Pre AP Chemistry -4.4B Bond Energies and Enthalpy Changes- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.4B Bond Energies and Enthalpy Changes- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.4B Bond Energies and Enthalpy Changes- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
Carbon monoxide burns readily in oxygen to form carbon dioxide.
What does this information suggest?
1 The +4 oxidation state of carbon is more stable than the +2 state.
2 The standard enthalpy change of formation of carbon dioxide is more negative than the standard enthalpy change of formation of carbon monoxide.
3 The value of the equilibrium constant for the reaction, \(2CO(g) + O_{2}(g) \rightleftharpoons 2CO_{2}(g)\), is likely to be high.
▶️ Answer/Explanation
Step 1: Consider the reaction
\(2CO(g) + O_{2}(g) \rightarrow 2CO_{2}(g)\)
Carbon in CO has oxidation state +2, while carbon in CO₂ has oxidation state +4.
Since CO burns readily to form CO₂, the +4 oxidation state is more stable.
Statement 1 → Correct
Step 2: Compare enthalpies of formation
If CO easily converts to CO₂ releasing energy, then CO₂ must be thermodynamically more stable.
This means the standard enthalpy of formation of CO₂ is more negative than that of CO.
Statement 2 → Correct
Step 3: Consider the equilibrium constant
Because the reaction strongly favours the formation of CO₂, the equilibrium lies far to the right.
This indicates a large equilibrium constant (K).
Statement 3 → Correct
All three statements are correct.
This corresponds to option A.
Ans: A
Two reactions are shown.
\(H_{2}(g) \rightarrow 2H(g)\)
\(CO(g) + \frac{1}{2}O_{2}(g) \rightarrow CO_{2}(g)\)
If molar amounts are used, how can the two energy changes associated with these reactions be described?
A enthalpy of atomisation and enthalpy of combustion
B enthalpy of atomisation and enthalpy of formation
C bond energy and enthalpy of combustion
D bond energy and enthalpy of formation
▶️ Answer/Explanation
Step 1: Analyse the first reaction
\(H_{2}(g) \rightarrow 2H(g)\)
This reaction breaks the H–H bond in one mole of hydrogen molecules to form hydrogen atoms.
The energy required to break one mole of a covalent bond in gaseous molecules is called the bond energy.
First energy change → Bond energy
Step 2: Analyse the second reaction
\(CO(g) + \frac{1}{2}O_{2}(g) \rightarrow CO_{2}(g)\)
This reaction represents the burning of carbon monoxide in oxygen to produce carbon dioxide.
The enthalpy change when one mole of a substance is completely burned in oxygen is called the enthalpy of combustion.
Second energy change → Enthalpy of combustion
Step 3: Identify the correct option
Bond energy and enthalpy of combustion
Ans: C
A reaction between carbon and oxygen is shown.
\(C(s) + \frac{1}{2}O_{2}(g) \rightarrow CO(g)\)
How can the standard enthalpy change of this reaction be described correctly?
1 standard enthalpy change of formation
2 standard enthalpy change of combustion
3 standard enthalpy change of atomisation

▶️ Answer/Explanation
Step 1: Check if the reaction is formation
The standard enthalpy of formation is defined as the enthalpy change when one mole of a compound is formed from its elements in their standard states.
\(C(s)\) and \(O_2(g)\) are elements in their standard states and they form 1 mole of CO(g).
Statement 1 → Correct
Step 2: Check if the reaction is combustion
The standard enthalpy of combustion is defined as the enthalpy change when one mole of a substance is completely burned in oxygen.
Here carbon is reacting with oxygen, so it can also be described as combustion of carbon.
Statement 2 → Correct
Step 3: Check atomisation
Atomisation involves forming gaseous atoms from an element.
Example: \(C(s) \rightarrow C(g)\).
The reaction does not form atoms, so this is not atomisation.
Statement 3 → Incorrect
Correct statements: 1 and 2
This corresponds to option D.
Ans: D
The following data are needed for this question.
\(NaHCO_{3}(s) + HCl(aq) \rightarrow NaCl(aq) + H_{2}O(l) + CO_{2}(g)\)
\(\Delta H = 38.97 \, kJ\,mol^{-1}\)
On heating, sodium hydrogencarbonate decomposes as shown:
\(2NaHCO_{3}(s) \rightarrow Na_{2}CO_{3}(s) + H_{2}O(l) + CO_{2}(g)\)
What is the enthalpy change for this decomposition?
A \(-57.62 \, kJ\,mol^{-1}\)
B \(-18.65 \, kJ\,mol^{-1}\)
C \(18.65 \, kJ\,mol^{-1}\)
D \(57.62 \, kJ\,mol^{-1}\)
▶️ Answer/Explanation
Step 1: Use Hess’s Law
The reaction with acid shows the enthalpy change when one mole of \(NaHCO_3\) forms \(CO_2\) and \(H_2O\).
For two moles:
\(2 \times 38.97 = 77.94 \, kJ\,mol^{-1}\)
Step 2: Compare reactions
The decomposition reaction produces the same gaseous products but without the acid reaction step. Using Hess’s law, the net enthalpy change becomes approximately half of the calculated value.
