Question 1
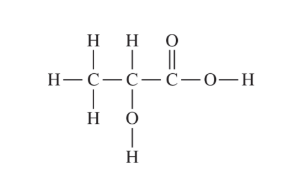
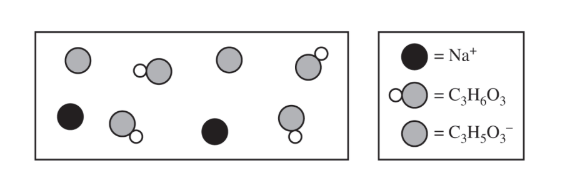
(d) The preceding diagram represents the relative amounts of major species in a sample of the solution in the flask at one point during the titration. (Note that water molecules are omitted.)
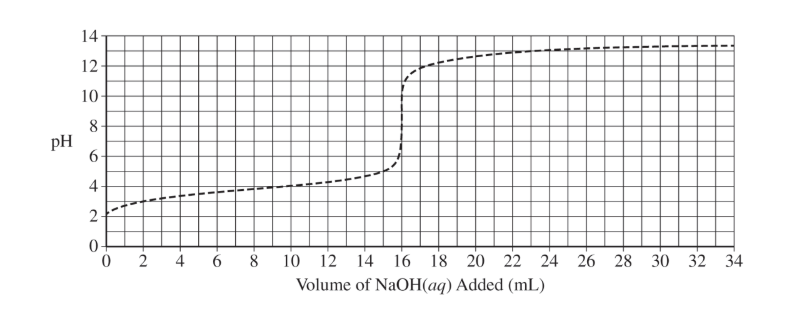
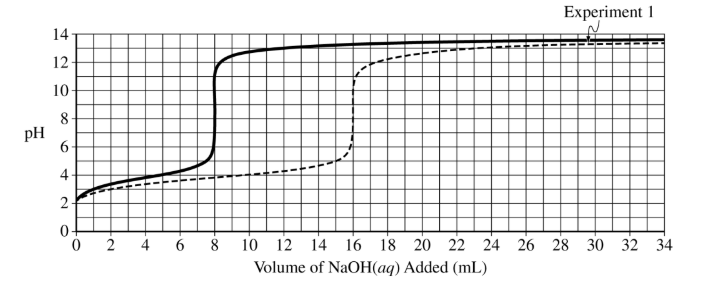
(i) Draw an X on the preceding titration curve at a point in the titration where the reaction mixture would be represented by this diagram.
(ii) Justify your answer.
| Experiment | Mass of \( \mathrm{NaOH(s)} \) (grams) | Volume of Solution (mL) | Titration Curve |
|---|---|---|---|
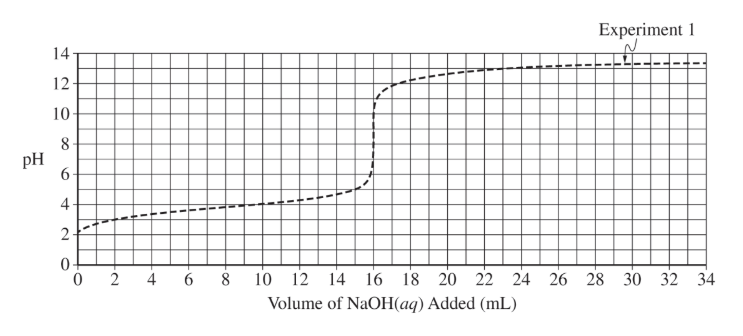
| 1 | \( 10.22 \) | \( 500. \) | Already shown on graph |
| 2 | \( 20.44 \) | \( 500. \) | ? |
(i) Calculate the quantity of heat produced, in J.
(ii) Calculate \( \Delta H_{rxn} \), in \( \mathrm{kJ/mol} \). Include the sign.
