Pre AP Chemistry - 4.5A Reaction Rates and Factors Affecting it- MCQ Exam Style Questions -New Syllabus 2025-2026
Pre AP Chemistry -4.5A Reaction Rates and Factors Affecting it- MCQ Exam Style Questions – New Syllabus 2025-2026
Pre AP Chemistry -4.5A Reaction Rates and Factors Affecting it- MCQ Exam Style Questions – Pre AP Chemistry – per latest Pre AP Chemistry Syllabus.
A student reacts strips of zinc with dilute sulfuric acid and measures the time taken to produce 100 cm3 of hydrogen.
The experiment is repeated using different conditions.
The results are shown in the table.
| experiment | time to produce 100 cm3 of hydrogen/s |
|---|---|
| 1 | 250 |
| 2 | 100 |
Which changes in conditions produce the results shown in experiment 2?
- Add a catalyst.
- Dilute the acid.
- Use zinc powder.
- Heat the acid.
A) 1, 3 and 4
B) 1 and 4 only
C) 2 and 3
D) 2 and 4
▶️ Answer/Explanation
Ans: A
To understand why option A is correct, let’s analyze each condition:
1. Adding a catalyst increases the reaction rate by providing an alternative pathway with lower activation energy.
2. Diluting the acid would actually slow down the reaction (decrease rate), so this can’t be correct.
3. Using zinc powder increases the surface area, which increases the reaction rate.
4. Heating the acid provides more energy to the particles, increasing their collision frequency and energy.
Since experiment 2 shows a faster reaction (100s vs 250s), we need conditions that increase the rate. Only options 1, 3, and 4 would increase the rate, making A the correct answer.
Reaction 1 and reaction 2 both produce a gas. The total volume of gas produced in each reaction is measured every minute for 10 minutes.
A graph of the results is shown.
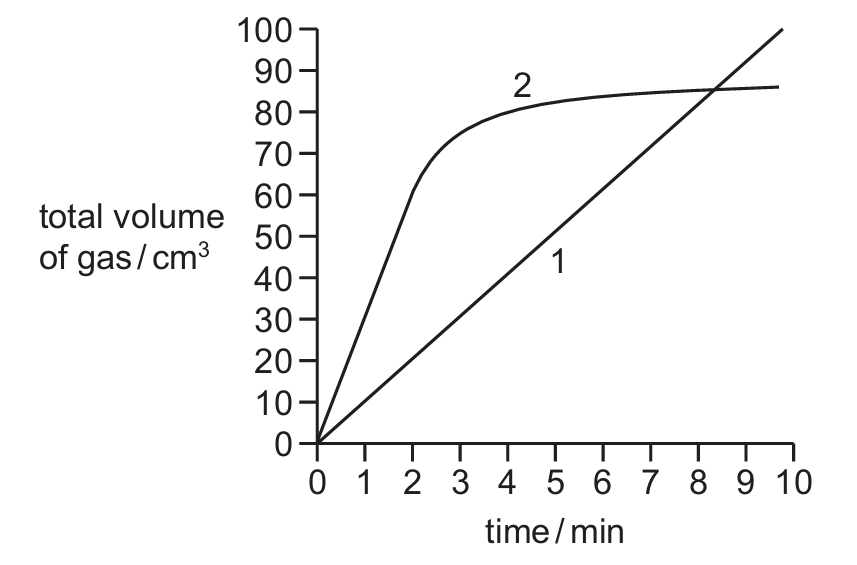
Which row describes how the rate of reaction changes, if at all, during each reaction?
| reaction 1 | reaction 2 | |
|---|---|---|
| A | the rate is constant | the rate decreases after 2 minutes |
| B | the rate increases | the rate increases |
| C | the rate increases | the rate decreases after 2 minutes |
| D | the rate is constant | the rate increases |
▶️ Answer/Explanation
Ans: A
From the graph description:
Reaction 1: The straight line indicates a constant rate of reaction (volume increases steadily with time).
Reaction 2: The steep initial slope followed by leveling off indicates the rate decreases after 2 minutes (likely because reactants are being used up).
Therefore, option A correctly describes both reactions – Reaction 1 has a constant rate while Reaction 2’s rate decreases after 2 minutes.
Which two pieces of apparatus are most useful to measure the rate of a reaction in which a gas is given off?
A) accurate balance and gas syringe
B) accurate balance and thermometer
C) gas syringe and stop-watch
D) stop-watch and thermometer
▶️ Answer/Explanation
Ans: C
To measure the rate of a gas-producing reaction, we need to measure either:
- The volume of gas produced over time (using a gas syringe)
- The time taken to produce a certain amount of gas (using a stop-watch)
Option C combines both these essential pieces of apparatus. A gas syringe measures the volume of gas produced, while a stop-watch measures the time taken. The other options either include unnecessary equipment (thermometer) or less direct measurement methods (balance measures mass change, which isn’t as straightforward for gas evolution).
Four students collect the gas produced from the reaction of calcium carbonate with dilute hydrochloric acid. Each student records the time taken to collect a volume of gas.
Which results show the highest average rate of reaction?
