Question 1
(ii) When manganese forms cations, electrons are lost from which subshell first? Identify both the number and letter associated with the subshell.
| Mass of empty beaker | \( 60.169\ \mathrm{g} \) |
| Mass of beaker and \( \mathrm{Mn}(s) \) | \( 61.262\ \mathrm{g} \) |
| Mass of beaker and \( \mathrm{Mn}_{x}\mathrm{Cl}_{y}(s) \) after heating to constant mass | \( 62.673\ \mathrm{g} \) |

(ii) Calculate the value of \( E^\circ_{\mathrm{cell}} \) for the overall reaction.
(iii) Calculate the value of \( \Delta G^\circ \) in \( \mathrm{kJ/mol}_{\mathrm{rxn}} \).
(iv) A student claims that the total mass of an alkaline battery decreases as the battery operates because the anode loses mass. Do you agree with the student’s claim? Justify your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 1.3 \) — Elemental Composition of Pure Substances (Parts \( \mathrm{(b)} \), \( \mathrm{(c)} \), \( \mathrm{(d)} \), \( \mathrm{(e)} \))
• Topic \( 9.8 \) — Galvanic (Voltaic) and Electrolytic Cells (Parts \( \mathrm{(f)(i)} \), \( \mathrm{(f)(iv)} \))
• Topic \( 9.9 \) — Cell Potential and Free Energy (Parts \( \mathrm{(f)(ii)} \), \( \mathrm{(f)(iii)} \))
▶️ Answer/Explanation
(a)(i)
Accept one of the following:
\( 1s^2\,2s^2\,2p^6\,3s^2\,3p^6\,4s^2\,3d^5 \)
or \( [\mathrm{Ar}]\,4s^2\,3d^5 \)
(a)(ii)
\( 4s \)
(b)
\( 62.673\ \mathrm{g} – 61.262\ \mathrm{g} = 1.411\ \mathrm{g\ Cl} \)
(c)
\( 1.411\ \mathrm{g\ Cl} \times \dfrac{1\ \mathrm{mol\ Cl}}{35.45\ \mathrm{g\ Cl}} = 0.03980\ \mathrm{mol\ Cl} \)
(d)
\( \dfrac{0.03980\ \mathrm{mol\ Cl}}{0.0199\ \mathrm{mol\ Mn}} = 2.00 \)
Therefore, the empirical formula is \( \mathrm{MnCl}_{2} \).
(e)
Less than. If some of the \( \mathrm{Mn}_{x}\mathrm{Cl}_{y} \) splatters out, the final measured mass is smaller, so the calculated mass and moles of chlorine are smaller.
(f)(i)
\( \mathrm{2\,MnO}_{2}(s) + \mathrm{H}_{2}\mathrm{O}(l) + 2\,e^- \rightarrow \mathrm{Mn}_{2}\mathrm{O}_{3}(s) + 2\,\mathrm{OH}^-(aq) \)
\( \mathrm{Zn}(s) + 2\,\mathrm{OH}^-(aq) \rightarrow \mathrm{ZnO}(s) + \mathrm{H}_{2}\mathrm{O}(l) + 2\,e^- \)
Overall:
\( \mathrm{2\,MnO}_{2}(s) + \mathrm{Zn}(s) \rightarrow \mathrm{Mn}_{2}\mathrm{O}_{3}(s) + \mathrm{ZnO}(s) \)
(f)(ii)
\( E^\circ_{\mathrm{cell}} = 0.15\ \mathrm{V} – (-1.28\ \mathrm{V}) = 1.43\ \mathrm{V} \)
(f)(iii)
\( \Delta G^\circ = -nFE^\circ \)
\( \Delta G^\circ = -(2)(96{,}485)(1.43)\left(\dfrac{1\ \mathrm{kJ}}{1000\ \mathrm{J}}\right) = -276\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
(f)(iv)
Disagree. The battery is sealed, so the total mass remains the same.