(iii) If heat is lost to surroundings, will the calculated \( \Delta H \) be smaller in magnitude? Justify.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.6 \) — Introduction to Titration (Part (b))
• Topic \( 8.5 \) — Acid–Base Titrations (Parts (c), (d))
• Topic \( 6.4 \) — Heat Capacity and Calorimetry (Part (e)(i))
• Topic \( 6.6 \) — Introduction to Enthalpy of Reaction (Parts (e)(ii), (e)(iii))
▶️ Answer/Explanation
(a)
The rightmost hydrogen atom should be circled.
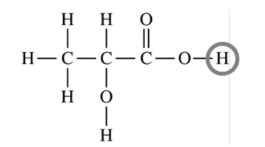
(b)
\( M = \dfrac{10.22}{40.00} \div 0.500 = 0.511\ \mathrm{M} \)
Moles \( = \dfrac{10.22}{40.00} = 0.2555\ \mathrm{mol} \).
(c)
\( pK_a \approx 3.9 \) (acceptable range \( 3.7 – 4.0 \)).
(d)(i)
The X should be at a point greater than or equal to \( 3\ \mathrm{mL} \) and less than \( 8\ \mathrm{mL} \).
(d)(ii)
More acid particles are present than conjugate base particles, meaning the solution is before the half-equivalence point.
(d)(iii)
The equivalence point should be at \( 8\ \mathrm{mL} \). The curve begins similarly but rises earlier due to higher concentration.
(e)(i)
\( q = mc\Delta T = (200.0)(4.2)(3.2) = 2700\ \mathrm{J} \).
(e)(ii)
\( q_{rxn} = -2.7\ \mathrm{kJ} \)
Moles \( = (0.100)(0.500) = 0.0500\ \mathrm{mol} \)
\( \Delta H_{rxn} = \dfrac{-2.7}{0.0500} = -54\ \mathrm{kJ/mol} \).
(e)(iii)
Agree. Heat loss lowers measured temperature change, so calculated \( \Delta H \) is smaller in magnitude.
Question 2
(ii) The \( \mathrm{CO_2(g)} \) produced from the reaction at \( 20.0^\circ \mathrm{C} \) was collected and found to have a pressure of \( 1.25\ \mathrm{atm} \). Calculate the volume of \( \mathrm{CO_2(g)} \), in liters.
(ii) The rate-determining step for the overall reaction is the dissolving of the solids. Would the time required for the dissolving of the solids in the second experiment be longer than, shorter than, or the same as the time required in the first experiment? Justify your answer based on the collisions between particles.
(iii) When the reaction is complete, will the volume of \( \mathrm{CO_2(g)} \) at the end of the second experiment be greater than, less than, or equal to the volume at the end of the first experiment? Justify your answer.
| Trial | Mass of \( \mathrm{H_2C_4H_2O_4} \) (g) | Mass of \( \mathrm{NaHCO_3} \) (g) | Mass of \( \mathrm{CO_2} \) Produced (g) |
|---|---|---|---|
| 1 | \( 0.581 \) | \( 0.627 \) | \( 0.220 \) |
| 2 | \( 1.162 \) | \( 0.627 \) | \( 0.328 \) |
| 3 | \( 1.543 \) | \( 1.251 \) | \( 0.550 \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.4 \) — Ideal Gas Law (Part (a)(ii))
• Topic \( 5.1 \) — Reaction Rates (Parts (b)(i), (b)(iii))
• Topic \( 5.5 \) — Collision Model (Part (b)(ii))
• Topic \( 9.2 \) — Absolute Entropy and Entropy Change (Part (d))
• Topic \( 9.3 \) — Gibbs Free Energy and Thermodynamic Favorability (Part (e))
• Topic \( 8.7 \) — pH and \( pK_a \) (Part (f))
• Topic \( 8.9 \) — Henderson-Hasselbalch Equation (Part (g))
▶️ Answer/Explanation
(a)(i) For the correct calculated value:
\( 0.0114\ \mathrm{mol\ CO_2} \times \dfrac{44.01\ \mathrm{g}}{1\ \mathrm{mol}} = 0.502\ \mathrm{g\ CO_2} \)
(a)(ii) For the correct calculated value:
\( PV = nRT \)
\( V = \dfrac{nRT}{P} = \dfrac{(0.0114\ \mathrm{mol})(0.08206\ \mathrm{L \cdot atm \cdot mol^{-1} \cdot K^{-1}})(293\ \mathrm{K})}{1.25\ \mathrm{atm}} = 0.219\ \mathrm{L} \)
(b)(i) For a correct claim:
The surface area of the solid reactants increases.
