Question 1
\( \mathrm{H_2NNH_2}(aq) + \mathrm{H_2O}(l) \rightleftharpoons \mathrm{H_2NNH_3^+}(aq) + \mathrm{OH^-}(aq) \qquad K_b = 1.3 \times 10^{-6} \)
(ii) Is the resulting solution acidic, basic, or neutral? Justify your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 8.3 \) — Weak Acid and Base Equilibria (Part \( \mathrm{(b)} \), Part \( \mathrm{(d)(ii)} \))
• Topic \( 2.5 \) — Lewis Diagrams (Part \( \mathrm{(c)} \))
• Topic \( 4.2 \) — Net Ionic Equations (Part \( \mathrm{(d)(i)} \))
• Topic \( 4.9 \) — Oxidation-Reduction (Redox) Reactions (Part \( \mathrm{(e)} \))
• Topic \( 3.4 \) — Ideal Gas Law (Part \( \mathrm{(f)} \))
• Topic \( 5.11 \) — Catalysis (Part \( \mathrm{(g)} \))
▶️ Answer/Explanation
(a)
For the correct expression:
\( K_a = \dfrac{[\mathrm{H_3O^+}][\mathrm{HCOO^-}]}{[\mathrm{HCOOH}]} \)
Liquid water is omitted from the equilibrium expression because it is a pure liquid and its activity is treated as constant.
(b)
For the correct calculated concentration of \( \mathrm{H_3O^+} \):
\( \mathrm{HCOOH} + \mathrm{H_2O} \rightleftharpoons \mathrm{H_3O^+} + \mathrm{HCOO^-} \)
Initial:
\( [\mathrm{HCOOH}] = 0.25 \)
\( [\mathrm{H_3O^+}] = 0 \)
\( [\mathrm{HCOO^-}] = 0 \)
Change:
\( -x \qquad +x \qquad +x \)
Equilibrium:
\( [\mathrm{HCOOH}] = 0.25 – x \)
\( [\mathrm{H_3O^+}] = x \)
\( [\mathrm{HCOO^-}] = x \)
Let \( [\mathrm{H_3O^+}] = x \), then
\( 1.8 \times 10^{-4} = \dfrac{x^2}{0.25 – x} \)
Assume \( x \ll 0.25 \), then
\( 1.8 \times 10^{-4} = \dfrac{x^2}{0.25} \Rightarrow x = 0.0067\ \mathrm{M} \)
For the correct calculated value of \( \mathrm{pH} \):
\( \mathrm{pH} = -\log[\mathrm{H_3O^+}] = -\log(0.0067) = 2.17 \)
The weak-acid approximation works well here because \( x \) is small compared with \( 0.25 \). So the pH is \( \boxed{2.17} \).
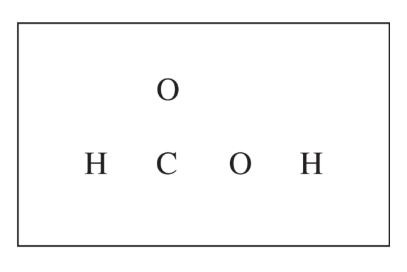
(c)
For the correct diagram:
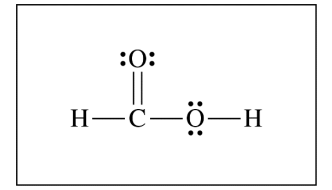
The completed Lewis structure is \( \mathrm{H{-}C(=O){-}O{-}H} \), with two lone pairs on each oxygen atom.
(d)(i)
For the correct balanced equation (state symbols not required):
\( \mathrm{H_2NNH_2}(aq) + \mathrm{HCOOH}(aq) \rightarrow \mathrm{H_2NNH_3^+}(aq) + \mathrm{HCOO^-}(aq) \)
This is a proton-transfer reaction: methanoic acid donates a proton and hydrazine accepts it.
(d)(ii)
For the correct answer and a valid justification:
Acidic. The \( K_a \) of \( \mathrm{H_2NNH_3^+} \) is greater than the \( K_b \) of \( \mathrm{HCOO^-} \), so the production of \( \mathrm{H_3O^+}(aq) \) occurs to a greater extent than the production of \( \mathrm{OH^-}(aq) \).
Since the conjugate acid is stronger than the conjugate base, the resulting solution is \( \boxed{\text{acidic}} \).