Question 2
| Reaction Number | Equation | \( \Delta H^\circ_{\mathrm{rxn}} \) \( \mathrm{(kJ/mol}_{\mathrm{rxn}}\mathrm{)} \) |
|---|---|---|
| \( 2 \) | \( \mathrm{Al}(s) + \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \rightarrow \mathrm{AlCl}_{3}(g) \) | \( -583 \) |
| \( 3 \) | \( \mathrm{Al}(s) \rightarrow \mathrm{Al}(g) \) | \( +326 \) |
| \( 4 \) | \( \mathrm{Cl}_{2}(g) \rightarrow 2\,\mathrm{Cl}(g) \) | \( +243 \) |
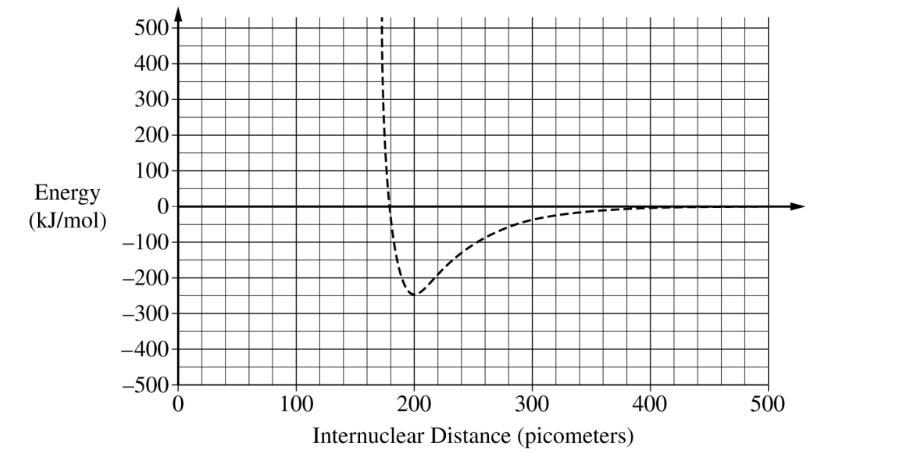
(ii) A student finds that the average \( \mathrm{Al-Cl} \) bond length is \( 220\ \mathrm{pm} \) and the average bond energy is \( 425\ \mathrm{kJ/mol} \). Draw the potential energy curve for the average \( \mathrm{Al-Cl} \) bond on the preceding graph.
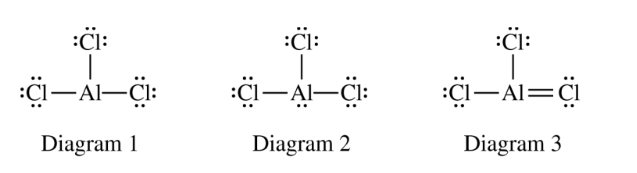
(ii) Which of the three diagrams is the best representation for the bonding in \( \mathrm{AlCl}_{3} \)? Justify your choice based on formal charges.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 6.9 \) — Hess’s Law (Part \( \mathrm{(b)} \))
• Topic \( 2.2 \) — Intramolecular Force and Potential Energy (Parts \( \mathrm{(c)(i)} \), \( \mathrm{(c)(ii)} \))
• Topic \( 2.7 \) — VSEPR and Hybridization (Part \( \mathrm{(d)(i)} \))
• Topic \( 2.6 \) — Resonance and Formal Charge (Part \( \mathrm{(d)(ii)} \))
• Topic \( 7.3 \) — Reaction Quotient and Equilibrium Constant (Part \( \mathrm{(e)} \))
• Topic \( 7.4 \) — Calculating the Equilibrium Constant (Part \( \mathrm{(f)} \))
• Topic \( 7.8 \) — Representations of Equilibrium (Part \( \mathrm{(f)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value reported with the correct number of significant figures:
\( 1.25\ \mathrm{mol\ AlCl}_{3} \times \dfrac{3\ \mathrm{mol\ Cl}}{1\ \mathrm{mol\ AlCl}_{3}} \times \dfrac{35.45\ \mathrm{g\ Cl}}{1\ \mathrm{mol\ Cl}} = 133\ \mathrm{g\ Cl} \)
First use the mole ratio from reaction \( 1 \), then convert moles of \( \mathrm{Cl} \) to grams using its molar mass.