A) 15 cm3 of gas collected in 20 seconds
B) 50 cm3 of gas collected in 40 seconds
C) 75 cm3 of gas collected in 80 seconds
D) 90 cm3 of gas collected in 100 seconds
▶️ Answer/Explanation
Ans: B
To determine the highest average rate of reaction, we calculate the rate of gas production (volume/time) for each option:
A) 15 cm3/20 s = 0.75 cm3/s
B) 50 cm3/40 s = 1.25 cm3/s
C) 75 cm3/80 s ≈ 0.94 cm3/s
D) 90 cm3/100 s = 0.90 cm3/s
Option B has the highest rate at 1.25 cm3/s. Even though option D produces more total gas, it takes proportionally longer, resulting in a lower average rate.
This shows that the highest rate isn’t necessarily the one with the most product, but rather the one that produces gas fastest relative to the time taken.
Hydrogen peroxide decomposes to form water and oxygen. The equation is shown.
\[ 2H_2O_2(l) \rightarrow 2H_2O(l) + O_2(g) \]
Manganese(IV) oxide catalyses this reaction.
Which statements about manganese(IV) oxide are correct?
- It increases the rate of the reaction.
- It increases the total volume of oxygen gas produced at the end of the reaction.
- It will have the same mass at the end of the reaction as it does at the start of the reaction.
A) 1, 2 and 3
B) 1 and 2 only
C) 1 and 3 only
D) 2 and 3 only
▶️ Answer/Explanation
Ans: C
Let’s evaluate each statement about the catalyst manganese(IV) oxide:
1. Correct: Catalysts by definition increase the rate of reaction by providing an alternative reaction pathway with lower activation energy.
2. Incorrect: A catalyst doesn’t affect the total amount of product formed, only how quickly it’s formed. The volume of oxygen depends only on the amount of hydrogen peroxide decomposed.
3. Correct: Catalysts are not consumed in the reaction and remain chemically unchanged, so their mass stays constant.
Therefore, statements 1 and 3 are correct, making option C the right answer.
Magnesium reacts with dilute hydrochloric acid, HCl, to produce hydrogen gas. Which row identifies the reaction conditions that give the fastest rate of reaction?

▶️ Answer/Explanation
Ans: B
1. Rate Factors: The reaction rate increases with:
• Higher temperature (60°C > 25°C)
• Powdered form (greater surface area than ribbon)
• More concentrated acid (2.0 mol/dm³ > 0.5 mol/dm³)
2. Option Analysis:
• B combines all rate-increasing factors (powder, 2.0M, 60°C)
• Other options lack one or more optimal conditions
Thus, B gives the fastest reaction rate.
Water is added to anhydrous copper(II) sulfate. Which row describes the direction of energy change and the colour change of the mixture during the reaction?

▶️ Answer/Explanation
Ans: D
The reaction of anhydrous copper(II) sulfate with water demonstrates two key changes:
- Energy Change (Exothermic): The reaction releases heat as water molecules bond to the copper ions (hydration energy).
- Color Change (White to Blue):
- Anhydrous CuSO₄: White powder (no water molecules)
- Hydrated CuSO₄·5H₂O: Bright blue crystals
The chemical equation for this highly exothermic process is:
CuSO₄ (white) + 5H₂O → CuSO₄·5H₂O (blue) + heat
This makes option D (exothermic + white to blue) correct.
The rate of reaction between magnesium and hydrochloric acid is investigated. The total volume of hydrogen given off is measured at different times. A graph of the results is shown.
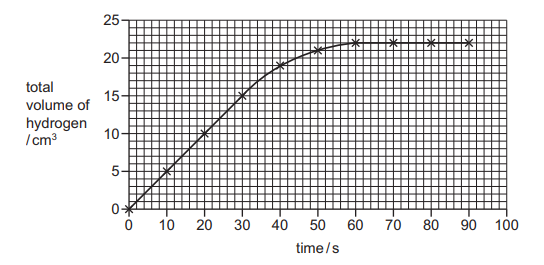
Which conclusions are correct?
1 The rate is fastest between 0 and 30 seconds.
2 The maximum volume of hydrogen given off is 22 cm³
3 At 40 seconds, 20 cm³ of hydrogen is given off.
A 1 and 2 only
B 1 and 3 only
C 2 and 3 only
D 1, 2 and 3
▶️ Answer/Explanation
Ans: A
From the graph analysis:
- Fastest rate (0-30s): The steepest slope occurs initially, indicating highest reaction rate (Correct)
- Maximum volume (22 cm³): The curve plateaus at 22 cm³ (Correct)
- Volume at 40s: The graph shows ~21 cm³ at 40s, not 20 cm³ (Incorrect)
Therefore, only conclusions 1 and 2 are correct, making option A the right choice.
A method used to investigate the rate of reaction of calcium carbonate with dilute hydrochloric acid under different conditions is shown.
Procedure:
- Place 50 cm³ of dilute hydrochloric acid in a conical flask
- Add a known volume of water to the conical flask
- Heat the conical flask to the required temperature
- Add 1.0 g of calcium carbonate to the conical flask
- Measure the time taken for the reaction to finish
Which volume of water and which temperature give the shortest time taken for the reaction to finish?

▶️ Answer/Explanation
Ans: B
To achieve the fastest reaction rate (shortest time), we need:
- Highest concentration of HCl (minimum water added)
- Option B adds 0 cm³ water → maintains original acid concentration
- Highest temperature (increases particle energy/collisions)
- Option B uses 50°C (highest among choices)
Key chemical principles:
- Reaction: \(CaCO_3 + 2HCl → CaCl_2 + H_2O + CO_2\)
- Rate increases with:
- Higher acid concentration (less dilution)
- Higher temperature (more kinetic energy)
Therefore, B (0 cm³ water at 50°C) provides both maximum concentration and highest temperature for fastest reaction.