(b)(ii) For the correct answer and a valid justification:
Shorter than. The powdered solids have a larger surface area than the solid chunks, so collisions between water and the surface particles occur more frequently, resulting in a faster rate of dissolution and a shorter time to dissolve the solids.
(b)(iii) For the correct answer and a valid justification:
Equal to. Both experiments begin with the same amount of reactants, so they produce the same number of moles of \( \mathrm{CO_2(g)} \) under the same conditions of pressure and temperature; therefore, the final volume is the same.
(c) For the correct answer and a valid justification:
\( \mathrm{NaHCO_3} \) is the limiting reactant because the amount present has the smaller theoretical yield of \( \mathrm{CO_2} \).
\( 1.543\ \mathrm{g\ H_2C_4H_2O_4} \times \dfrac{1\ \mathrm{mol\ H_2C_4H_2O_4}}{116.07\ \mathrm{g}} \times \dfrac{2\ \mathrm{mol\ CO_2}}{1\ \mathrm{mol\ H_2C_4H_2O_4}} = 0.02659\ \mathrm{mol\ CO_2} \)
\( 1.251\ \mathrm{g\ NaHCO_3} \times \dfrac{1\ \mathrm{mol\ NaHCO_3}}{84.01\ \mathrm{g}} \times \dfrac{2\ \mathrm{mol\ CO_2}}{2\ \mathrm{mol\ NaHCO_3}} = 0.01489\ \mathrm{mol\ CO_2} \)
Because \( 0.01489\ \mathrm{mol} < 0.02659\ \mathrm{mol} \), \( \mathrm{NaHCO_3} \) is the limiting reactant.
(d) For a valid explanation:
The entropy change is positive because the reactants produce \( 2 \) moles of gas particles according to the balanced equation. Gas particles are much more dispersed than particles in aqueous solution, so the products have greater disorder and therefore greater entropy.
(e) For the correct answer and a valid justification:
Disagree. Because the reaction is endothermic, \( \Delta H > 0 \), and because \( \Delta S > 0 \), the reaction is thermodynamically favorable only at temperatures high enough that the magnitude of \( -T\Delta S \) is greater than that of \( \Delta H \). Thus, the reaction is not favorable at all temperatures.
(f) For the correct calculated value:
\( pK_{a2} = -\log(8.5 \times 10^{-7}) = 6.07 \)
(g) For the correct calculated value:
\( \mathrm{pH} = pK_{a2} + \log \left( \dfrac{[\mathrm{C_4H_2O_4^{2-}}]}{[\mathrm{HC_4H_2O_4^-}]} \right) \)
\( \dfrac{[\mathrm{C_4H_2O_4^{2-}}]}{[\mathrm{HC_4H_2O_4^-}]} = 10^{(\mathrm{pH} – pK_{a2})} = 10^{(7.00 – 6.07)} = 8.5 \)
Question 3
| Element | Silver \( \mathrm{(Ag)} \) | Copper \( \mathrm{(Cu)} \) |
|---|---|---|
| Atomic radius \( \mathrm{(pm)} \) | \( 165 \) | \( 145 \) |
(ii) Using principles of atomic structure and Coulomb’s law, explain why silver has a larger atomic radius than copper does.
| Before Tarnish Removal | After Tarnish Removal | |
|---|---|---|
| Mass | \( 409.21\ \mathrm{g} \) | \( 398.94\ \mathrm{g} \) |
| Half-Reaction | \( E^\circ \) \( \mathrm{(V)} \) |
|---|---|
| \( \mathrm{Rh^{3+}(aq) + 3\,e^- \rightarrow Rh(s)} \) | \( +0.80 \) |
| \( \mathrm{O_2(g) + 4\,H^+(aq) + 4\,e^- \rightarrow 2\,H_2O(l)} \) | \( +1.23 \) |
(ii) Calculate the value of \( E^\circ_{\mathrm{cell}} \) for the reaction in part \( \mathrm{(d)(i)} \).