(b)
For the correct algebraic manipulation of either \( \Delta H^\circ_{2} \) or \( \Delta H^\circ_{4} \) (may be implicit):
Accept one of the following:
• Reversing reaction \( 2 \):
\( \mathrm{AlCl}_{3}(g) \rightarrow \mathrm{Al}(s) + \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \qquad \Delta H^\circ_{\mathrm{rxn}} = -(-583) = +583\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
• Multiplying reaction \( 4 \) by \( \dfrac{3}{2} \):
\( \dfrac{3}{2}\,\mathrm{Cl}_{2}(g) \rightarrow 3\,\mathrm{Cl}(g) \qquad \Delta H^\circ_{\mathrm{rxn}} = \dfrac{3}{2}(243) = +365\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
For the correct calculated value:
\( \Delta H^\circ_{1} = -\Delta H^\circ_{2} + \Delta H^\circ_{3} + 1.5(\Delta H^\circ_{4}) = -(-583) + 326 + 1.5(243) = 1274\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
This is a Hess’s law problem: reverse reaction \( 2 \), keep reaction \( 3 \), and multiply reaction \( 4 \) by \( \dfrac{3}{2} \), then add.
(c)(i)
For the correct answer:
\( 200\ \mathrm{pm} \) \((\pm 10\ \mathrm{pm})\)
The bond length is the internuclear distance at the minimum point of the potential energy curve.
(c)(ii)
For a curve with a minimum at an internuclear distance of \( 220 \pm 10\ \mathrm{pm} \):
See sample curve below.
For a curve with a minimum energy value of \( -425 \pm 20\ \mathrm{kJ/mol} \) that approaches zero as the internuclear distance approaches \( 500\ \mathrm{pm} \):
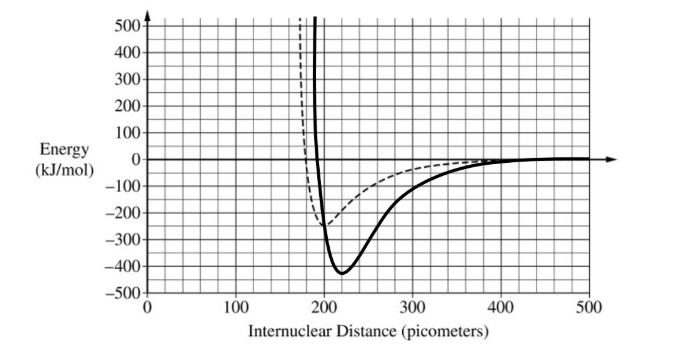
Because the \( \mathrm{Al-Cl} \) bond is longer than the \( \mathrm{Cl-Cl} \) bond, its minimum is shifted slightly to the right. Because its bond energy is greater, the well is deeper.
(d)(i)
For the correct answer and a valid justification:
Diagram \( 2 \). Aluminum has four electron domains in Diagram \( 2 \), which would be trigonal pyramidal, not trigonal planar.
A trigonal planar molecule has \( 3 \) electron domains around the central atom, not \( 4 \).
(d)(ii)
For the correct answer and a valid justification:
Diagram \( 1 \). All atoms in diagram \( 1 \) have a formal charge of zero, whereas atoms in diagrams \( 2 \) and \( 3 \) have nonzero formal charges.
The best Lewis structure is usually the one with the smallest magnitude of formal charges, especially when all formal charges can be zero.
(e)
For the correct answer:
\( K_p = \dfrac{P_{\mathrm{Al}_{2}\mathrm{Cl}_{6}}}{\left(P_{\mathrm{AlCl}_{3}}\right)^2} \)
The exponent \( 2 \) on \( P_{\mathrm{AlCl}_{3}} \) comes directly from the coefficient \( 2 \) in the balanced equilibrium equation.
(f)
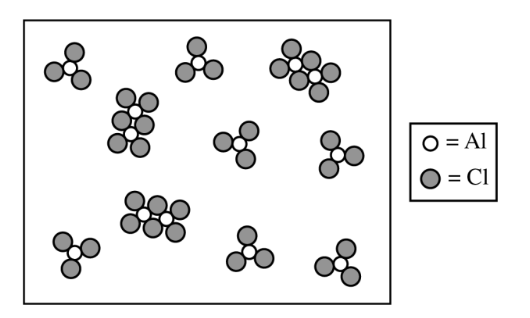
For the correct calculated value, consistent with part \( \mathrm{(e)} \):
From the particulate diagram, there are \( 3 \) molecules of \( \mathrm{Al}_{2}\mathrm{Cl}_{6} \) and \( 7 \) molecules of \( \mathrm{AlCl}_{3} \).