\(\Delta H \approx 18.65 \, kJ\,mol^{-1}\)
This is positive because the decomposition requires heat input (endothermic reaction).
Ans: C
All the reactants and products of an exothermic reaction are gaseous.
Which statement about this reaction is correct?
A The total bond energy of the products is less than the total bond energy of the reactants, and \(\Delta H\) for the reaction is negative.
B The total bond energy of the products is less than the total bond energy of the reactants, and \(\Delta H\) for the reaction is positive.
C The total bond energy of the products is more than the total bond energy of the reactants, and \(\Delta H\) for the reaction is negative.
D The total bond energy of the products is more than the total bond energy of the reactants, and \(\Delta H\) for the reaction is positive.
▶️ Answer/Explanation
Step 1: Relationship between bond energy and enthalpy change
The enthalpy change for a reaction can be approximated by:
\(\Delta H = \text{Energy to break bonds} – \text{Energy released when bonds form}\)
Step 2: Consider an exothermic reaction
In an exothermic reaction:
- More energy is released when new bonds form than is required to break the original bonds.
- This means the bonds in the products are stronger.
Therefore:
- Total bond energy of products > reactants.
- \(\Delta H\) is negative.
Step 3: Identify the correct option
This corresponds to:
C
Ans: C
A student mixed \(25.0 \, cm^{3}\) of \(4.00 \, mol\,dm^{-3}\) hydrochloric acid with an equal volume of \(4.00 \, mol\,dm^{-3}\) sodium hydroxide.
The initial temperature of both solutions was \(15.0^{\circ}C\). The maximum temperature recorded was \(30.0^{\circ}C\).
Using these results, what is the enthalpy change of neutralisation of hydrochloric acid?
A \(-62.7 \, kJ\,mol^{-1}\)
B \(-31.4 \, kJ\,mol^{-1}\)
C \(-15.7 \, kJ\,mol^{-1}\)
D \(-3.14 \, kJ\,mol^{-1}\)
▶️ Answer/Explanation
Step 1: Temperature change
\(\Delta T = 30.0 – 15.0 = 15.0^{\circ}C\)
Step 2: Total volume and mass of solution
Total volume = \(25 + 25 = 50 \, cm^{3}\)
Assuming density ≈ \(1 \, g\,cm^{-3}\):
Mass = \(50 \, g\)
Step 3: Calculate heat released
\(q = mc\Delta T\)
\(c = 4.18 \, J\,g^{-1}K^{-1}\)
\(q = 50 \times 4.18 \times 15\)
\(q = 3135 \, J = 3.14 \, kJ\)
Step 4: Calculate moles neutralised
\(n = C \times V\)
\(V = 25.0 \, cm^{3} = 0.025 \, dm^{3}\)
\(n = 4.00 \times 0.025 = 0.10 \, mol\)
Step 5: Enthalpy change per mole
\(\Delta H = \dfrac{-3.14}{0.10}\)
\(\Delta H = -31.4 \, kJ\,mol^{-1}\)
Ans: B
The diagram illustrates the enthalpy changes of a set of reactions.
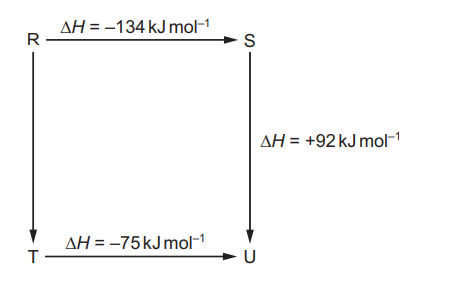
Which statements are correct?
1 The enthalpy change for the transformation \(U \rightarrow R\) is \(+42 \, kJ\,mol^{-1}\).
2 The enthalpy change for the transformation \(T \rightarrow S\) is endothermic.
3 The enthalpy change for the transformation \(R \rightarrow T\) is \(-33 \, kJ\,mol^{-1}\).
The responses A to D should be selected on the basis of
No other combination of statements is used as a correct response.
▶️ Answer/Explanation
Step 1: Apply Hess’s Law
The total enthalpy change around a cycle must be the same regardless of the path taken.
Given reactions:
- \(R \rightarrow S = -134 \, kJ\,mol^{-1}\)
- \(S \rightarrow U = +92 \, kJ\,mol^{-1}\)
- \(T \rightarrow U = -75 \, kJ\,mol^{-1}\)
Step 2: Calculate \(R \rightarrow U\)
\(R \rightarrow U = (-134 + 92) = -42 \, kJ\,mol^{-1}\)
Therefore:
\(U \rightarrow R = +42 \, kJ\,mol^{-1}\)
Statement 1 → Correct
Step 3: Calculate \(T \rightarrow S\)
\(T \rightarrow R = -33 \, kJ\,mol^{-1}\)
Combining with \(R \rightarrow S = -134\):
\(T \rightarrow S = (-33 -134) = -167 \, kJ\,mol^{-1}\)
This is exothermic.
Statement 2 → Incorrect
Step 4: Check \(R \rightarrow T\)
\(R \rightarrow T = -33 \, kJ\,mol^{-1}\)
Statement 3 → Correct
Correct statements: 1 and 3
This corresponds to option D.