(e)
For the correct answer and a valid justification:
Accept one of the following:
• Yes. The oxidation number of hydrogen changes from \( +1 \) in \( \mathrm{HCOOH} \) to zero in \( \mathrm{H_2} \).
• Yes. The oxidation number of carbon changes from \( +2 \) in \( \mathrm{HCOOH} \) to \( +4 \) in \( \mathrm{CO_2} \).
Because one element is reduced and another is oxidized, the reaction is a \( \boxed{\text{redox reaction}} \).
(f)
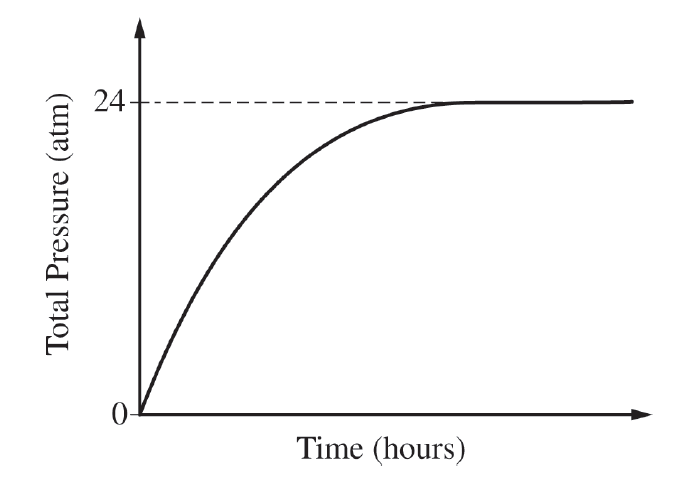
For the correct calculated value of the pressure of \( \mathrm{CO_2} \) (may be implicit):
\( 24\ \mathrm{atm\ total} \times \dfrac{1\ \mathrm{atm\ CO_2}}{2\ \mathrm{atm\ product}} = 12\ \mathrm{atm\ CO_2} \)
Since the reaction makes \( \mathrm{H_2} \) and \( \mathrm{CO_2} \) in a \( 1:1 \) mole ratio, each gas accounts for half the total pressure.
For the correct calculated number of moles of \( \mathrm{CO_2} \):
\( PV = nRT \)
\( n = \dfrac{PV}{RT} = \dfrac{(12\ \mathrm{atm})(4.3\ \mathrm{L})}{(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(298\ \mathrm{K})} = 2.1\ \mathrm{mol\ CO_2} \)
Therefore, the amount of \( \mathrm{CO_2} \) produced is \( \boxed{2.1\ \mathrm{mol}} \).
(g)
For the correct answer and a valid justification:
It would remain the same. In a catalyzed reaction the net amount of catalyst is constant.
A catalyst may participate in intermediate steps, but it is regenerated by the end of the mechanism, so its overall amount does not change.
Question 2
(ii) Write the ground-state electron configuration of \( \mathrm{Si} \).
| Substance | \( S^\circ \) \( \mathrm{(J/(mol \cdot K))} \) |
|---|---|
| \( \mathrm{H_2}(g) \) | \( 131 \) |
| \( \mathrm{Si}(s) \) | \( 18 \) |
| \( \mathrm{SiH_4}(g) \) | \( 205 \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 1.5 \) — Atomic Structure and Electron Configuration (Part \( \mathrm{(a)(ii)} \))
• Topic \( 3.1 \) — Intermolecular and Interparticle Forces (Part \( \mathrm{(b)} \))
• Topic \( 9.2 \) — Absolute Entropy and Entropy Change (Parts \( \mathrm{(d)} \), \( \mathrm{(e)} \))
• Topic \( 5.11 \) — Catalysis (Part \( \mathrm{(f)} \))
• Topic \( 1.6 \) — Photoelectron Spectroscopy (Part \( \mathrm{(g)} \))
• Topic \( 1.7 \) — Periodic Trends (Part \( \mathrm{(h)} \))
• Topic \( 3.12 \) — Properties of Photons (Part \( \mathrm{(i)} \))
▶️ Answer/Explanation
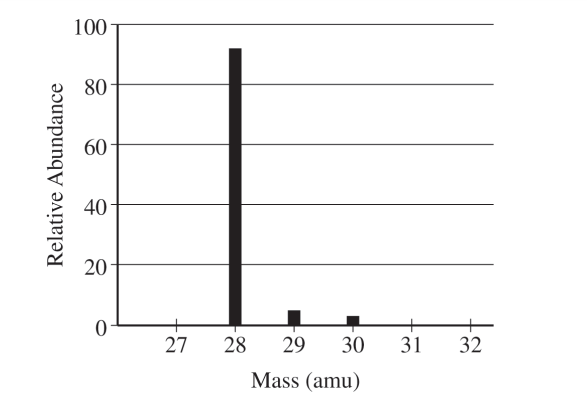
(a)(i)
For the correct answer:
\( 14 \) protons and \( 14 \) neutrons