Therefore, \( \chi_{\mathrm{Al}_{2}\mathrm{Cl}_{6}} = \dfrac{3}{10} \) and \( \chi_{\mathrm{AlCl}_{3}} = \dfrac{7}{10} \)
So, \( P_{\mathrm{Al}_{2}\mathrm{Cl}_{6}} = \dfrac{3}{10}(22.1) \) and \( P_{\mathrm{AlCl}_{3}} = \dfrac{7}{10}(22.1) \)
\( K_p = \dfrac{\chi_{\mathrm{Al}_{2}\mathrm{Cl}_{6}}(P_{\mathrm{total}})}{\left(\chi_{\mathrm{AlCl}_{3}}(P_{\mathrm{total}})\right)^2} = \dfrac{\dfrac{3}{10}(22.1)}{\left(\dfrac{7}{10}(22.1)\right)^2} = 0.0277 \)
Thus, \( \boxed{K_p = 0.0277} \).
Question 3
| Trial | Concentration of \( \mathrm{HCl}(aq) \) \( (\mathrm{M}) \) | Particle Size of \( \mathrm{CaCO}_{3}(s) \) | Time of Reaction \( (\mathrm{s}) \) |
|---|---|---|---|
| \( 1 \) | \( 1.00 \) | Fine powder | \( 67 \) |
| \( 2 \) | \( 1.00 \) | Small chunks | \( 112 \) |
| \( 3 \) | \( 1.00 \) | Large chunk | \( 342 \) |
| \( 4 \) | \( 3.00 \) | Fine powder | \( 22 \) |
| \( 5 \) | \( 3.00 \) | Small chunks | \( 227 \) |
| \( 6 \) | \( 3.00 \) | Large chunk | \( 114 \) |
| Time \( (\mathrm{s}) \) | Measured Temperature of Solution \( \left( ^\circ \mathrm{C} \right) \) |
|---|---|
| \( 0 \) | \( 21.20 \) |
| \( 20 \) | \( 21.51 \) |
| \( 40 \) | \( 21.70 \) |
| \( 60 \) | \( 21.85 \) |
| \( 80 \) | \( 21.90 \) |
| \( 100 \) | \( 21.90 \) |
(ii) Calculate the enthalpy of reaction in units of \( \mathrm{kJ/mol}_{\mathrm{rxn}} \). Include the algebraic sign on your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 5.1 \) — Reaction Rates (Parts \( \mathrm{(b)} \), \( \mathrm{(d)} \))
• Topic \( 5.5 \) — Collision Model (Part \( \mathrm{(c)} \))
• Topic \( 5.2 \) — Introduction to Rate Law (Part \( \mathrm{(d)} \))
• Topic \( 4.5 \) — Stoichiometry (Part \( \mathrm{(e)} \))
• Topic \( 6.1 \) — Endothermic and Exothermic Processes (Part \( \mathrm{(f)} \))
• Topic \( 6.4 \) — Heat Capacity and Calorimetry (Part \( \mathrm{(g)(i)} \))
• Topic \( 6.6 \) — Introduction to Enthalpy of Reaction (Part \( \mathrm{(g)(ii)} \))
▶️ Answer/Explanation
(a)
For the correct balanced equation (state symbols not required):
Accept one of the following:
• \( \mathrm{CaCO}_{3}(s) + 2\,\mathrm{H}^{+}(aq) \rightarrow \mathrm{Ca}^{2+}(aq) + \mathrm{CO}_{2}(g) + \mathrm{H}_{2}\mathrm{O}(l) \)
• \( \mathrm{CaCO}_{3}(s) + 2\,\mathrm{H}_{3}\mathrm{O}^{+}(aq) \rightarrow \mathrm{Ca}^{2+}(aq) + \mathrm{CO}_{2}(g) + 3\,\mathrm{H}_{2}\mathrm{O}(l) \)
Chloride is a spectator ion, so it does not appear in the net ionic equation.
(b)
For a correct explanation:
Accept one of the following:
• Even though the concentration of \( \mathrm{HCl} \) is greater in trial \( 5 \) than in trial \( 2 \), the reaction time is significantly longer. Both trial \( 2 \) and \( 5 \) occur under otherwise identical conditions. The trend for trial \( 1 \) and \( 4 \) indicates that higher concentration of \( \mathrm{HCl} \) results in a shorter time of reaction.
• The time of reaction in trial \( 5 \), with small chunks of calcium carbonate, is longer than trial \( 6 \) with large chunks. Both trial \( 5 \) and \( 6 \) occur under otherwise identical conditions. The trend for trials \( 1 \), \( 2 \), and \( 3 \) shows that larger chunks of the solid result in longer time of reaction.
So trial \( 5 \) does not fit the pattern shown by the other trials and is therefore inconsistent.