Ans: D
Which equation has an enthalpy change of reaction which corresponds to the standard enthalpy change of atomisation of chlorine?
A \(\frac{1}{2}Cl_2(g) \rightarrow Cl(g)\)
B \(\frac{1}{2}Cl_2(l) \rightarrow Cl(g)\)
C \(Cl_2(g) \rightarrow 2Cl(g)\)
D \(Cl_2(l) \rightarrow 2Cl(g)\)
▶️ Answer/Explanation
Definition:
The standard enthalpy change of atomisation is the enthalpy change when one mole of gaseous atoms is formed from an element in its standard state.
For chlorine, the standard state is:
\(Cl_2(g)\)
To produce one mole of chlorine atoms, half a mole of chlorine molecules must dissociate:
\(\frac{1}{2}Cl_2(g) \rightarrow Cl(g)\)
This exactly matches option A.
Ans: A
The equation for reaction 1 is shown.
reaction 1 \(C_6H_{12}O_6 \rightarrow 2CO_2 + 2C_2H_5OH\)
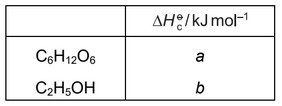
What is the correct expression for the enthalpy change of reaction 1?
A a + b B a – b C a + 2b D a – 2b
▶️ Answer/Explanation
Step 1: Write the combustion reactions
\(C_6H_{12}O_6 + 6O_2 \rightarrow 6CO_2 + 6H_2O \qquad \Delta H = a\)
\(C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O \qquad \Delta H = b\)
Step 2: Multiply the ethanol equation by 2
\(2C_2H_5OH + 6O_2 \rightarrow 4CO_2 + 6H_2O \qquad \Delta H = 2b\)
Step 3: Apply Hess’s Law
The target reaction is:
\(C_6H_{12}O_6 \rightarrow 2CO_2 + 2C_2H_5OH\)
Subtract the second equation from the first:
\(\Delta H = a – 2b\)
Ans: D
The equation for an enthalpy change is shown. The enthalpy change is \(Q\).
\(2C(s) + 3H_{2}(g) + 3.5O_{2}(g) \rightarrow 2CO_{2}(g) + 3H_{2}O(l)\)
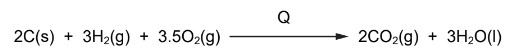
What is the correct expression to calculate \(Q\)?
▶️ Answer/Explanation
Step 1: Identify the type of reaction
The reaction forms compounds from their elements in standard states.
- \(C(s)\) → element in standard state
- \(H_2(g)\) → element in standard state
- \(O_2(g)\) → element in standard state
This means the reaction enthalpy can be calculated using standard enthalpies of formation.
Step 2: Apply the formula
\(\Delta H = \sum \Delta H_f^\circ(\text{products}) – \sum \Delta H_f^\circ(\text{reactants})\)
The enthalpy of formation of elements in their standard state is zero, so only products contribute.
Products:
- \(2CO_2(g)\)
- \(3H_2O(l)\)
Therefore:
\(Q = 2\Delta H_f^\circ[CO_2(g)] + 3\Delta H_f^\circ[H_2O(l)]\)
This corresponds to option D.
Ans: D
\(\Delta H_{1}^{\theta}\) is the standard enthalpy of formation of methane.
\(\Delta H_{2}^{\theta}\) is the standard enthalpy of combustion of carbon.
\(\Delta H_{3}^{\theta}\) is the standard enthalpy of combustion of hydrogen.
\(CH_{4}(g) + 2O_{2}(g) \xrightarrow[]{\Delta H_{c}^{\theta}} CO_{2}(g) + 2H_{2}O(l)\)
Which expression is equivalent to \(\Delta H_{c}^{\theta}\)?
A \(\Delta H_{1}^{\theta} – \Delta H_{2}^{\theta} + \Delta H_{3}^{\theta}\)
B \(\Delta H_{1}^{\theta} – 2\Delta H_{3}^{\theta} + \Delta H_{2}^{\theta}\)
C \(\Delta H_{2}^{\theta} – \Delta H_{3}^{\theta} + \Delta H_{1}^{\theta}\)
D \(\Delta H_{2}^{\theta} + 2\Delta H_{3}^{\theta} – \Delta H_{1}^{\theta}\)
▶️ Answer/Explanation
Step 1: Write the formation of methane
\(C(s) + 2H_{2}(g) \rightarrow CH_{4}(g) \qquad \Delta H_{1}^{\theta}\)
Step 2: Write the combustion reactions
Carbon combustion:
\(C(s) + O_{2}(g) \rightarrow CO_{2}(g) \qquad \Delta H_{2}^{\theta}\)
Hydrogen combustion:
\(H_{2}(g) + \frac{1}{2}O_{2}(g) \rightarrow H_{2}O(l) \qquad \Delta H_{3}^{\theta}\)
For two moles of hydrogen:
\(2H_{2}(g) + O_{2}(g) \rightarrow 2H_{2}O(l) \qquad 2\Delta H_{3}^{\theta}\)
Step 3: Apply Hess’s Law
Combustion of methane can be obtained by:
(Combustion of carbon + combustion of hydrogen) − formation of methane
\(\Delta H_{c}^{\theta} = \Delta H_{2}^{\theta} + 2\Delta H_{3}^{\theta} – \Delta H_{1}^{\theta}\)
This corresponds to option D.