(iii) Based on your answer to part \( \mathrm{(d)(ii)} \), explain why this process requires the use of an external power source.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.2 \) — Properties of Solids (Part (b)(i))
• Topic \( 1.7 \) — Periodic Trends (Part (b)(ii))
• Topic \( 4.5 \) — Stoichiometry (Part (c))
• Topic \( 9.8 \) — Galvanic (Voltaic) and Electrolytic Cells (Part (d)(i))
• Topic \( 9.9 \) — Cell Potential and Free Energy (Parts (d)(ii), (d)(iii))
• Topic \( 9.11 \) — Electrolysis and Faraday’s Law (Part (e))
▶️ Answer/Explanation
(a)
For the correct answer:
\( \mathrm{Ag(s)} \): \( 0 \) \( \mathrm{Ag_2S(s)} \): \( +1 \)
An element in its standard form has oxidation number \( 0 \). In \( \mathrm{Ag_2S} \), sulfur is typically \( -2 \), so the two silver atoms together must total \( +2 \), giving each silver atom \( +1 \).
(b)(i)
For a valid explanation:
Sterling silver is better classified as a substitutional alloy because silver and copper have similar atomic radii, \( 165\ \mathrm{pm} \) and \( 145\ \mathrm{pm} \), so copper atoms can replace silver atoms in the metal lattice rather than fitting into small holes between them.
(b)(ii)
For a valid explanation:
Silver has more occupied electron shells \((n=5)\) than copper \((n=4)\), so its valence electrons are farther from the nucleus and experience weaker Coulombic attraction. Therefore, silver has the larger atomic radius.
(c)
For the correct calculated mass of \( \mathrm{Ag_2S} \):
\( 409.21\ \mathrm{g} – 398.94\ \mathrm{g} = 10.27\ \mathrm{g} \)
For the correct calculated moles of silver atoms:
\( 10.27\ \mathrm{g\ Ag_2S} \times \dfrac{1\ \mathrm{mol\ Ag_2S}}{247.80\ \mathrm{g\ Ag_2S}} \times \dfrac{2\ \mathrm{mol\ Ag}}{1\ \mathrm{mol\ Ag_2S}} = 0.08289\ \mathrm{mol\ Ag} \)
So, the number of moles of silver atoms removed is \( \boxed{0.0829\ \mathrm{mol}} \).
(d)(i)
For the correct balanced equation (state symbols not required):
\( \mathrm{4\,Rh^{3+}(aq) + 6\,H_2O(l) \rightarrow 4\,Rh(s) + 3\,O_2(g) + 12\,H^+(aq)} \)
The reduction half-reaction is multiplied by \( 4 \), and the oxidation of water is the reverse of the given oxygen reduction half-reaction, multiplied by \( 3 \), so that \( 12 \) electrons cancel.
(d)(ii)
For the correct calculated value, consistent with part \( \mathrm{(d)(i)} \):
\( E^\circ_{\mathrm{cell}} = 0.80\ \mathrm{V} – 1.23\ \mathrm{V} = -0.43\ \mathrm{V} \)
The plating reaction uses the \( \mathrm{Rh^{3+}/Rh} \) reduction and the oxidation of water.
Because the oxidation potential of water is \( -1.23\ \mathrm{V} \) when written from the reduction table, the total cell potential is negative.
(d)(iii)
For a correct explanation, consistent with part \( \mathrm{(d)(ii)} \):
Since \( E^\circ_{\mathrm{cell}} \) is negative, the reaction is not thermodynamically favorable as written. Therefore, an external power source is required to drive the electroplating process.