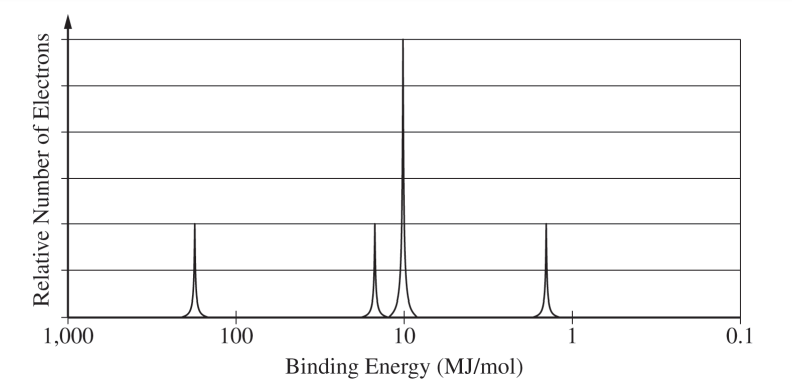
The tallest peak is at mass number \( 28 \), so the most abundant isotope is \( \mathrm{Si}\text{-}28 \). Silicon has atomic number \( 14 \), so neutrons \( = 28 – 14 = 14 \).
(a)(ii)
For the correct answer:
Accept one of the following:
• \( 1s^2\,2s^2\,2p^6\,3s^2\,3p^2 \)
• \( [\mathrm{Ne}]\,3s^2\,3p^2 \)
Silicon has \( 14 \) electrons, so after the neon core, the remaining four electrons go into the \( 3s \) and \( 3p \) sublevels.
(b)
For a correct explanation:
\( \mathrm{SiH_4} \) is composed of molecules, for which the only intermolecular forces are London dispersion forces. \( \mathrm{SiO_2} \) is a network covalent compound with covalent bonds between silicon and oxygen atoms. London dispersion forces are much weaker than covalent bonds, so \( \mathrm{SiH_4} \) boils at a much lower temperature than \( \mathrm{SiO_2} \).
In short, boiling \( \mathrm{SiH_4} \) only requires overcoming weak intermolecular attractions, whereas changing \( \mathrm{SiO_2} \) requires breaking part of a giant covalent network.
(c)
For the correct balanced equation (state symbols not required):
\( \mathrm{SiH_4}(g) \rightarrow \mathrm{Si}(s) + 2\,\mathrm{H_2}(g) \)
A quick atom check shows \( 1 \) silicon atom and \( 4 \) hydrogen atoms on each side.
(d)
For a correct explanation:
The \( \mathrm{H_2}(g) \) molecules are more highly dispersed than the \( \mathrm{Si}(s) \) atoms and, therefore, have a higher absolute molar entropy. Silicon is a solid; therefore, its atoms are in fixed positions, are less dispersed, and have a lower absolute molar entropy.
Gases have many more possible arrangements and much greater freedom of motion than solids, so they generally have higher entropy.
(e)
For the correct calculated value:
\( \Delta S^\circ_{\mathrm{rxn}} = (18 + 2(131)) – 205 = +75\ \mathrm{J/(mol_{rxn} \cdot K)} \)
The positive sign makes sense because the reaction forms more gas particles than it starts with.
(f)
For a correct explanation:
High temperature is required for the reactant particles to have sufficient thermal energy to overcome the activation energy of the reaction.
So even though the reaction is thermodynamically favorable, it is still kinetically slow unless enough energy is supplied.
(g)
For the correct peak height and location:
The peak should be drawn to the right of the other peaks, and it should reach the second line above the horizontal axis.