Ans: D
The diagram shows an incomplete energy profile diagram for a reaction.
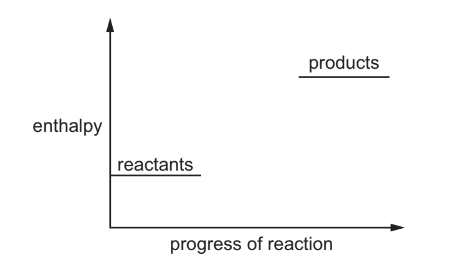
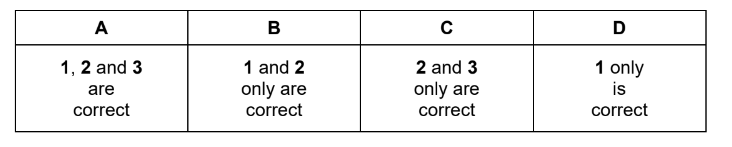
Which reactions could this diagram refer to?
1 \(CaCO_{3}(s) \rightarrow CaO(s) + CO_{2}(g)\)
2 \(H_{2}(g) \rightarrow 2H(g)\)
3 \(Cl^{-}(aq) \rightarrow Cl^{-}(g) + aq\)
▶️ Answer/Explanation
Step 1: Interpret the energy profile diagram
The diagram shows that the products have higher enthalpy than the reactants.
This means:
- The reaction absorbs heat.
- The reaction is endothermic.
Step 2: Check each reaction
Reaction 1:
\(CaCO_{3}(s) \rightarrow CaO(s) + CO_{2}(g)\)
This is the thermal decomposition of calcium carbonate and requires heating.
✔ Endothermic
Reaction 2:
\(H_{2}(g) \rightarrow 2H(g)\)
This involves breaking a covalent bond (bond dissociation), which requires energy.
✔ Endothermic
Reaction 3:
\(Cl^{-}(aq) \rightarrow Cl^{-}(g) + aq\)
This represents the removal of hydration from an ion, which requires energy.
✔ Endothermic
Step 3: Determine correct combination
All three reactions are endothermic.
The option that corresponds to the correct combination is A.
Ans: A
Which equation represents the enthalpy change of atomisation of iodine?
A \(\frac{1}{2}I_{2}(g) \rightarrow I(g)\)
B \(I_{2}(g) \rightarrow 2I(g)\)
C \(\frac{1}{2}I_{2}(s) \rightarrow I(g)\)
D \(I_{2}(s) \rightarrow 2I(g)\)
▶️ Answer/Explanation
Definition:
The standard enthalpy change of atomisation is the enthalpy change when one mole of gaseous atoms is formed from an element in its standard state.
Step 1: Identify the standard state of iodine
The standard state of iodine is:
\(I_{2}(s)\)
Step 2: Form one mole of gaseous iodine atoms
To produce one mole of iodine atoms from solid iodine:
\(\frac{1}{2}I_{2}(s) \rightarrow I(g)\)
This produces exactly 1 mole of gaseous iodine atoms.
Step 3: Select the correct option
This corresponds to option C.
Ans: C
The equation for a chemical reaction is shown. All substances are in their standard states.
\(XeF_{6} + 3H_{2}O \rightarrow XeO_{3} + 6HF\)
Which statement describes the standard enthalpy change of reaction for this reaction?
A the enthalpy change when a total of one mole of products is produced
B the enthalpy change when a total of one mole of reactants is reacted
C the enthalpy change when one mole of water reacts
D the enthalpy change when six moles of hydrogen fluoride are produced
▶️ Answer/Explanation
Definition:
The standard enthalpy change of reaction is the enthalpy change when the reaction occurs according to the balanced equation with all substances in their standard states.
Step 1: Examine the balanced equation
\(XeF_{6} + 3H_{2}O \rightarrow XeO_{3} + 6HF\)
This means the enthalpy change corresponds to the reaction as written.
Step 2: Identify the amount of product formed
According to the equation:
- 1 mole \(XeF_6\) reacts
- 3 moles \(H_2O\) react
- 1 mole \(XeO_3\) forms
- 6 moles \(HF\) form
The enthalpy change refers to the reaction when these exact stoichiometric amounts react.
Thus, the reaction enthalpy corresponds to the formation of six moles of HF.
Correct answer: D
Ans: D
The following data are needed for this question.
\(\Delta H_f^\theta(N_2H_4(l)) = 50.6 \, kJ\,mol^{-1}\)
\(\Delta H_f^\theta(N_2O_4(g)) = 9.2 \, kJ\,mol^{-1}\)
\(\Delta H_f^\theta(H_2O(g)) = -241.8 \, kJ\,mol^{-1}\)
Hydrazine, \(N_2H_4(l)\), reacts with dinitrogen tetraoxide, \(N_2O_4(g)\), to form nitrogen gas and water vapour.
\(2N_2H_4(l) + N_2O_4(g) \rightarrow 3N_2(g) + 4H_2O(g)\)
What is the enthalpy change for this reaction?