(e)
For the correct calculated value of moles of electrons:
\( 2.8\ \mathrm{g\ Rh} \times \dfrac{1\ \mathrm{mol\ Rh}}{102.9\ \mathrm{g\ Rh}} \times \dfrac{3\ \mathrm{mol\ e^-}}{1\ \mathrm{mol\ Rh}} = 0.0816\ \mathrm{mol\ e^-} \)
For the correct calculated value of time:
\( 0.0816\ \mathrm{mol\ e^-} \times \dfrac{96{,}485\ \mathrm{C}}{1\ \mathrm{mol\ e^-}} \times \dfrac{1\ \mathrm{s}}{2.0\ \mathrm{C}} = 3.94\times10^3\ \mathrm{s} \)
Therefore, the time required is approximately \( \boxed{3900\ \mathrm{s}} \).
The small difference between \( 3940\ \mathrm{s} \) and \( 3900\ \mathrm{s} \) is due to rounding.
Question 4
Most-appropriate topic codes (AP Chemistry):
• Topic \( 6.3 \) — Heat Transfer and Thermal Equilibrium (Part (b))
▶️ Answer/Explanation
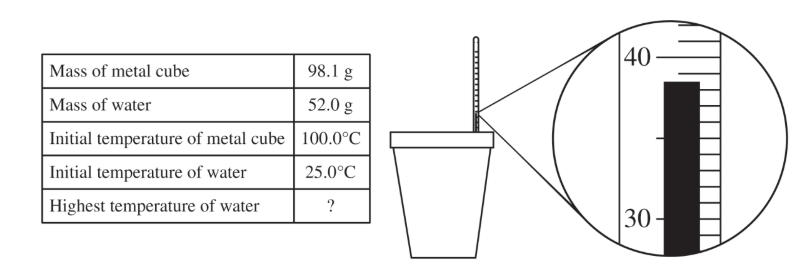
(a)
For the correct answer, reported to the correct decimal place:
\( 38.5^\circ\mathrm{C} \)
The liquid level is halfway between \( 38^\circ\mathrm{C} \) and \( 39^\circ\mathrm{C} \), so the reading is \( 38.5^\circ\mathrm{C} \).
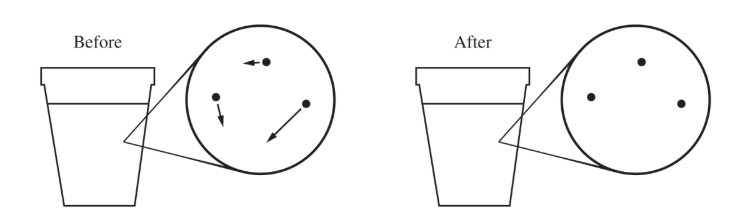
(b)
For a correct drawing:
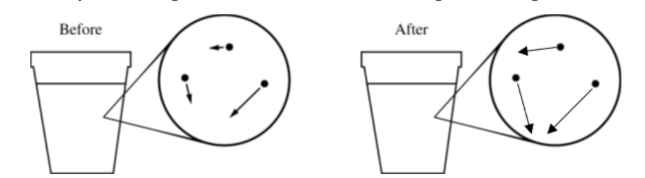
The “After” drawing should contain arrows that are longer, on average.
Because the metal is hotter than the water, thermal energy is transferred to the water, so the average kinetic energy and speed of the water molecules increase.
(c)
For the correct calculated value, consistent with part \( \mathrm{(a)} \):
\( q = mc\Delta T \)
For the metal, \( q_{\mathrm{metal}} = -2940\ \mathrm{J} \) because the metal loses thermal energy.