(c)
For a correct explanation of the effect of surface area on reaction time:
The time of reaction in trial \( 2 \) is shorter than trial \( 3 \) because the calcium carbonate in trial \( 2 \) has a larger surface area \( (\text{meaning that more particles of calcium carbonate are exposed to the } \mathrm{H}^{+} \text{ particles in solution}) \).
For a correct explanation of the effect of particle collisions on reaction rate:
The larger interface between the two reacting substances means there will be more collisions between the particles in a given amount of time, and thus, a higher frequency of successful collisions in which the particles react to form the products.
In short, more exposed surface means more opportunities for acid particles to collide with the solid each second.
(d)
For the correct answer and a valid justification:
Accept one of the following:
• Disagree. If the reaction was zeroth order with respect to \( \mathrm{HCl} \), then changing the concentration of \( \mathrm{HCl} \) would not affect the rate of reaction, and the time of reaction would be the same for trials in which the only difference was \( [\mathrm{HCl}] \). The student’s data for trials \( 1 \) and \( 4 \) \( (\text{and likewise for } 3 \text{ and } 6) \) show that changing \( [\mathrm{HCl}] \) significantly alters the time of reaction.
• Disagree. The reaction appears to be first order, not zeroth order, with respect to \( [\mathrm{HCl}] \). Tripling \( [\mathrm{HCl}] \) results in a reaction time that is about \( \dfrac{1}{3} \) of that when \( [\mathrm{HCl}] = 1.00\ \mathrm{M} \), which means the reaction rate has also tripled, indicating a first-order process.
Since rate changes when concentration changes, the order with respect to \( \mathrm{HCl} \) cannot be zero.
(e)
For the correct calculated moles of \( \mathrm{HCl} \) reacted (may be implicit):
\( 1.00\ \mathrm{g\ CaCO}_{3} \times \dfrac{1\ \mathrm{mol}}{100.09\ \mathrm{g}} = 0.00999\ \mathrm{mol\ CaCO}_{3} \)
\( 0.00999\ \mathrm{mol\ CaCO}_{3} \times \dfrac{2\ \mathrm{mol\ HCl}}{1\ \mathrm{mol\ CaCO}_{3}} = 0.0200\ \mathrm{mol\ HCl\ reacted} \)
For the correct calculated \( [\mathrm{HCl}] \) remaining, consistent with the number of moles reacted:
\( 0.0500\ \mathrm{L} \times \dfrac{1.00\ \mathrm{mol\ HCl}}{1\ \mathrm{L}} = 0.0500\ \mathrm{mol\ HCl\ initially\ present} \)
\( 0.0500\ \mathrm{mol} – 0.0200\ \mathrm{mol} = 0.0300\ \mathrm{mol\ remaining} \)
\( \dfrac{0.0300\ \mathrm{mol}}{0.0500\ \mathrm{L}} = 0.600\ \mathrm{M\ HCl\ remaining} \)
Therefore, the molarity of \( \mathrm{HCl}(aq) \) after completion in trial \( 2 \) is \( \boxed{0.600\ \mathrm{M}} \).
(f)
For the correct answer and a valid justification:
Exothermic. The solution temperature increases as the reaction proceeds, rising from \( 21.20^\circ \mathrm{C} \) to \( 21.90^\circ \mathrm{C} \). This means thermal energy is released by the reaction to the surroundings.
Since the solution warms up, \( q_{\mathrm{sur}} \) is positive and the reaction itself must have \( q_{\mathrm{sys}} < 0 \).
(g)(i)
For the correct calculated value (sign not required):
\( q_{\mathrm{sur}} = mc\Delta T = (51.0\ \mathrm{g})(4.0\ \mathrm{J/(g \cdot ^\circ C)})(21.90^\circ \mathrm{C} – 21.20^\circ \mathrm{C}) = 140\ \mathrm{J} \)
Here, \( \Delta T = 0.70^\circ \mathrm{C} \), so the heat absorbed by the solution is \( \boxed{140\ \mathrm{J}} \).