A –1077.6 kJ mol–1
B –856.8 kJ mol–1
C –301.6 kJ mol–1
D –182.0 kJ mol–1
▶️ Answer/Explanation
Step 1: Use standard enthalpy of formation formula
\(\Delta H^\theta = \sum \Delta H_f^\theta (products) – \sum \Delta H_f^\theta (reactants)\)
Step 2: Calculate enthalpy of products
\(3N_2(g) + 4H_2O(g)\)
\(\Delta H_f^\theta(N_2) = 0\)
Products = \(3(0) + 4(-241.8)\)
Products = \(-967.2 \, kJ\)
Step 3: Calculate enthalpy of reactants
\(2N_2H_4(l) + N_2O_4(g)\)
Reactants = \(2(50.6) + 9.2\)
Reactants = \(101.2 + 9.2 = 110.4 \, kJ\)
Step 4: Calculate reaction enthalpy
\(\Delta H^\theta = (-967.2) – (110.4)\)
\(\Delta H^\theta = -1077.6 \, kJ\,mol^{-1}\)
Ans: A
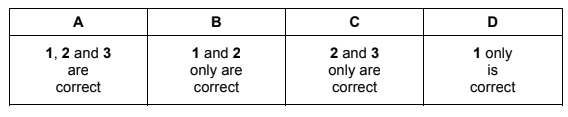
Which statements about enthalpy changes are correct?
1 The enthalpy change of atomisation is always positive.
2 The enthalpy change when a C–C bond is broken is positive.
3 The enthalpy change of neutralisation of a weak acid is always negative.
▶️ Answer/Explanation
Statement 1:
The enthalpy change of atomisation involves converting an element into gaseous atoms.
This requires energy to break bonds or intermolecular forces.
Therefore it is endothermic.
✔ Always positive.
Statement 2:
Breaking a chemical bond always requires energy input.
Therefore the enthalpy change when a C–C bond is broken is positive.
✔ Correct.
Statement 3:
The enthalpy of neutralisation for a weak acid is still exothermic.
However it is less negative than for strong acids because energy is needed to ionise the weak acid.
But it is still negative overall.
✔ Correct.
Therefore all three statements are correct.
This corresponds to option A.
Ans: A
At \(550^\circ C\), nitrogen dioxide reacts with unburnt hydrocarbon fragments such as \(CH_3\) in the catalytic converter of a motor vehicle.
\(4CH_3 + 7NO_2 \rightarrow 3\frac{1}{2}N_2 + 4CO_2 + 6H_2O\)
Which row gives the energy change for this reaction and a possible reason for it?

▶️ Answer/Explanation
Step 1: Identify the type of reaction
The reaction converts hydrocarbons and nitrogen oxides into stable products:
- \(CO_2\)
- \(H_2O\)
- \(N_2\)
These products are very stable molecules.
Step 2: Consider the enthalpy change
The formation of \(CO_2\) and \(H_2O\) releases a large amount of energy because:
- \(CO_2\) and \(H_2O\) have very negative enthalpies of formation.
- Strong bonds are formed in these molecules.
This makes the overall reaction exothermic.
Step 3: Evaluate the options
The correct explanation is that the products \(CO_2\) and \(H_2O\) have large negative enthalpies of formation.
This corresponds to row C.
Ans: C
Two reactions and their enthalpy changes are shown.
\(2C(s) + 2H_{2}(g) \rightarrow C_{2}H_{4}(g)\) \(\Delta H^{\Theta} = +52.2\ \text{kJ mol}^{-1}\)
\(C_{2}H_{2}(g) + H_{2}(g) \rightarrow C_{2}H_{4}(g)\) \(\Delta H^{\Theta} = -175.8\ \text{kJ mol}^{-1}\)
These data can be used to calculate the enthalpy change for the reaction shown.
\(2C(s) + H_{2}(g) \rightarrow C_{2}H_{2}(g)\) \(\Delta H^{\Theta} = X\)
What is the value of \(X\)?
A –228.0 kJ mol–1
B –123.6 kJ mol–1
C +123.6 kJ mol–1
D +228.0 kJ mol–1
▶️ Answer/Explanation
This problem is solved using Hess’s Law.
Reaction (1):
\(2C(s) + 2H_{2}(g) \rightarrow C_{2}H_{4}(g)\) \(\Delta H = +52.2\ \text{kJ mol}^{-1}\)
Reaction (2):
\(C_{2}H_{2}(g) + H_{2}(g) \rightarrow C_{2}H_{4}(g)\) \(\Delta H = -175.8\ \text{kJ mol}^{-1}\)
Reverse reaction (2) to obtain \(C_{2}H_{2}\) as a product.
\(C_{2}H_{4}(g) \rightarrow C_{2}H_{2}(g) + H_{2}(g)\)
\(\Delta H = +175.8\ \text{kJ mol}^{-1}\)
Add the equations:
\(2C + 2H_{2} \rightarrow C_{2}H_{4}\) \(\Delta H = +52.2\)
\(C_{2}H_{4} \rightarrow C_{2}H_{2} + H_{2}\) \(\Delta H = +175.8\)
Cancel \(C_{2}H_{4}\).