\( \Delta T_{\mathrm{metal}} = 38.5^\circ\mathrm{C} – 100.0^\circ\mathrm{C} = -61.5^\circ\mathrm{C} \)
\( c_{\mathrm{metal}} = \dfrac{q_{\mathrm{metal}}}{m_{\mathrm{metal}}\Delta T_{\mathrm{metal}}} = \dfrac{-2940\ \mathrm{J}}{(98.1\ \mathrm{g})(38.5^\circ\mathrm{C} – 100.0^\circ\mathrm{C})} = 0.487\ \mathrm{J/(g\cdot^\circ C)} \)
So, the specific heat capacity of the metal is \( \boxed{0.487\ \mathrm{J/(g\cdot^\circ C)}} \).
(d)
For a valid explanation, consistent with part \( \mathrm{(c)} \):
The value of \( \left|\Delta T_{\mathrm{Al}}\right| \) will be smaller because aluminum has a greater specific heat capacity than the metal in the original experiment. Therefore, the same thermal energy transfer applied to the same mass gives a smaller temperature change, according to \( q = mc\Delta T \).
\( q = mc\Delta T \)
\( \left|\Delta T_{\mathrm{Al}}\right| = \left|\dfrac{q_{\mathrm{Al}}}{m_{\mathrm{Al}}c_{\mathrm{Al}}}\right| = \left|\dfrac{-2940\ \mathrm{J}}{(98.1\ \mathrm{g})(0.897\ \mathrm{J/(g\cdot^\circ C)})}\right| = 33.4^\circ\mathrm{C} \)
\( \left|\Delta T_{\mathrm{metal}}\right| = \left|38.5^\circ\mathrm{C} – 100.0^\circ\mathrm{C}\right| = 61.5^\circ\mathrm{C} \)
Thus, \( \left|\Delta T_{\mathrm{Al}}\right| < \left|\Delta T_{\mathrm{metal}}\right| \).
Numerically, \( 33.4^\circ\mathrm{C} < 61.5^\circ\mathrm{C} \).
Question 5
Most-appropriate topic codes (AP Chemistry):
• Topic \( 7.8 \) — Representations of Equilibrium (Part \( \mathrm{(b)(i)} \))
• Topic \( 7.10 \) — Reaction Quotient and Le Châtelier’s Principle (Parts \( \mathrm{(b)(ii)} \), \( \mathrm{(b)(iii)} \))
▶️ Answer/Explanation
(a)
For the correct expression:
\( K_c = \dfrac{[\mathrm{HI}]^2}{[\mathrm{H_2}][\mathrm{I_2}]} \)
The coefficient \( 2 \) in front of \( \mathrm{HI} \) becomes the exponent \( 2 \) in the equilibrium expression.
(b)(i)
For the correct drawing, consistent with part \( \mathrm{(a)} \):
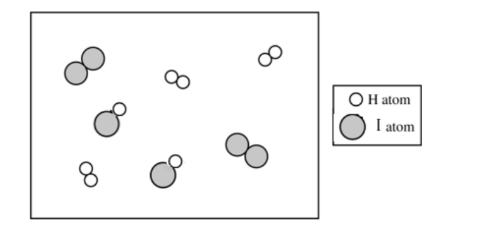
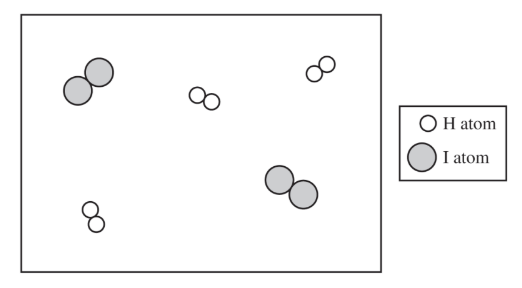
There are \( 2 \) molecules of \( \mathrm{H_2} \) and \( 2 \) molecules of \( \mathrm{I_2} \) shown.
Since \( Q = \dfrac{[\mathrm{HI}]^2}{[\mathrm{H_2}][\mathrm{I_2}]} = 0.67 \), we have \( [\mathrm{HI}]^2 \approx 0.67 \times (2)(2) = 2.68 \), so \( [\mathrm{HI}] \approx \sqrt{2.68} \approx 1.6 \).