An experiment is set up as shown. The mass of the conical flask and its contents is measured at 30-second intervals.
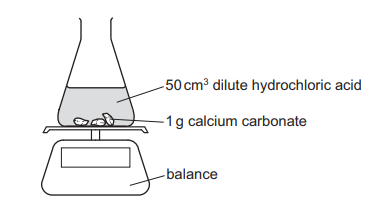
Which statement about the reaction and changes to the reaction conditions is correct?
A Adding \(10 cm^3\) of water to the \(50 cm^3\) of acid increases the rate of the reaction.
B Increasing the size of the pieces of calcium carbonate increases the rate of the reaction.
C Increasing the temperature increases the rate of the reaction.
D The mass of the conical flask and its contents increases as carbon dioxide is formed.
▶️ Answer/Explanation
Ans: C
1. Temperature effect: Higher temperature increases particle energy and collision frequency → faster reaction (C is correct).
2. Other options analysis:
– A: Adding water dilutes acid → decreases reaction rate.
– B: Larger CaCO3 pieces decrease surface area → slower reaction.
– D: Mass decreases as CO2 gas escapes.
Only statement C correctly describes a factor that increases the reaction rate.
Magnesium is added to dilute hydrochloric acid. 25 cm3 of gas is given off in the first 30 s of the reaction. The experiment is repeated at a lower temperature. All other reaction conditions are the same. Which volume of gas is produced in the first 30 s of this reaction?
A. 15 cm3 B. 25 cm3 C. 30 cm3 D. 50 cm3
▶️ Answer/Explanation
Ans: A
The reaction between magnesium and hydrochloric acid is:
Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g)
Key points:
- Temperature effect: Lowering the temperature decreases the reaction rate because particles have less kinetic energy and collide less frequently.
- Gas volume: In the same 30 s time period, less gas (15 cm3) will be produced at the lower temperature compared to the original 25 cm3.
- Why not others:
- B: Same volume would mean no rate change
- C/D: More gas would require faster rate, opposite of temperature decrease effect
The only plausible answer is A (15 cm3), showing reduced reaction rate at lower temperature.
The graph shows the results of two experiments investigating the rate of reaction between excess calcium carbonate and dilute hydrochloric acid. In each experiment the volume of carbon dioxide produced is measured at fixed time intervals.

Which statement describes the difference in conditions between experiments 1 and 2?
- In experiment 2 a higher concentration of dilute hydrochloric acid is used.
- In experiment 2 a higher temperature is used.
- In experiment 2 the mass of calcium carbonate is greater.
- In experiment 2 the particle size of calcium carbonate is greater.
▶️ Answer/Explanation
Ans: D
Key observations from the graph:
- Experiment 2 shows a slower reaction rate (less steep curve).
- Both experiments produce the same total volume of CO2, indicating the same amount of CaCO3 reacted.
Analysis of options:
- A/B: Higher concentration or temperature would increase the rate (incorrect).
- C: Greater mass would produce more CO2 (graph shows equal volumes).
- D: Larger particle size reduces surface area, decreasing reaction rate (correct).
Thus, the difference is due to larger particle size in experiment 2.
The rate of reaction between magnesium and hydrochloric acid is investigated. The volume of hydrogen given off at different times is measured. The results are shown.

Which conclusions are correct?
1 The rate is fastest between 0 and 20 seconds.
2 The maximum volume of hydrogen given off is $22 \mathrm{~cm}^3$.
3 At 40 seconds, $20 \mathrm{~cm}^3$ of hydrogen is given off.
A 1 and 2 only
B 1 and 3 only
C 2 and 3 only
D 1,2 and 3
▶️ Answer/Explanation
Ans: A
Graph Analysis:
- Statement 1 (Correct):
- Steepest slope occurs between 0-20 seconds
- Rate = Δvolume/Δtime is maximum here
- Statement 2 (Correct):
- Graph plateaus at $22 \mathrm{~cm}^3$
- This is the maximum H2 volume produced
- Statement 3 (Incorrect):
- At 40s, graph shows ~$19 \mathrm{~cm}^3$ (not 20)
- Reading error of ±0.5 cm3 possible, but clearly not 20
Therefore, only statements 1 and 2 are correct, corresponding to Option A.
Zinc reacts with an acid to form a gas. The volume of gas produced is measured at intervals. The results are shown as curve Z.
The reaction is repeated in the presence of a catalyst.
Which curve shows the results for the catalysed reaction?
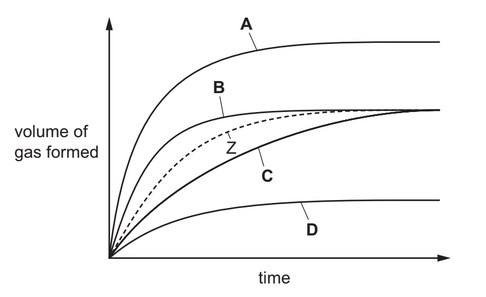
▶️ Answer/Explanation
Ans: B
Key characteristics of a catalysed reaction:
- Same Final Volume: A catalyst doesn’t affect the total amount of product, only the rate.
