sodium carbonate produces CO₂ that turns limewater milky
- Only ammonium chloride produces ammonia with NaOH
- Sodium chloride shows no reactions
Therefore, the correct identification is A (Mg, Na₂CO₃, NH₄Cl, NaCl).
Part of the Periodic Table is shown. Which element forms an oxide that reacts with dilute acid to form a salt and water?
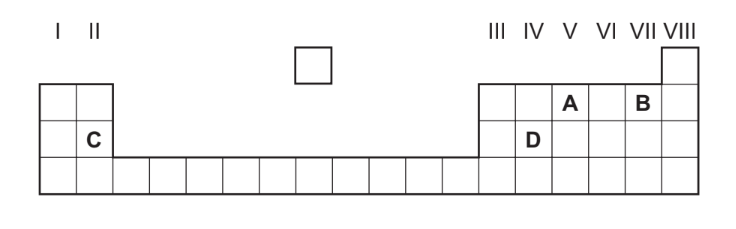
▶️ Answer/Explanation
Ans: C
Key Concept: Basic oxides (typically from Groups 1-2 metals) react with acids in neutralization reactions.
Analysis of Options:
| Option | Element | Oxide Type | Reaction with Acid |
|---|---|---|---|
| A | Carbon (C) | Acidic (CO2) | No salt formation |
| B | Nitrogen (N) | Acidic (NO2) | No salt formation |
| C | Magnesium (Mg) | Basic (MgO) | Forms salt + water |
| D | Silicon (Si) | Amphoteric (SiO2) | Complex reaction |
Detailed Explanation for Option C:
- Magnesium (Group 2 metal) forms MgO – a basic oxide
- Neutralization reaction with acid:
MgO + 2HCl → MgCl2 + H2O
- Characteristics:
- Forms ionic salt (MgCl2)
- Water as byproduct
- Exothermic reaction
Why Other Options Are Incorrect:
- A/B: Non-metal oxides are acidic (don’t neutralize acids)
- D: SiO2 is amphoteric but requires strong bases/acids
Therefore, C (Magnesium) is the correct answer as it forms a basic oxide that readily neutralizes acids.