Thus, the particulate diagram should show about \( 2 \) molecules of \( \mathrm{HI} \).
(b)(ii)
For a valid hypothesis:
Accept one of the following:
• Decreased the temperature.
• Added more \( \mathrm{H_2} \) and/or \( \mathrm{I_2} \) to the reaction vessel.
A small extra explanation: because the forward reaction is exothermic \(\left(\Delta H_{\mathrm{rxn}} < 0\right)\), decreasing the temperature shifts equilibrium toward products, so the number of moles of \( \mathrm{HI(g)} \) increases.
(b)(iii)
For the correct answer and a valid justification:
Accept one of the following:
• Remain unchanged. The number of moles in the numerator and denominator of \( Q \) (or \( K \)) are equal; changing the volume of the container would not alter the value of \( Q \), which is still equal to \( K \), so the number of moles of \( \mathrm{HI} \) will remain the same.
• Remain unchanged. The increase in volume will decrease the concentration of reactants and products by an equal proportion. Because there are equal moles of gaseous reactants and products in the balanced chemical equation, there is no shift in the equilibrium position, and the number of moles of \( \mathrm{HI} \) will remain the same.
In short, \( \Delta n_{\mathrm{gas}} = 2 – 2 = 0 \), so changing volume at constant temperature does not favor either side.
Question 6
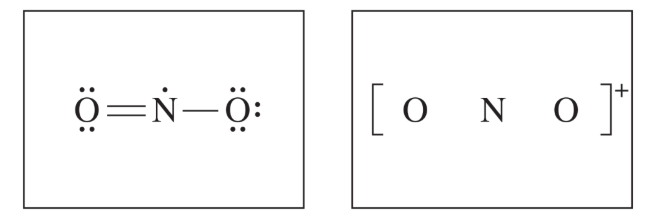
(ii) A student makes the claim that the bond angles in \( \mathrm{NO_2} \) and \( \mathrm{NO_2^+} \) are different from each other. Do you agree or disagree with the student’s claim? Justify your answer.
Most-appropriate topic codes (AP Chemistry):
• Topic \( 4.5 \) — Stoichiometry (Part \( \mathrm{(b)} \))
• Topic \( 2.5 \) — Lewis Diagrams (Part \( \mathrm{(c)(i)} \))
• Topic \( 2.7 \) — VSEPR and Hybridization (Part \( \mathrm{(c)(ii)} \))
▶️ Answer/Explanation
(a)
For a correct explanation:
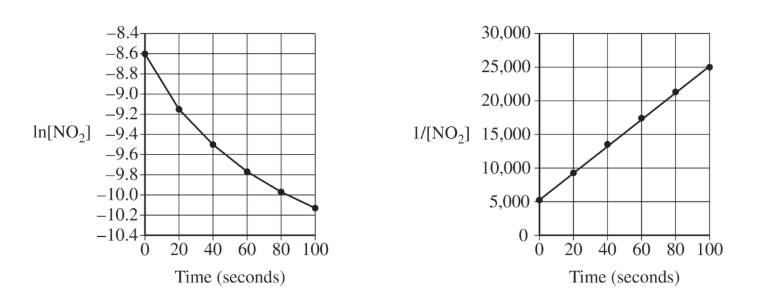
The plot of \( \dfrac{1}{[\mathrm{NO_2}]} \) versus time is the most linear, indicating that the reaction is second order with respect to \( \mathrm{NO_2} \).
For a second-order reaction in one reactant, the integrated rate law predicts a linear relationship between \( \dfrac{1}{[\mathrm{A}]} \) and time, so the straight-line graph supports the claim.