- Faster Initial Rate: The catalysed reaction will show a steeper initial slope.
Analyzing the graph:
- Curve Z (original reaction) shows gradual gas production
- Curve B has:
- Steeper initial slope (faster reaction)
- Same final volume as Z
- Other curves either:
- Show different final volumes (invalid)
- Or slower rates (invalid)
The reaction equation (Zn + 2H⁺ → Zn²⁺ + H₂) confirms:
- Same amount of zinc → same theoretical H₂ volume
- Catalyst only provides alternative reaction pathway
Therefore, Curve B correctly represents the catalysed reaction.
The apparatus shown is used to measure the speed of a reaction.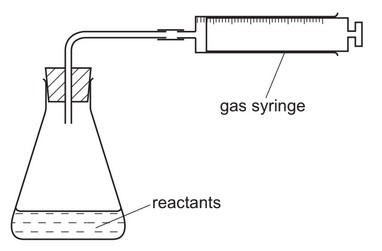
Which equation represents a reaction where the speed can be measured using this apparatus?
A \(Mg(s) + 2HCl(aq) → MgCl_2(aq) + H_2(g)\)
B \(HCl(aq) + NaOH(aq) → NaCl(aq) + H_2O(l)\)
C \(Fe(s) + CuSO_4(aq) → Cu(s) + FeSO_4(aq)\)
D \(2Na(s) + Br_2(l) → 2NaBr(s)\)
▶️ Answer/Explanation
Ans: A
The apparatus shown is designed to measure gas evolution, which means it can only be used to monitor reactions that produce a gas. Let’s analyze each option:
Option A: \(Mg(s) + 2HCl(aq) → MgCl_2(aq) + H_2(g)\)
- Produces hydrogen gas (H₂)
- The rate can be measured by gas collection
- This is the correct choice
Option B: Neutralization reaction (no gas produced)
Option C: Displacement reaction (no gas produced)
Option D: Solid-solid reaction (no gas produced)
Only reaction A produces a measurable gas (hydrogen), making it the only reaction whose rate can be measured with this apparatus. The other reactions either don’t produce gas or involve only solids/liquids.
Solid copper(II) carbonate reacts with dilute sulfuric acid.
\(CuCO_3 + H_2SO_4 → CuSO_4 + CO_2 + H_2O\)
The rate of the reaction can be changed by varying the conditions.
Which changes always increase the rate of this chemical reaction?
1 increasing the concentration of sulfuric acid
2 increasing the size of the pieces of copper(II) carbonate
3 increasing the temperature
4 increasing the volume of sulfuric acid
A 1, 3 and 4 B 1 and 3 only C 2 and 3 D 3 and 4 only
▶️ Answer/Explanation
Ans: B
1. Acid concentration: Higher [H2SO4] means more reactant particles per unit volume, increasing collision frequency and reaction rate.
2. Particle size: Decreasing size (not increasing) raises surface area and rate. Larger pieces would slow the reaction.
3. Temperature: Provides more kinetic energy, making collisions more frequent and energetic (higher success rate).
4. Acid volume: Only increases total reactant amount without changing concentration, so no rate effect unless more surface area is exposed.
Thus, only 1 and 3 (option B) are universally correct for increasing rate.
Marble chips (calcium carbonate) react with hydrochloric acid in an exothermic reaction.
calcium carbonate + hydrochloric acid → calcium chloride + water + carbon dioxide
When excess marble chips are added to dilute hydrochloric acid the rate of the reaction starts off fast, then gets slower until the reaction stops.
Why does the reaction rate get slower?
A) The concentration of the hydrochloric acid is decreasing.
B) The concentration of calcium chloride is increasing.
C) The calcium carbonate is completely used up.
D) The temperature of the mixture decreases.
▶️ Answer/Explanation
Ans: A
1. Key Concept: Reaction rate depends on reactant concentration (Collision Theory).
2. Process Analysis:
• HCl is being consumed (2HCl + CaCO₃ → CaCl₂ + H₂O + CO₂)
• With excess marble chips, HCl becomes limiting reactant
• [HCl] decreases → fewer collisions → slower rate
3. Why Not Others:
• B: Product concentration doesn’t affect rate
• C: Marble is in excess (not fully used)
• D: Reaction is exothermic (temp increases initially)
Thus, A is the correct explanation.
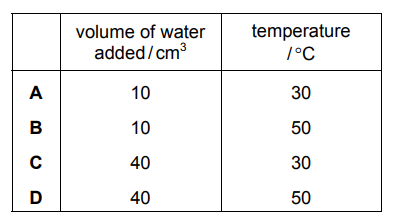
A method used to investigate the rate of reaction of calcium carbonate with dilute hydrochloric acid under different conditions is shown.
- Place 50 cm³ of dilute hydrochloric acid in a conical flask.
- Add a known volume of water to the conical flask.
- Heat the conical flask to the required temperature.
- Add 1.0 g of calcium carbonate to the conical flask.
- Measure the time taken for the reaction to finish.
Which volume of water and which temperature gives the shortest time taken for the reaction to finish?
▶️ Answer/Explanation
Ans: B
The fastest reaction occurs with:
- 0 cm³ water added: Maintains the highest HCl concentration (no dilution), maximizing collision frequency between H⁺ ions and CaCO₃.