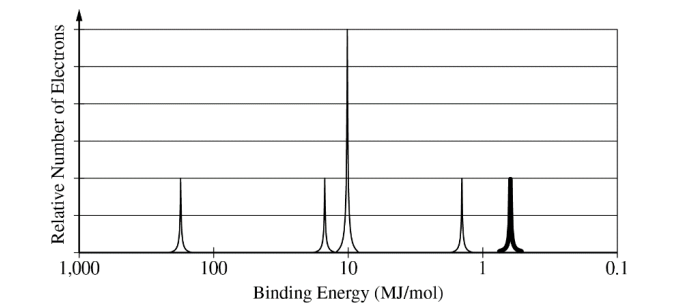
The \( 3p \) electrons have lower binding energy than the \( 3s \) electrons, so their peak is farther to the right. Since there are \( 2 \) electrons in \( 3p \), the peak height corresponds to \( 2 \) electrons.
(h)
For a correct explanation:
The valence electrons of a \( \mathrm{Ge} \) atom occupy a higher shell \( (n = 4) \) than those of a \( \mathrm{Si} \) atom \( (n = 3) \), so the average distance between the nucleus and the valence electrons is greater in \( \mathrm{Ge} \) than in \( \mathrm{Si} \). This greater separation results in weaker Coulombic attractions between the \( \mathrm{Ge} \) nucleus and its valence electrons, making them less tightly bound and, therefore, easier to remove compared to those in \( \mathrm{Si} \).
More shielding and a larger atomic radius in germanium both help lower the first ionization energy.
(i)
For the correct calculated value:
\( E = h\nu = h\left(\dfrac{c}{\lambda}\right) = (6.626 \times 10^{-34}\ \mathrm{J \cdot s}) \left( \dfrac{2.998 \times 10^8\ \mathrm{m \cdot s^{-1}}}{4.00 \times 10^{-7}\ \mathrm{m}} \right) = 4.97 \times 10^{-19}\ \mathrm{J} \)
The energy is inversely proportional to wavelength, so a shorter wavelength would have given an even higher-energy photon.
Question 3
| Mass of dry filter paper | \( 0.764\ \mathrm{g} \) |
| Volume of \( \mathrm{CuSO_4}(aq) \) | \( 50.0\ \mathrm{mL} \) |
| Volume of \( 0.200\ \mathrm{M\ Ba(NO_3)_2} \) | \( 20.0\ \mathrm{mL} \) |
| Mass of filter paper and dried precipitate | \( 1.136\ \mathrm{g} \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.5 \) — Stoichiometry (Parts \( \mathrm{(b)} \), \( \mathrm{(c)} \), \( \mathrm{(d)} \))
• Topic \( 3.7 \) — Solutions and Mixtures (Part \( \mathrm{(e)} \))
• Topic \( 3.13 \) — Beer-Lambert Law (Parts \( \mathrm{(f)} \), \( \mathrm{(g)} \))
▶️ Answer/Explanation
(a)
For the correct balanced equation (state symbols not required):
\( \mathrm{Ba}^{2+}(aq) + \mathrm{SO_4}^{2-}(aq) \rightarrow \mathrm{BaSO_4}(s) \)
\( \mathrm{BaSO_4} \) is the white precipitate, so the net ionic equation includes only the ions that form that solid.
(b)
For the correct calculated value of the mass of precipitate \( (\text{may be implicit}) \):
\( 1.136\ \mathrm{g} – 0.764\ \mathrm{g} = 0.372\ \mathrm{g\ BaSO_4} \)
For the correct calculated value of the number of moles, consistent with mass of precipitate:
\( 0.372\ \mathrm{g} \times \dfrac{1\ \mathrm{mol}}{233.39\ \mathrm{g}} = 0.00159\ \mathrm{mol} \)
So the precipitate formed is \( \boxed{0.00159\ \mathrm{mol\ BaSO_4}} \).
Since the precipitate is dry, its mass can be used directly to find moles.
(c)
For the correct calculated value, consistent with part \( \mathrm{(b)} \):
\( 0.00159\ \mathrm{mol\ BaSO_4} \times \dfrac{1\ \mathrm{mol\ CuSO_4}}{1\ \mathrm{mol\ BaSO_4}} = 0.00159\ \mathrm{mol\ CuSO_4} \)
\( \dfrac{0.00159\ \mathrm{mol\ CuSO_4}}{0.0500\ \mathrm{L}} = 0.0318\ \mathrm{M\ CuSO_4} \)
Therefore, the molarity of the original solution is \( \boxed{0.0318\ \mathrm{M}} \) \( \left( 0.0319\ \mathrm{M}\ \text{if decimals are carried} \right) \).