(g)(ii)
For the correct calculated value, consistent with part \( \mathrm{(g)(i)} \), and the correct sign, consistent with part \( \mathrm{(f)} \):
\( q_{\mathrm{sys}} = -q_{\mathrm{sur}} = -140\ \mathrm{J} = -0.14\ \mathrm{kJ} \)
\( 1.00\ \mathrm{g\ CaCO}_{3} \times \dfrac{1\ \mathrm{mol\ CaCO}_{3}}{100.09\ \mathrm{g\ CaCO}_{3}} \times \dfrac{1\ \mathrm{mol}_{\mathrm{rxn}}}{1\ \mathrm{mol\ CaCO}_{3}} = 0.00999\ \mathrm{mol}_{\mathrm{rxn}} \)
\( \Delta H^\circ_{\mathrm{rxn}} = \dfrac{-0.14\ \mathrm{kJ}}{0.00999\ \mathrm{mol}_{\mathrm{rxn}}} = -14\ \mathrm{kJ/mol}_{\mathrm{rxn}} \)
Therefore, the enthalpy of reaction is \( \boxed{-14\ \mathrm{kJ/mol}_{\mathrm{rxn}}} \).
Question 4
• \( 0.100\ \mathrm{M\ CH_3NH_2}(aq) \) • Weighing paper • \( 10.0\ \mathrm{mL} \) graduated cylinder • pH meter
• Solid \( \mathrm{CH_3NH_3Cl} \) • \( 50.00\ \mathrm{mL} \) buret • Small spatula
| Step | Procedure |
|---|---|
| \( 1 \) | |
| \( 2 \) | Place the solid in the \( 50\ \mathrm{mL} \) beaker. |
| \( 3 \) | Clean the buret and rinse with distilled water. |
| \( 4 \) | |
| \( 5 \) | Use the buret to add \( 25.00\ \mathrm{mL} \) of \( 0.100\ \mathrm{M\ CH_3NH_2}(aq) \) to the beaker. |
| \( 6 \) | Mix well. |
| \( 7 \) | Check the pH with the pH meter. |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 8.4 \) — Acid-Base Reactions and Buffers (Part \( \mathrm{(b)} \))
• Topic \( 8.9 \) — Henderson-Hasselbalch Equation (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value:
\( 0.00250\ \mathrm{mol\ CH_3NH_3Cl} \times \dfrac{67.52\ \mathrm{g}}{1\ \mathrm{mol}} = 0.169\ \mathrm{g} \)
A quick check: the problem says the buffer uses equimolar amounts, so the salt must also provide \( 0.00250\ \mathrm{mol} \), matching the moles of \( \mathrm{CH_3NH_2} \).
(b)
For a correct description of step \( 1 \):
Accept one of the following:
• Use the spatula, balance, and weighing paper to measure out exactly \( 0.169\ \mathrm{g} \) of \( \mathrm{CH_3NH_3Cl}(s) \).
• Use the balance to weigh out the mass of solid found in part \( \mathrm{(a)} \).
For a correct description of step \( 4 \):
Rinse the buret with a small amount of \( 0.100\ \mathrm{M\ CH_3NH_2}(aq) \), drain, and refill with \( 0.100\ \mathrm{M\ CH_3NH_2}(aq) \).
The buret should be rinsed with the solution it will contain so the concentration is not changed by leftover water.
(c)
For the correct answer and a valid justification:
Equal to. The ratio of weak acid to conjugate base is still \( 1:1 \).
In the second buffer, both the base \( \mathrm{CH_3NH_2} \) and its conjugate acid \( \mathrm{CH_3NH_3^+} \) are reduced by the same factor, so \( \dfrac{[\mathrm{CH_3NH_2}]}{[\mathrm{CH_3NH_3^+}]} \) stays the same.
Since buffer pH depends on this ratio, the pH remains unchanged.
Question 5
(ii) The rigid \( 6.00\ \mathrm{L} \) container of \( \mathrm{HCl}(g) \) is cooled to a temperature of \( 271\ \mathrm{K} \). Calculate the new pressure, in atm, of the \( \mathrm{HCl}(g) \).