- 60°C temperature: Provides maximum kinetic energy to particles, ensuring both more frequent and more energetic collisions.
The reaction rate is governed by the equation:
Rate ∝ [HCl] × e^(-Eₐ/RT)
where higher concentration ([HCl]) and temperature (T) both increase the rate.
This combination (0 cm³ water + 60°C) gives the shortest reaction time as it optimizes both concentration and temperature factors.
Lumps of limestone react with dilute hydrochloric acid according to the equation shown.
CaCO3 + 2HCl → CaCl2 + H2O + CO2
Which change in conditions decreases the rate of the reaction?
- increase the concentration of the acid
- increase the volume of the acid
- increase the size of the lumps of limestone
- increase the temperature
▶️ Answer/Explanation
Ans: C
The rate of reaction between limestone and HCl depends on:
- Surface area: Larger lumps (C) decrease surface area, slowing the reaction
- Other factors:
- Higher acid concentration (A) increases rate
- More acid volume (B) doesn’t affect rate (unless concentration changes)
- Higher temperature (D) increases rate
Only increasing lump size (C) reduces the reaction rate by decreasing available surface area for collisions.
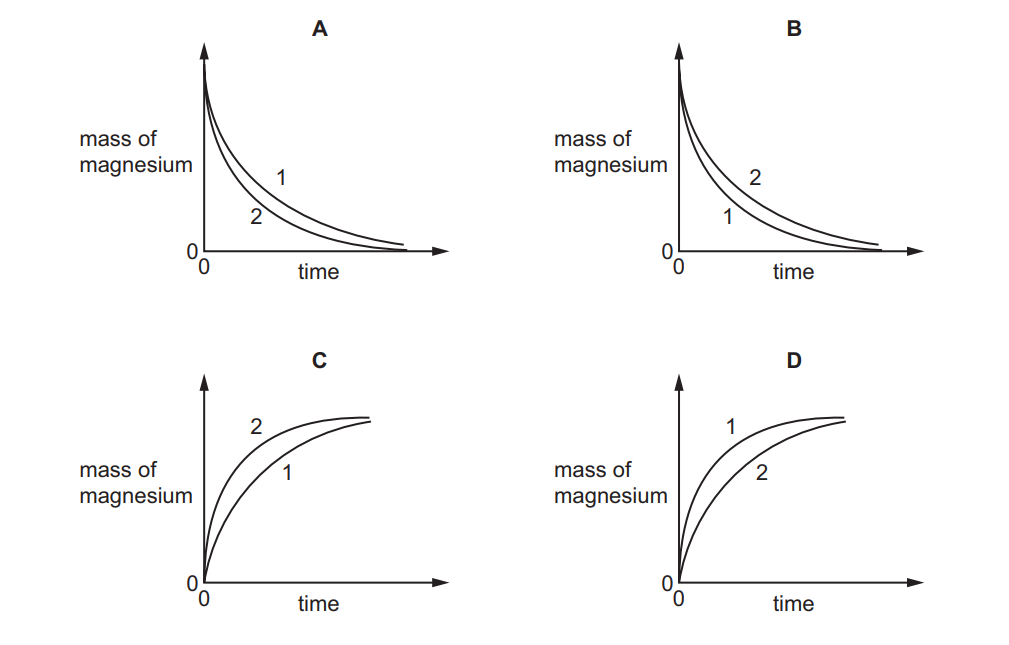
The rate of reaction between magnesium and excess dilute hydrochloric acid was followed by measuring the mass of magnesium present at regular time intervals.
Two experiments were performed:
- Both experiments used 0.1 g of magnesium ribbon
- The acid in experiment 1 was less concentrated than in experiment 2
Which graph shows the results of the experiments?
▶️ Answer/Explanation
Ans: A
Key analysis of the reaction:
- Reaction equation:
\(Mg + 2HCl \rightarrow MgCl_2 + H_2\) - Graph characteristics:
- Mass of Mg decreases over time (reactant being consumed)
- Experiment 2 (higher [HCl]) shows steeper slope → faster rate
- Both experiments start at 0.1g and end at 0g
- Concentration effect:
- Higher [HCl] means more frequent collisions
- Results in faster consumption of Mg
Graph A correctly shows:
- Two decreasing curves from 0.1g to 0g
- Steeper curve for Experiment 2 (more concentrated acid)
- Shallower curve for Experiment 1 (less concentrated acid)
This matches the expected behavior where higher acid concentration leads to faster magnesium consumption.
Which changes increase the rate of reaction between calcium carbonate and dilute hydrochloric acid?
- increasing the concentration of the acid
- increasing the temperature
- increasing the size of the pieces of calcium carbonate
A 1, 2 and 3 B 1 and 2 only C 1 and 3 only D 2 and 3 only
▶️ Answer/Explanation
Ans: B
1. Higher acid concentration (1) → more H+ ions → increased collision frequency → faster reaction.
2. Higher temperature (2) → more energetic particles → more successful collisions → faster reaction.
3. Larger CaCO3 pieces (3) → decreased surface area → slower reaction (incorrect).
Therefore, only changes 1 and 2 increase the reaction rate (Option B).
Copper(II) carbonate reacts with dilute sulfuric acid.
CuCO3(s) + H2SO4(aq) → CuSO4(aq) + CO2(g) + H2O(l)
The rate of the reaction can be changed by varying the conditions.