(d)
For the correct calculated value:
\( M_1V_1 = M_2V_2 \)
\( V_1 = \dfrac{M_2V_2}{M_1} = \dfrac{(0.0500\ \mathrm{M})(50.00\ \mathrm{mL})}{0.1000\ \mathrm{M}} = 25.0\ \mathrm{mL} \)
So, \( \boxed{25.0\ \mathrm{mL}} \) of the stock solution is required.
(e)
For a correct technique to measure the volume of solution:
First, measure out the correct volume of \( 0.1000\ \mathrm{M\ CuSO_4} \) solution with a \( 25.0\ \mathrm{mL} \) volumetric pipet \( (\text{graduated cylinder or buret is acceptable}) \).
For a correct technique to dilute the solution to the final volume:
Transfer the \( 25.0\ \mathrm{mL} \) of solution to a \( 50.00\ \mathrm{mL} \) volumetric flask and dilute the solution with water up to the \( 50.00\ \mathrm{mL} \) mark.
After filling to the mark, stopper and invert the flask several times so the solution is thoroughly mixed.
(f)
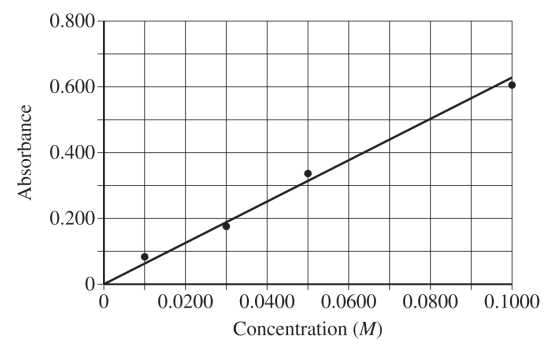
For the correct value \( (\text{between } 0.032\ \mathrm{M} \text{ and } 0.038\ \mathrm{M}) \):
Accept one of the following:
• \( y = mx \)
\( \dfrac{0.63}{0.1000}x = 6.3x \)
\( x = \dfrac{y}{6.3} = \dfrac{0.219}{6.3} = 0.035\ \mathrm{M} \)
• Estimated value from the graph within the specified range.
Therefore, the unknown concentration is \( \boxed{0.035\ \mathrm{M}} \) approximately.
(g)
For the correct answer:
The concentration will be less than that determined in part \( \mathrm{(f)} \).
For a valid justification:
The additional water will decrease the concentration of \( \mathrm{CuSO_4} \) in the cuvette. Therefore, there will be a decrease in absorbance \( (\text{according to the Beer-Lambert law}) \). This dilution results in a lower estimated concentration of \( \mathrm{CuSO_4} \).
Since the sample is unintentionally diluted before measurement, the colorimeter reads a smaller absorbance and the concentration appears lower than it really is.
Question 4
| Time \( (\mathrm{min}) \) | Temperature of Mixture \( (^\circ\mathrm{C}) \) |
|---|---|
| \( 0 \) | \( 22.0 \) |
| \( 1 \) | \( 25.1 \) |
| \( 2 \) | \( 34.6 \) |
| \( 3 \) | \( 37.3 \) |
| \( 4 \) | \( 39.7 \) |
| \( 5 \) | \( 39.4 \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 6.4 \) — Heat Capacity and Calorimetry (Part \( \mathrm{(a)} \))
• Topic \( 6.6 \) — Introduction to Enthalpy of Reaction (Part \( \mathrm{(b)} \))
• Topic \( 4.5 \) — Stoichiometry (Part \( \mathrm{(b)} \), Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value with units:
\( q = mc\Delta T = (15.0\ \mathrm{g})(0.72\ \mathrm{J/(g\cdot^\circ C)})(39.7^\circ\mathrm{C} – 22.0^\circ\mathrm{C}) = 190\ \mathrm{J} \)
Here, \( \Delta T = 17.7^\circ\mathrm{C} \). Since the mixture temperature increases, the mixture absorbs heat.
So, the amount of heat absorbed is \( \boxed{190\ \mathrm{J}} \).