(b)
For the correct calculated value:
From the balanced equation, \( \mathrm{2\,NO_2 \rightarrow 2\,NO + O_2} \)
This means: \( \dfrac{1\ \mathrm{mol\ O_2}}{2\ \mathrm{mol\ NO_2}} \)
Therefore, \( 6.52 \times 10^{-7}\ \mathrm{M/s} \times \dfrac{1\ \mathrm{mol\ O_2}}{2\ \mathrm{mol\ NO_2}} = 3.26 \times 10^{-7}\ \mathrm{M/s} \)
So, the rate of appearance of \( \mathrm{O_2} \) is \( \boxed{3.26 \times 10^{-7}\ \mathrm{M/s}} \).
(c)(i)
For the correct Lewis diagram:
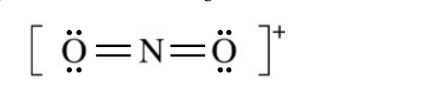
In \( \mathrm{NO_2^+} \), nitrogen forms two double bonds to oxygen and has no unpaired electron.
Each oxygen has two lone pairs, and the entire structure is enclosed in brackets with an overall \( + \) charge.
(c)(ii)
For the correct answer and a valid justification, consistent with part \( \mathrm{(c)(i)} \):
Agree. The angle of \( \mathrm{NO_2^+} \) is different from the angle in \( \mathrm{NO_2} \) because there would no longer be a nonbonding electron on the central atom in \( \mathrm{NO_2^+} \), and the oxygen atoms would spread farther apart, forming a linear structure with a \( 180^\circ \) bond angle.
Another valid way to justify this is by hybridization:
The hybridization of \( \mathrm{N} \) in \( \mathrm{NO_2} \) is \( \mathrm{sp^2} \), which gives a bond angle of approximately \( 120^\circ \). The hybridization of \( \mathrm{N} \) in \( \mathrm{NO_2^+} \) is \( \mathrm{sp} \), which gives a bond angle of \( 180^\circ \).
Question 7
• Balance • \( 100.0\ \mathrm{mL} \) volumetric flask • \( 50.0\ \mathrm{mL} \) graduated cylinder
• Pipet • \( 150\ \mathrm{mL} \) beakers • Chromatography paper
| Step | Step Description and Materials Used |
|---|---|
| 1. | Use the weighing paper and scoop to measure the correct mass of solid \( \mathrm{NaCl} \) on the balance. |
| 2. | |
| 3. | Swirl the mixture to dissolve the solid \( \mathrm{NaCl} \). |
| 4. | |
| 5. | Stopper and invert the mixture several times to ensure that the mixture is homogeneous. |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.9 \) — Separation of Solutions and Mixtures (Part \( \mathrm{(c)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value:
\( 0.1000\ \mathrm{L} \times \dfrac{0.340\ \mathrm{mol}}{1\ \mathrm{L}} \times \dfrac{58.44\ \mathrm{g}}{1\ \mathrm{mol}} = 1.99\ \mathrm{g\ NaCl} \)
First find the moles needed:
\( n = MV = (0.340)(0.1000) = 0.0340\ \mathrm{mol} \)
Then convert moles to grams using the molar mass.
(b)
For a correct description of step \( 2 \):
Combine the solid \( \mathrm{NaCl} \) and some distilled water in a \( 100.0\ \mathrm{mL} \) volumetric flask.
A little extra explanation: only add some water first so the solid can dissolve completely before the flask is filled to the final mark.
For a correct description of step \( 4 \):
Fill the volumetric flask with distilled water to the calibration \((100.0\ \mathrm{mL})\) mark.
The volumetric flask is used because it gives the correct final solution volume more accurately than a beaker or graduated cylinder.
(c)
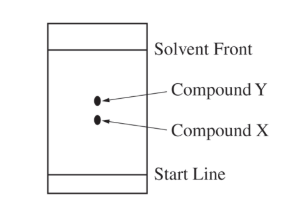
For the correct prediction and a valid explanation:
It would decrease. The solvent front would not travel as far in the second experiment, so the separation would be smaller.
Because the paper is removed sooner, both compounds have less time to move with the solvent. Therefore, the distance between the \( X \) and \( Y \) spots would be reduced.