Which changes always increase the rate of this chemical reaction?
- increasing the concentration of sulfuric acid
- increasing the size of the pieces of copper(II) carbonate
- increasing the temperature
- increasing the volume of sulfuric acid
A 1, 3 and 4 B 1 and 3 only C 2 and 3 D 3 and 4 only
▶️ Answer/Explanation
Ans: B
Analysis of each factor:
- Increasing acid concentration (✓): More H2SO4 particles per unit volume increases collision frequency.
- Increasing particle size (✗): Larger pieces decrease surface area, reducing reaction rate.
- Increasing temperature (✓): Higher kinetic energy leads to more frequent and energetic collisions.
- Increasing acid volume (✗): More solution doesn’t increase concentration or collision frequency if concentration remains constant.
Key points:
- Only factors 1 and 3 always increase rate
- Factor 2 decreases rate (opposite effect)
- Factor 4 has no effect unless concentration changes
Therefore, B (1 and 3 only) is correct.
The diagram shows the apparatus used to measure the rate of a chemical reaction.
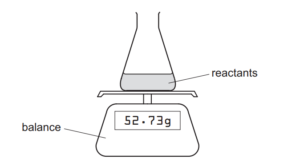
For which reaction can the rate be measured using this apparatus?
- 2Na + Cl2 → 2NaCl
- NaOH + HCl → NaCl + H2O
- Na2O + 2HCl → 2NaCl + H2O
- Na2CO3 + 2HCl → 2NaCl + H2O + CO2
▶️ Answer/Explanation
Ans: D
The apparatus measures reaction rate by tracking mass loss due to gas evolution:
- A/B/C: No gas produced (cannot be measured with this setup).
- D: Produces CO2 gas → mass decreases as gas escapes.
Key points:
• The balance detects mass change from CO2 release.
• Rate = Δmass/Δtime (directly measurable for reaction D).
Thus, only D is compatible with this measurement method.
A student was investigating the reaction between marble chips and dilute hydrochloric acid.
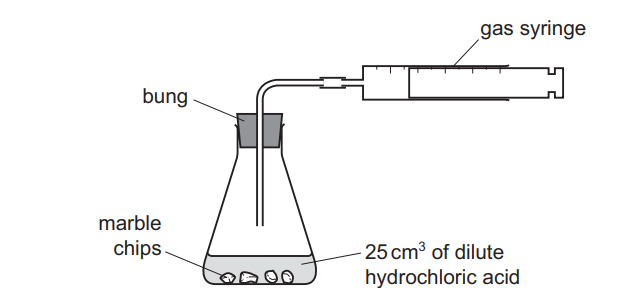
Which changes slow down the rate of reaction?
▶️ Answer/Explanation
Ans: A
Factors Affecting Reaction Rate:
- Temperature (Change 1):
- Lower temperature → slower particle movement
- Fewer effective collisions between HCl and CaCO3
- Concentration (Change 2):
- Diluting acid → fewer H+ ions available
- Reduces collision frequency with marble chips
- Surface Area (Change 3):
- Larger chips → smaller surface area exposed
- Less contact area for acid to react
All three changes (1, 2, and 3) will slow down the reaction rate. However, the question asks which changes slow the reaction, implying the correct answer is the option containing all three factors (A).
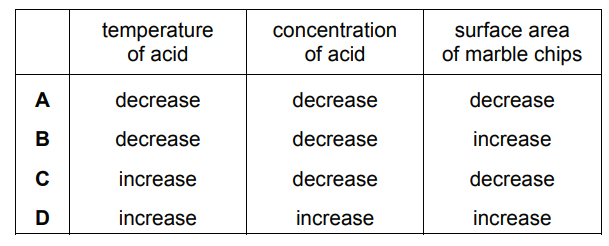
Which changes increase the rate of reaction?
1 increasing the concentration of the reactants
2 increasing the particle size of a solid reactant
3 increasing the temperature
A 1, 2 and 3 B 1 and 2 only C 1 and 3 only D 2 and 3 only
▶️ Answer/Explanation
Ans: C
Analysis of each factor’s effect on reaction rate:
- Increasing concentration (Factor 1):
- More reactant particles per unit volume
- Leads to more frequent collisions → increases rate
- Valid factor for rate increase
- Increasing particle size (Factor 2):
- Larger particles = smaller surface area
- Fewer particles exposed for collisions
- Actually decreases reaction rate
- Increasing temperature (Factor 3):
- Particles gain kinetic energy
- More collisions with sufficient activation energy
- Valid factor for rate increase
Therefore, only factors 1 and 3 increase reaction rate, making C the correct answer.
Key concept: Reaction rate depends on effective collision frequency, which is enhanced by concentration and temperature but hindered by larger particle size.
A liquid X reacts with solid Y to form a gas. Which two diagrams show suitable methods for investigating the rate (speed) of the reaction?