(b)
For the correct calculated value of the moles of reaction, consistent with part \( \mathrm{(a)} \) \( (\text{may be implicit}) \):
\( q_{\mathrm{sys}} = -q_{\mathrm{surr}} \)
\( -190\ \mathrm{J} \times \dfrac{1\ \mathrm{kJ}}{1000\ \mathrm{J}} \times \dfrac{1\ \mathrm{mol_{rxn}}}{-1650\ \mathrm{kJ}} = 0.00012\ \mathrm{mol_{rxn}} \)
For the correct calculated value of the mass of iron:
\( 0.00012\ \mathrm{mol_{rxn}} \times \dfrac{4\ \mathrm{mol\ Fe}}{1\ \mathrm{mol_{rxn}}} \times \dfrac{55.85\ \mathrm{g\ Fe}}{1\ \mathrm{mol\ Fe}} = 0.027\ \mathrm{g\ Fe} \)
The reaction releases heat, so the reaction system loses the same amount of heat that the surroundings gain.
Therefore, the mass of iron that reacted is \( \boxed{0.027\ \mathrm{g}} \) \( \left( 0.026\ \mathrm{g}\ \text{if decimals are carried} \right) \).
(c)
For the correct answer and a valid justification:
Greater than. A greater mass of iron provides a greater number of moles of reaction, which would transfer a greater quantity of thermal energy to the same mass of sand and therefore lead to a greater maximum temperature.
Since the amount of sand stays the same, more reacting iron means more heat released into the same surroundings, so the final maximum temperature must be higher.
Question 5
| Half-Reaction | \( E^\circ \) \( (\mathrm{V}) \) |
|---|---|
| \( \mathrm{Mg^{2+}} + 2e^- \rightarrow \mathrm{Mg} \) | \( -2.37 \) |
| \( \mathrm{Cl_2} + 2e^- \rightarrow 2\,\mathrm{Cl^-} \) | \( +1.36 \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 9.9 \) — Cell Potential and Free Energy (Part \( \mathrm{(b)} \))
• Topic \( 9.11 \) — Electrolysis and Faraday’s Law (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
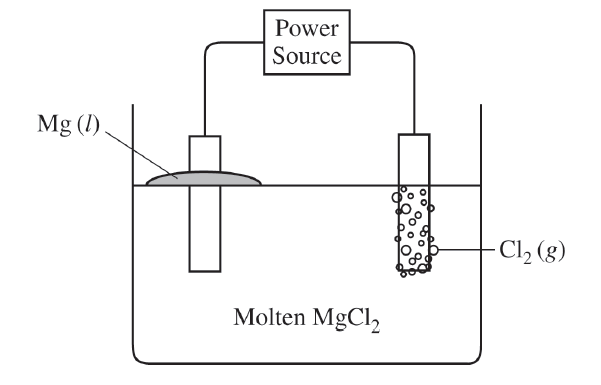
For the correct answer:
Electron flow should be indicated only in a counter-clockwise direction in the external circuit, from the \( \mathrm{Cl_2} \) anode to the \( \mathrm{Mg} \) cathode.
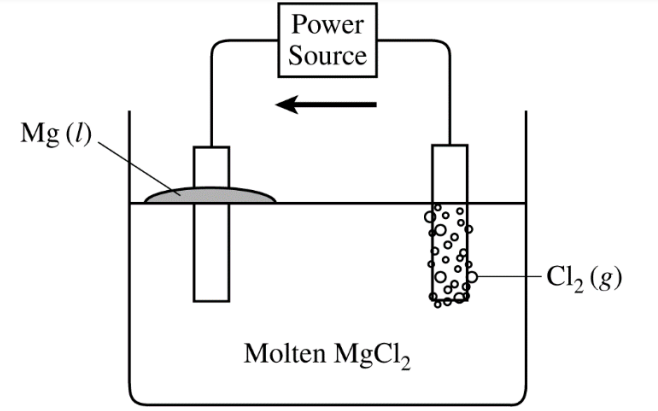
Oxidation occurs at the anode and reduction occurs at the cathode, so electrons move through the wire from the chlorine side toward the magnesium side.
(b)
For the correct answer and calculated value:
No, because \( 2.0\ \mathrm{V} \) is less than \( 3.73\ \mathrm{V} \), which is the minimum voltage needed for electrolysis to occur.
\( E^\circ_{\mathrm{cell}} = -2.37\ \mathrm{V} + (-1.36\ \mathrm{V}) = -3.73\ \mathrm{V} \)
The chlorine half-reaction is listed as a reduction, but in this electrolytic cell chlorine is actually formed at the anode, so that process runs in reverse. Because the overall \( E^\circ_{\mathrm{cell}} \) is negative, an external voltage greater than \( 3.73\ \mathrm{V} \) is needed.