| Acid \( (\mathrm{HA}) \) | Anion \( (\mathrm{A^-}) \) | \( K_a \) Value |
|---|---|---|
| \( \mathrm{HNO_2} \) | \( \mathrm{NO_2^-} \) | \( 5.6 \times 10^{-4} \) |
| \( \mathrm{HCl} \) | \( \mathrm{Cl^-} \) | \( 2.0 \times 10^{7} \) |
| \( \mathrm{HClO_4} \) | \( \mathrm{ClO_4^-} \) | \( 1.6 \times 10^{15} \) |
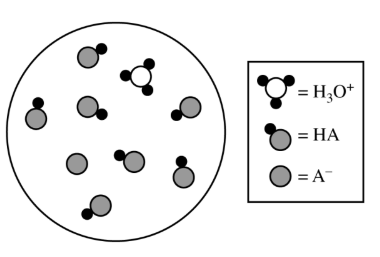
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.7 \) — Solutions and Mixtures (Part \( \mathrm{(b)} \))
• Topic \( 8.6 \) — Molecular Structure of Acids and Bases (Part \( \mathrm{(b)} \))
• Topic \( 8.3 \) — Weak Acid and Base Equilibria (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)(i)
For the correct calculated value:
\( n = \dfrac{PV}{RT} = \dfrac{(7.45\ \mathrm{atm})(6.00\ \mathrm{L})}{(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(296\ \mathrm{K})} = 1.84\ \mathrm{mol} \)
Since pressure, volume, and temperature are given, the ideal gas law directly gives the number of moles of \( \mathrm{HCl}(g) \).
So, the container holds \( \boxed{1.84\ \mathrm{mol}} \) of \( \mathrm{HCl}(g) \).
(a)(ii)
For the correct calculated value:
Accept one of the following:
• \( \dfrac{P_1}{T_1} = \dfrac{P_2}{T_2} \)
\( P_2 = \dfrac{(P_1)(T_2)}{T_1} = \dfrac{(7.45\ \mathrm{atm})(271\ \mathrm{K})}{296\ \mathrm{K}} = 6.82\ \mathrm{atm} \)
• \( P = \dfrac{nRT}{V} = \dfrac{(1.84\ \mathrm{mol})(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(271\ \mathrm{K})}{6.00\ \mathrm{L}} = 6.82\ \mathrm{atm} \)
Because the container is rigid, \( V \) and \( n \) stay constant, so pressure is directly proportional to temperature.
Therefore, the new pressure is \( \boxed{6.82\ \mathrm{atm}} \).
(b)
For a correct drawing:
The drawing should show three water molecules with a hydrogen atom \( (\text{dark circle}) \) oriented toward the \( \mathrm{Cl^-} \) ion.
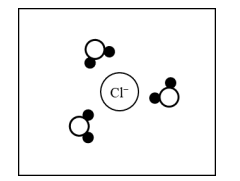
Water is polar, so the partially positive hydrogen ends point toward the negative \( \mathrm{Cl^-} \) ion.
The oxygen ends point away from the anion.
(c)
For the correct answer and a valid justification:
\( \mathrm{HNO_2} \). The diagram shows most of the molecules in their unionized form, indicating a weak acid with a \( K_a \) value less than \( 1 \), which is consistent with \( \mathrm{HNO_2} \).
In the particulate diagram, there are many \( \mathrm{HA} \) particles and only a smaller number of \( \mathrm{H_3O^+} \) and \( \mathrm{A^-} \) ions, so the acid only partially ionizes.
Since \( \mathrm{HCl} \) and \( \mathrm{HClO_4} \) have very large \( K_a \) values, they would ionize much more completely. Thus, the acid shown must be \( \boxed{\mathrm{HNO_2}} \).
Question 6
| Liquid | \( \mathrm{HBr}(l) \) | \( \mathrm{HF}(l) \) |
|---|---|---|
| Intermolecular forces present |
| Liquid | \( \mathrm{HBr}(l) \) | \( \mathrm{HF}(l) \) |
|---|---|---|
| \( \Delta H^\circ_{\mathrm{vap}} \) | \( 17.3\ \mathrm{kJ/mol} \) | \( 25.2\ \mathrm{kJ/mol} \) |
(ii) Calculate the amount of thermal energy, in \( \mathrm{kJ} \), required to vaporize \( 6.85\ \mathrm{g} \) of \( \mathrm{HF}(l) \).
Most-appropriate topic codes (AP Chemistry):
• Topic \( 6.5 \) — Energy of Phase Changes (Part \( \mathrm{(b)(ii)} \))
• Topic \( 1.7 \) — Periodic Trends (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
For the correct answer:
\( \mathrm{HBr}(l) \): London dispersion forces, dipole-dipole attractions
\( \mathrm{HF}(l) \): London dispersion forces, dipole-dipole attractions, hydrogen bonding
Both molecules are polar, so both have dipole-dipole forces and London dispersion forces. Only \( \mathrm{HF} \) has hydrogen bonding because hydrogen is directly bonded to fluorine.