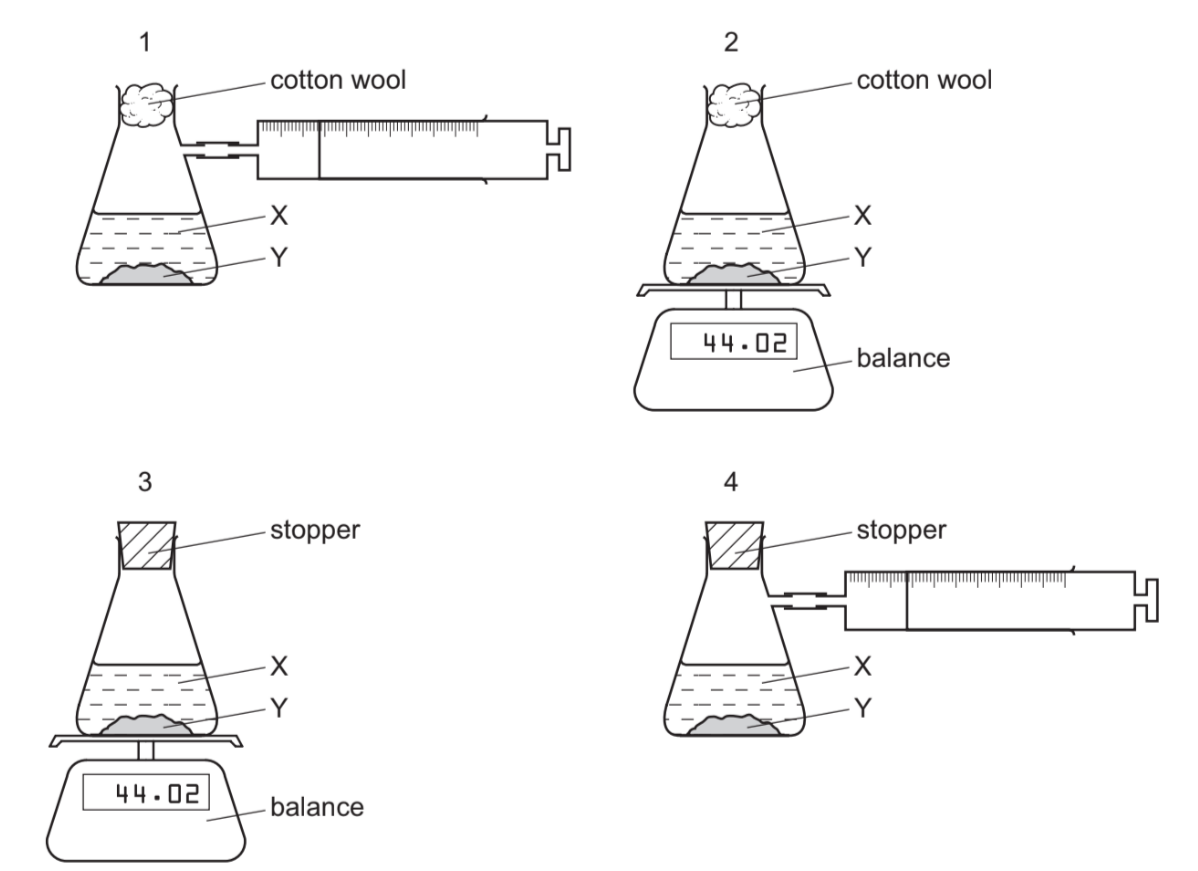
A 1 and 3 B 1 and 4 C 2 and 3 D 2 and 4
▶️ Answer/Explanation
Ans: D
The question describes a reaction between a liquid and solid that produces gas. We need to identify which two methods can measure the reaction rate:
Method 2 (Mass Measurement):
- As the reaction progresses, solid Y is consumed and gas is released
- This causes a decrease in mass that can be measured over time
- The rate of mass loss corresponds to the reaction rate
Method 4 (Gas Collection):
- The gas produced can be collected in a gas syringe or over water
- The volume of gas collected at regular intervals shows the reaction rate
Why not other methods?
- Method 1: Measures pH change – not applicable as no H⁺/OH⁻ involved
- Method 3: Measures temperature – while exothermic, less direct than mass/gas
Therefore, the two most suitable methods are 2 (mass loss) and 4 (gas collection), making D the correct answer.
In separate experiments, a catalyst is added to a reaction mixture and the temperature of the mixture is decreased.
What are the effects of these changes on the rate of the reaction?
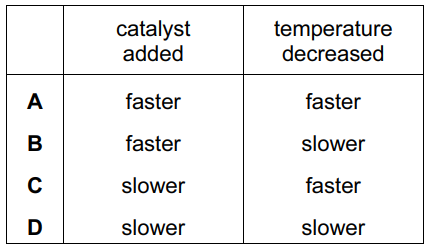
▶️ Answer/Explanation
Ans: B
Catalyst effect: Increases reaction rate by providing an alternative pathway with lower activation energy (Ea). More particles now have sufficient energy to react.
Temperature effect: Decreasing temperature reduces kinetic energy of particles, resulting in fewer successful collisions per second and a slower reaction rate.
The table shows these opposing effects: rate increases with catalyst (left column) but decreases with lower temperature (right column). Only option B matches this combination.
An experiment is carried out to investigate the rate of reaction when calcium carbonate is reacted with hydrochloric acid.
The volume of carbon dioxide gas given off is measured at different intervals of time.
The diagram shows pieces of apparatus used to collect gases.