(c)
For the correct calculated value of moles of electrons \( (\text{may be implicit}) \):
\( 2.00\ \mathrm{g\ Mg} \times \dfrac{1\ \mathrm{mol\ Mg}}{24.30\ \mathrm{g\ Mg}} \times \dfrac{2\ \mathrm{mol\ e^-}}{1\ \mathrm{mol\ Mg}} = 0.165\ \mathrm{mol\ e^-} \)
For the correct calculated number of seconds:
\( 0.165\ \mathrm{mol\ e^-} \times \dfrac{96{,}485\ \mathrm{C}}{1\ \mathrm{mol\ e^-}} \times \dfrac{1\ \mathrm{s}}{5.00\ \mathrm{C/s}} = 3180\ \mathrm{s} \)
Since making \( 1 \) mole of \( \mathrm{Mg} \) requires \( 2 \) moles of electrons, first convert grams of magnesium to moles, then to moles of electrons, then to charge, and finally to time using \( I = \dfrac{Q}{t} \).
Therefore, the time required is \( \boxed{3180\ \mathrm{s}} \).
Question 6
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.10 \) — Solubility (Part \( \mathrm{(b)} \), Part \( \mathrm{(d)} \))
• Topic \( 3.8 \) — Representations of Solutions (Part \( \mathrm{(c)} \))
• Topic \( 7.12 \) — Common-Ion Effect (Part \( \mathrm{(d)} \))
▶️ Answer/Explanation
(a)
For a correct description:
Ionic solids do not have free-moving ions that are required to carry an electric current. Therefore, there is no conduction of electricity.
In both solids, the ions are locked into fixed positions in a crystal lattice, so they cannot move from place to place and carry charge.
(b)
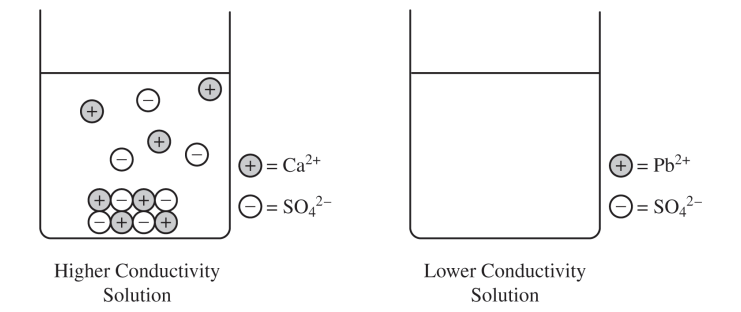
For the correct answer and a valid justification:
\( \mathrm{CaSO_4} \). The greater electrical conductivity of the \( \mathrm{CaSO_4} \) solution relative to the \( \mathrm{PbSO_4} \) solution implies a higher concentration of ions, which comes from the dissolution \( (\text{dissociation}) \) of \( \mathrm{CaSO_4} \) to a greater extent.
More dissolved ions in solution means better conductivity, so the more conductive beaker must contain the more soluble salt.
(c)
For a correct drawing that shows an equal number of cations and anions:
The drawing shows solid \( \mathrm{PbSO_4} \) at the bottom of the beaker \( (\text{similar to the solid shown for } \mathrm{CaSO_4}) \) and fewer dissolved \( \mathrm{Pb^{2+}} \) and \( \mathrm{SO_4^{2-}} \) ions in the solution.
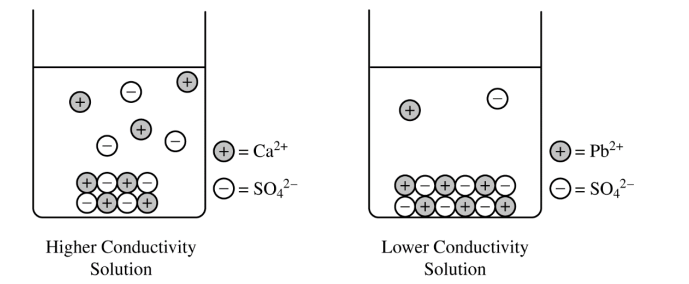
The number of dissolved \( \mathrm{Pb^{2+}} \) ions must equal the number of dissolved \( \mathrm{SO_4^{2-}} \) ions, because the dissolved salt separates in a \( 1:1 \) ratio.