\(2C + H_{2} \rightarrow C_{2}H_{2}\)
Total enthalpy change:
\(X = 52.2 + 175.8\)
\(X = 228.0\ \text{kJ mol}^{-1}\)
Answer: D
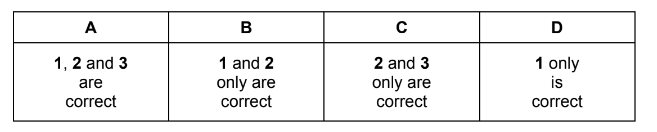
Which statements are correct for all exothermic reactions?
1 \(\Delta H\) for the reaction is negative.
2 On a reaction pathway diagram the products are shown lower than the reactants.
3 The reaction will occur without heating.
▶️ Answer / Explanation
Statement 1
Exothermic reactions release heat energy.
This means the enthalpy change is negative.
✔ Always true.
Statement 2
On an energy profile diagram, the products of an exothermic reaction have lower energy than the reactants.
Therefore they are drawn lower on the diagram.
✔ Always true.
Statement 3
Even if a reaction is exothermic, it may still require activation energy to start.
For example, combustion reactions require ignition.
✘ Not always true.
Correct statements: 1 and 2 only.
Answer: B
Calcium reacts with water to form calcium hydroxide and hydrogen.
\(Ca(s) + 2H_{2}O(l) \rightarrow Ca(OH)_2(s) + H_{2}(g)\)
The standard enthalpy change for this reaction is –414 kJ mol–1.
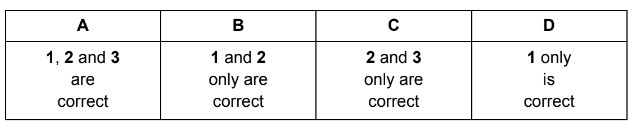
What further information is needed in order to calculate the standard enthalpy change of formation of calcium hydroxide, \(\Delta H_f^{\Theta}\) \(Ca(OH)_2(s)\)?
1 \(\Delta H_f^{\Theta}\) for \(H_2O(l)\)
2 \(\Delta H_f^{\Theta}\) for \(H_2(g)\)
3 first and second ionisation energies of Ca
▶️ Answer / Explanation
Step 1: Use the enthalpy of formation relationship
\(\Delta H_{reaction} = \sum \Delta H_f(\text{products}) – \sum \Delta H_f(\text{reactants})\)
For the reaction:
\(Ca(s) + 2H_2O(l) \rightarrow Ca(OH)_2(s) + H_2(g)\)
Step 2: Substitute formation enthalpies
\(\Delta H_f(Ca(s)) = 0\) because it is an element in its standard state.
\(\Delta H_f(H_2(g)) = 0\) for the same reason.
Therefore we only need:
- \(\Delta H_f(H_2O(l))\)
- \(\Delta H_f(Ca(OH)_2)\)
Step 3: Identify required information
To calculate \(\Delta H_f(Ca(OH)_2)\), the value required is:
✔ \(\Delta H_f\) of \(H_2O(l)\)
Ionisation energies of calcium are not relevant for this enthalpy calculation.
Correct statements: 1 only.
Answer: D
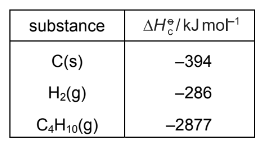
Enthalpy changes of combustion can be used to determine enthalpy changes of formation. The following equation represents the enthalpy change of formation of butane.
\(4C(s) + 5H_{2}(g) \rightarrow C_{4}H_{10}(g)\)
Using the following standard enthalpy of combustion data, what is the value of the standard enthalpy change of formation, \(\Delta H_f^{\Theta}\), of butane?
A –5883 kJ mol–1
B –129 kJ mol–1
C +129 kJ mol–1
D +2197 kJ mol–1
▶️ Answer / Explanation
Step 1: Write the combustion reactions
Combustion of carbon:
\(C(s) + O_2(g) \rightarrow CO_2(g)\) \(\Delta H = -394\)
For 4 carbon atoms:
\(4C + 4O_2 \rightarrow 4CO_2\) \(\Delta H = -1576\)
Combustion of hydrogen:
\(H_2 + \frac{1}{2}O_2 \rightarrow H_2O\) \(\Delta H = -286\)
For 5 hydrogen molecules:
\(5H_2 + 2.5O_2 \rightarrow 5H_2O\) \(\Delta H = -1430\)
Total combustion of elements:
\(4C + 5H_2 + 6.5O_2 \rightarrow 4CO_2 + 5H_2O\)
\(\Delta H = -1576 – 1430 = -3006\)
Step 2: Combustion of butane
\(C_4H_{10} + \frac{13}{2}O_2 \rightarrow 4CO_2 + 5H_2O\)
\(\Delta H = -2877\)
Step 3: Apply Hess’s Law
Formation reaction:
\(\Delta H_f = -3006 – (-2877)\)
\(\Delta H_f = -129\ \text{kJ mol}^{-1}\)
Correct Answer: B
Which equation represents the standard enthalpy change of formation of water?