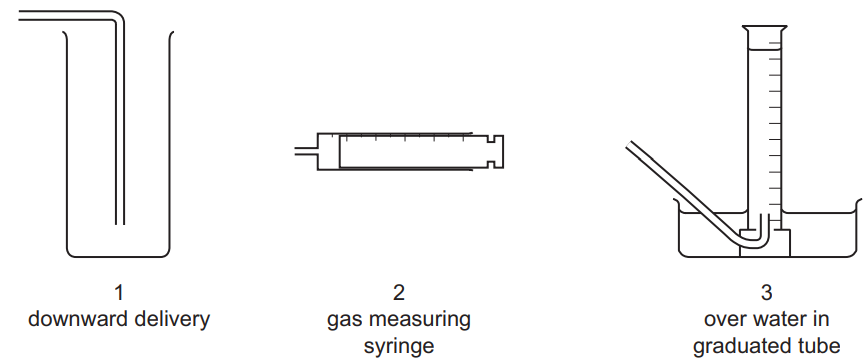
Which apparatus is suitable to collect and measure the volume of the carbon dioxide?
A) 1, 2 and 3
B) 2 and 3 only
C) 1 only
D) 3 only
▶️ Answer/Explanation
Ans: B
1. Apparatus Analysis:
• 1 (Downward delivery): Unsuitable – CO₂ is denser than air and cannot be collected upward
• 2 (Gas syringe): Ideal – directly measures gas volume with precision
• 3 (Inverted measuring cylinder): Suitable – collects CO₂ by downward displacement of water
2. Key Properties: CO₂ has density = 1.98 g/L (heavier than air) and is moderately soluble in water
3. Why Not Others:
• A/C include unsuitable apparatus 1
• D excludes accurate gas syringe method
Thus, B (2 and 3 only) is correct.
Which changes decrease the rate of reaction between magnesium and air?
- heating the magnesium to a higher temperature
- using a higher proportion of oxygen in the air
- using magnesium ribbon instead of powdered magnesium
A 1, 2 and 3 B 1 only C 2 only D 3 only
▶️ Answer/Explanation
Ans: D
Analysis of each factor’s effect on reaction rate:
- Heating: Increases rate (more kinetic energy → more collisions)
- Higher oxygen: Increases rate (more reactant particles available)
- Ribbon vs powder: Decreases rate (less surface area for reaction)
Key concept: Surface area affects reaction rate because only atoms at the surface can react. Powdered magnesium has much greater surface area than ribbon, leading to faster reactions.
Therefore, only change 3 decreases the reaction rate, making D the correct answer.
The rate of a reaction depends on temperature, concentration, particle size and catalysts.
Which statement is not correct?
- Catalysts can be used to increase the rate of reaction.
- Higher concentration decreases the rate of reaction.
- Higher temperature increases the rate of reaction.
- Larger particle size decreases the rate of reaction.
▶️ Answer/Explanation
Ans: B
The incorrect statement is:
B) Higher concentration decreases the rate of reaction.
This is false because:
- Higher concentration increases the rate by providing more reactant particles per unit volume
- This leads to more frequent effective collisions between particles
The other options are correct:
A) Catalysts do increase reaction rates
C) Temperature increases kinetic energy and collision frequency
D) Larger particles have less surface area, slowing reactions
A student investigates the rate of reaction between zinc and an excess of sulfuric acid.
The graph shows the results of two experiments, X and Y.
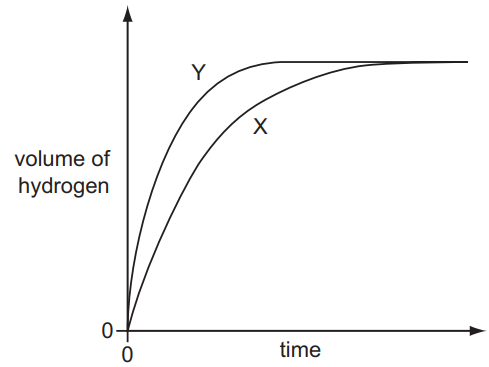
Which change explains the difference between X and Y?
- A catalyst is added in Y.
- A lower temperature is used in Y.
- Larger pieces of zinc are used in Y.
- Less concentrated acid is used in Y.
▶️ Answer/Explanation
Ans: A
Analysis of the graph and reaction conditions:
- Graph observation:
- Y shows a steeper initial slope → faster reaction rate
- Both experiments produce same total gas volume (same endpoint)
- Reaction characteristics:
- Zn + H₂SO₄ → ZnSO₄ + H₂(g)
- Excess acid means zinc is limiting reactant
- Eliminating incorrect options:
- B: Lower temperature would decrease rate (opposite of observation)
- C: Larger pieces would decrease surface area and rate
- D: Less concentrated acid would decrease rate
- Correct explanation:
- Only a catalyst (like copper) could increase rate without changing final yield
- Catalysts provide alternative reaction pathway with lower activation energy
Therefore, A (catalyst added in Y) is the only change that explains the faster reaction rate while maintaining the same final gas volume.
Which change does not increase the speed of reaction between zinc and hydrochloric acid?
A adding a catalyst
B decreasing the particle size of the zinc
C decreasing the temperature
D using more concentrated acid
▶️ Answer/Explanation
Ans: C
1. Adding a catalyst (A) → lowers activation energy → faster reaction (increases speed).
2. Smaller zinc particles (B) → increased surface area → faster reaction (increases speed).
3. Lower temperature (C) → decreases particle energy → fewer collisions → slower reaction (does not increase speed).
4. More concentrated acid (D) → more reactant particles → faster reaction (increases speed).
Therefore, only C (decreasing temperature) does not increase the reaction speed.