(d)
For a correct explanation:
The additional precipitate is \( \mathrm{CaSO_4} \) that forms in response to the increased \( [\mathrm{SO_4^{2-}}] \) in solution. According to Le Châtelier’s principle \( (Q > K_{sp}) \), the introduction of \( \mathrm{SO_4^{2-}} \) as a common ion shifts the equilibrium towards the formation of more \( \mathrm{CaSO_4}(s) \).
Adding \( \mathrm{H_2SO_4} \) increases sulfate ion concentration, so the dissolution equilibrium \( \mathrm{CaSO_4}(s) \rightleftharpoons \mathrm{Ca^{2+}}(aq) + \mathrm{SO_4^{2-}}(aq) \) shifts left and more solid forms.
Question 7
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.5 \) — Kinetic Molecular Theory (Part \( \mathrm{(c)} \))
• Topic \( 3.6 \) — Deviation from Ideal Gas Law (Part \( \mathrm{(d)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value:
Accept one of the following:
• \( 0.325\ \mathrm{mol\ O_2} \times \dfrac{32.00\ \mathrm{g\ O_2}}{1\ \mathrm{mol\ O_2}} = 10.4\ \mathrm{g\ O_2} \)
\( D = \dfrac{m}{V} = \dfrac{10.4\ \mathrm{g}}{7.95\ \mathrm{L}} = 1.31\ \mathrm{g/L} \)
• \( D = \dfrac{m}{V} = \dfrac{P(\mathrm{MM})}{RT} = \dfrac{(1.0\ \mathrm{atm})(32.00\ \mathrm{g/mol})}{(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(298\ \mathrm{K})} = 1.31\ \mathrm{g/L} \)
So, the density is \( \boxed{1.31\ \mathrm{g/L}} \).
Both methods agree, which is a nice check on the calculation.
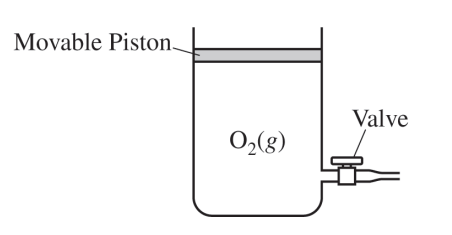
(b)
For the correct answer and a valid justification:
Accept one of the following:
• No, the density of the gas remains constant because \( P \), \( R \), and \( T \) remain constant and the mass and volume of \( \mathrm{O_2} \) decrease proportionately.
• A mathematical justification is shown below.
\( D = \dfrac{m}{V} = \dfrac{n\ \mathrm{moles\ of\ O_2} \times \mathrm{molar\ mass\ of\ O_2}}{nRT/P} = \dfrac{P \times (\mathrm{molar\ mass\ of\ O_2})}{RT} \)
Because the temperature stays constant and the piston is movable, the gas readjusts to the same pressure. That keeps the density unchanged.
(c)
For a valid explanation:
Accept one of the following:
• As the gas cools, the average kinetic energy \( (\text{speed}) \) of the \( \mathrm{O_2} \) molecules decreases. The molecules rebound with less energy when they collide with each other and the walls of the container. The spacing between particles decreases, causing the volume occupied by the gas to decrease.
• As the gas cools, the average kinetic energy \( (\text{speed}) \) of the \( \mathrm{O_2} \) molecules decreases. The molecules rebound with less energy when they collide with each other and the walls of the container. The only way for the molecules to maintain a constant rate of collisions with the walls of the container \( (\text{maintaining a pressure of } 1.00\ \mathrm{atm}) \) is for the volume of the gas to decrease.
Lower temperature means slower particles, so the piston moves downward until the gas once again exerts the outside pressure.
(d)
For a valid explanation:
The ideal gas law assumes that gas particles do not experience interparticle attractions. As a real gas cools further, the intermolecular forces have greater effect as the average speed of the molecules decreases, resulting in inelastic collisions. To maintain a gas pressure of \( 1.00\ \mathrm{atm} \), the volume must decrease to accommodate more collisions with less energy.
At \( -180^\circ \mathrm{C} \), the temperature is very close to the boiling point of oxygen, so the gas behaves much less ideally. Attractive forces become important, and some particles are close to condensing, which makes the measured volume smaller than the ideal-gas prediction.