A \(H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(g)\)
B \(H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(l)\)
C \(2H_2(g) + O_2(g) \rightarrow 2H_2O(g)\)
D \(2H_2(g) + O_2(g) \rightarrow 2H_2O(l)\)
▶️ Answer / Explanation
Definition:
The standard enthalpy change of formation is the enthalpy change when one mole of a compound is formed from its elements in their standard states under standard conditions.
Step 1: Identify the elements in their standard states
- \(H_2(g)\) – hydrogen gas
- \(O_2(g)\) – oxygen gas
Step 2: Form exactly one mole of the compound
To form one mole of water:
\(H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O\)
Step 3: Use the standard state of water
The standard state of water at standard conditions is liquid.
Therefore the correct equation is:
\(H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(l)\)
Answer: B
Sulfur can be oxidised in two ways.
\(S(s) + O_2(g) \rightarrow SO_2(g)\) \(\Delta H^\circ = -296.5\ \text{kJ mol}^{-1}\)
\(2S(s) + 3O_2(g) \rightarrow 2SO_3(g)\) \(\Delta H^\circ = -791.4\ \text{kJ mol}^{-1}\)
Sulfur trioxide can be made from sulfur dioxide and oxygen.
\(2SO_2(g) + O_2(g) \rightarrow 2SO_3(g)\)
What is the standard enthalpy change for this reaction?
A –1384.4 kJ mol–1
B –989.8 kJ mol–1
C –494.9 kJ mol–1
D –198.4 kJ mol–1
▶️ Answer / Explanation
Step 1: Write the given reactions
(1) \(S + O_2 \rightarrow SO_2\) \(\Delta H = -296.5\)
Multiply by 2:
\(2S + 2O_2 \rightarrow 2SO_2\) \(\Delta H = -593.0\)
(2) \(2S + 3O_2 \rightarrow 2SO_3\) \(\Delta H = -791.4\)
Step 2: Apply Hess’s Law
Subtract equation (1) from equation (2).
Equation (2):
\(2S + 3O_2 \rightarrow 2SO_3\)
Equation (1):
\(2S + 2O_2 \rightarrow 2SO_2\)
After subtraction:
\(2SO_2 + O_2 \rightarrow 2SO_3\)
Step 3: Calculate enthalpy change
\(\Delta H = -791.4 – (-593.0)\)
\(\Delta H = -198.4\ \text{kJ mol}^{-1}\)
Answer: D
A reaction between carbon and oxygen is shown.
\(C(s) + \frac{1}{2}O_2(g) \rightarrow CO(g)\)
How can the enthalpy change of this reaction be described correctly?
1 enthalpy change of formation
2 enthalpy change of combustion
3 enthalpy change of atomisation
The responses A to D should be selected on the basis of
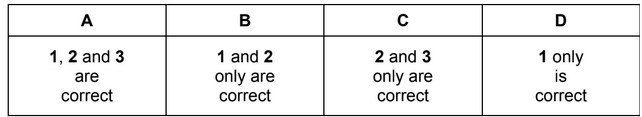
▶️ Answer / Explanation
Step 1: Identify the reaction type
The reaction forms 1 mole of CO from its elements in their standard states:
\(C(s)\) and \(O_2(g)\).
This matches the definition of the standard enthalpy of formation.
✔ Statement 1 is correct.
Step 2: Check if it is combustion
Combustion is the complete reaction with oxygen producing the most stable oxides such as \(CO_2\).
Here the product is \(CO\), not \(CO_2\).
Therefore it is not a combustion reaction.
✘ Statement 2 is incorrect.
Step 3: Check atomisation
Atomisation forms gaseous atoms from an element.
This reaction forms a molecule \(CO\), not atoms.
✘ Statement 3 is incorrect.
Correct statement: 1 only.
Answer: D
Which expression gives the standard enthalpy change of combustion of methane?
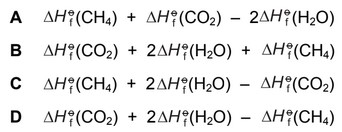
▶️ Answer / Explanation
Step 1: Write the combustion reaction of methane
\(CH_4(g) + 2O_2(g) \rightarrow CO_2(g) + 2H_2O(l)\)
Step 2: Use the enthalpy of formation formula
\(\Delta H^\circ_{reaction} = \sum \Delta H^\circ_f(\text{products}) – \sum \Delta H^\circ_f(\text{reactants})\)
Step 3: Substitute the species
Products: \(CO_2 + 2H_2O\)
Reactants: \(CH_4 + O_2\)
Note: \(\Delta H^\circ_f(O_2) = 0\) because it is an element in its standard state.
Step 4: Final expression
\(\Delta H^\circ_c = \Delta H^\circ_f(CO_2) + 2\Delta H^\circ_f(H_2O) – \Delta H^\circ_f(CH_4)\)
Correct Answer: D
For which enthalpy changes is the value of \(\Delta H\) always negative?
1 combustion
2 hydration
3 solution
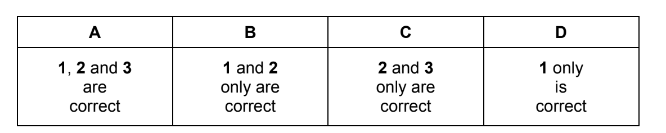
▶️ Answer / Explanation
Statement 1: Combustion
Combustion reactions always release heat because substances react with oxygen to form very stable products such as \(CO_2\) and \(H_2O\).