(b)(i)
For a correct explanation:
\( \Delta H^\circ_{\mathrm{vap}} \) is greater for \( \mathrm{HF}(l) \) than \( \mathrm{HBr}(l) \) because the overall intermolecular forces in \( \mathrm{HF}(l) \) are stronger than those in \( \mathrm{HBr}(l) \) due to hydrogen bonding attractions present in \( \mathrm{HF}(l) \). Therefore, more energy is required to separate the molecules in \( \mathrm{HF}(l) \).
Since vaporization requires molecules to overcome intermolecular attractions, the liquid with stronger attractions has the larger \( \Delta H^\circ_{\mathrm{vap}} \).
(b)(ii)
For the correct calculated value:
\( 6.85\ \mathrm{g\ HF} \times \dfrac{1\ \mathrm{mol}}{20.01\ \mathrm{g}} \times \dfrac{25.2\ \mathrm{kJ}}{1\ \mathrm{mol}} = 8.63\ \mathrm{kJ} \)
First convert grams of \( \mathrm{HF} \) to moles, then multiply by \( \Delta H^\circ_{\mathrm{vap}} \).
Therefore, the thermal energy required is \( \boxed{8.63\ \mathrm{kJ}} \).
(c)
For a correct explanation:
\( \mathrm{Br} \) has two additional occupied electron shells \( (n = 3\ \text{and}\ n = 4) \) compared to \( \mathrm{F} \) \( (n = 2) \). The extra electron shells increase the distance between the \( \mathrm{H} \) and \( \mathrm{Br} \) nuclei, giving \( \mathrm{HBr} \) the greater bond length.
In simple terms, bromine is a larger atom than fluorine, so the bonded nuclei in \( \mathrm{HBr} \) are farther apart than in \( \mathrm{HF} \).
Question 7
(ii) Calculate the value of \( K_{sp} \) for \( \mathrm{Sr(OH)}_{2} \).
Most-appropriate topic codes (AP Chemistry):
• Topic \( 7.12 \) — Common-Ion Effect (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
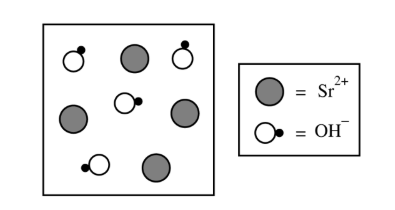
For a correct answer:
Accept one of the following:
• The student’s drawing shows an incorrect ratio of \( \mathrm{Sr}^{2+} \) and \( \mathrm{OH}^{-} \) ions.
• The student’s drawing is not charge-balanced.
Since each \( \mathrm{Sr}^{2+} \) ion must be accompanied by two \( \mathrm{OH}^{-} \) ions, the diagram should show twice as many hydroxide ions as strontium ions.
(b)(i)
For the correct calculated value:
\( 0.043\ \mathrm{mol\ Sr}^{2+}\!/\mathrm{L} \times \dfrac{2\ \mathrm{mol\ OH}^{-}}{1\ \mathrm{mol\ Sr}^{2+}} = 0.086\ \mathrm{M\ OH}^{-} \)
The coefficient \( 2 \) in the dissolution equation means hydroxide concentration is twice the strontium ion concentration.
Therefore, \( \boxed{[\mathrm{OH}^{-}] = 0.086\ \mathrm{M}} \).
(b)(ii)
For the correct calculated value, consistent with part \( \mathrm{(b)(i)} \):
\( K_{sp} = [\mathrm{Sr}^{2+}][\mathrm{OH}^{-}]^{2} = (0.043)(0.086)^{2} = 3.2 \times 10^{-4} \)
Squaring \( [\mathrm{OH}^{-}] \) is important because there are \( 2 \) hydroxide ions in the balanced dissolution equation.
Thus, \( \boxed{K_{sp} = 3.2 \times 10^{-4}} \).
(c)
For the correct answer and a valid justification:
Less than. Because the \( \mathrm{Sr(NO}_{3})_{2}(aq) \) solution already contains a common ion, \( \mathrm{Sr}^{2+}(aq) \), the solubility of \( \mathrm{Sr(OH)}_{2} \) will be decreased, resulting in a lower value of \( [\mathrm{OH}^{-}] \).
Adding extra \( \mathrm{Sr}^{2+} \) shifts the dissolution equilibrium to the left, so less \( \mathrm{Sr(OH)}_{2} \) dissolves and fewer hydroxide ions are produced.