Therefore combustion reactions are always exothermic.
✔ \(\Delta H\) is always negative.
Statement 2: Hydration
Hydration occurs when gaseous ions become surrounded by water molecules.
Attraction between the ions and water molecules releases energy.
Therefore hydration enthalpy is always negative.
✔ \(\Delta H\) is always negative.
Statement 3: Solution
The enthalpy of solution can be either endothermic or exothermic, depending on the balance between lattice enthalpy and hydration enthalpy.
✘ Not always negative.
Correct statements: 1 and 2 only.
Answer: B
At \(550^\circ C\) nitrogen dioxide reacts with unburnt hydrocarbon fragments such as \(CH_3\cdot\) in the catalytic converter of a motor vehicle.
\(4CH_{3}\cdot + 7NO_{2} \rightarrow 3\frac{1}{2}N_{2} + 4CO_{2} + 6H_{2}O\)
The following table lists types of energy change for this reaction and possible reasons for them.
Which row gives the energy change for this reaction and the reason for it?

▶️ Answer/Explanation
Step 1: Examine the reaction
The reaction converts hydrocarbon fragments and nitrogen dioxide into stable molecules:
- \(N_2\)
- \(CO_2\)
- \(H_2O\)
Step 2: Consider bond formation
These products contain very strong bonds:
- \(N \equiv N\) triple bond in \(N_2\)
- \(C=O\) bonds in \(CO_2\)
- \(O-H\) bonds in \(H_2O\)
When these strong bonds form, a large amount of energy is released.
Step 3: Determine the enthalpy change
The energy released when forming these strong bonds is greater than the energy needed to break the bonds in the reactants.
Therefore the reaction is exothermic.
Step 4: Identify the correct row
The correct explanation is that the products have large negative enthalpies of formation.
This corresponds to row C.
Answer: C
Which equation shows the reaction that occurs during the standard enthalpy change of atomisation of bromine?
A \(Br_2(l) \rightarrow 2Br(g)\)
B \(Br_2(g) \rightarrow 2Br(g)\)
C \(\frac{1}{2}Br_2(l) \rightarrow Br(g)\)
D \(\frac{1}{2}Br_2(g) \rightarrow Br(g)\)
▶️ Answer / Explanation
Definition:
The standard enthalpy change of atomisation is the enthalpy change when one mole of gaseous atoms is formed from an element in its standard state.
Step 1: Identify the standard state of bromine
Bromine is a liquid at standard conditions.
So we must start with:
\(Br_2(l)\)
Step 2: Form one mole of gaseous atoms
To produce exactly one mole of Br(g), half a mole of bromine molecules must dissociate.
\(\frac{1}{2}Br_2(l) \rightarrow Br(g)\)
Therefore the correct equation is:
C
Answer: C
Which quantity gives the best indication of the relative strengths of the hydrogen bonds between the molecules in liquid hydrogen halides?
A bond dissociation energies
B enthalpy changes of formation
C enthalpy changes of solution
D enthalpy changes of vaporisation
▶️ Answer / Explanation
Step 1: Identify what hydrogen bonding affects
Hydrogen bonds are intermolecular forces. These forces hold molecules together in the liquid phase.
Step 2: Consider what happens during vaporisation
During vaporisation:
\( \text{liquid} \rightarrow \text{gas} \)
The intermolecular forces between molecules must be overcome.
The stronger the intermolecular forces (including hydrogen bonds), the more energy is required to vaporise the liquid.
Step 3: Conclusion
The quantity that best reflects the strength of intermolecular forces such as hydrogen bonding is the enthalpy change of vaporisation.
Correct Answer: D
Use of the Data Booklet is relevant for this question.
In an experiment, the burning of 1.45 g (0.025 mol) of propanone was used to heat 100 g of water.
The initial temperature of the water was 20.0 °C and the final temperature of the water was 78.0 °C.
Which experimental value for the enthalpy change of combustion for propanone can be calculated from these results?
A \( -1304 \, kJ \, mol^{-1} \)
B \( -970 \, kJ \, mol^{-1} \)
C \( -352 \, kJ \, mol^{-1} \)
D \( -24.2 \, kJ \, mol^{-1} \)
▶️ Answer / Explanation
Step 1: Calculate temperature change
\(\Delta T = 78.0 – 20.0 = 58^\circ C\)
Step 2: Heat gained by water
\(q = mc\Delta T\)
\(m = 100\,g\)
\(c = 4.18\,J\,g^{-1}\,K^{-1}\)
\(\Delta T = 58\)
\(q = 100 \times 4.18 \times 58\)
\(q = 24244\,J\)
\(q = 24.2\,kJ\)
This is the heat released by 0.025 mol of propanone.
Step 3: Convert to per mole
\(\Delta H = \dfrac{24.2}{0.025}\)
\(\Delta H = 968\,kJ\,mol^{-1}\)
Combustion reactions are exothermic, so the sign is negative.
\(\Delta H = -970\,kJ\,mol^{-1}\)
Correct Answer: B
