Edexcel iGCSE Physics -5.16–5.17 Absolute Zero and the Kelvin Temperature Scale- Study Notes- New Syllabus
Edexcel iGCSE Physics -5.16–5.17 Absolute Zero and the Kelvin Temperature Scale- Study Notes- New syllabus
Edexcel iGCSE Physics -5.16–5.17 Absolute Zero and the Kelvin Temperature Scale- Study Notes -Edexcel iGCSE Physics – per latest Syllabus.
Key Concepts:
5.16 understand why there is an absolute zero of temperature, which is −273 °C
5.17 describe the Kelvin scale of temperature and be able to convert between the Kelvin and Celsius scales
Absolute Zero
Absolute zero is the lowest possible temperature. It has a value of –273 °C (or 0 K on the Kelvin scale).
At absolute zero, particles have the minimum possible kinetic energy.
Particle Explanation of Absolute Zero
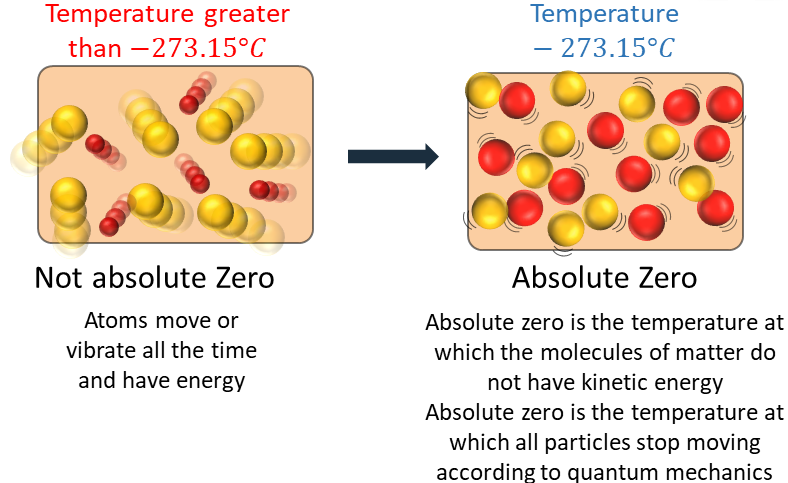
- Temperature is a measure of the average kinetic energy of particles.
- As temperature decreases, particles move more slowly.
- At absolute zero, particle motion is at its minimum.
- Particles cannot have less kinetic energy than this.
This means temperatures below –273 °C are impossible.
Why Absolute Zero Exists
- Particles cannot have zero or negative kinetic energy.
- Cooling removes kinetic energy from particles.
- Eventually, no more kinetic energy can be removed.
Therefore, there is a natural lower limit to temperature.
Absolute Zero and the Kelvin Scale
![]()
- The Kelvin scale starts at absolute zero.
- 0 K = –273 °C.
- There are no negative values on the Kelvin scale.
\( \mathrm{T(K) = T(^\circ C) + 273} \)
Evidence for Absolute Zero
- As gases are cooled, pressure decreases.
- If pressure is plotted against temperature, it reaches zero at –273 °C.
- This shows molecular motion would stop at this temperature.
Key Idea
- Temperature is linked to particle motion.
- Absolute zero is when kinetic energy is minimal.
- It is the lowest possible temperature.
Important Points to Remember
- Absolute zero is –273 °C or 0 K.
- Particles still exist but have minimum motion.
- Negative Kelvin temperatures do not exist.
Example
Explain why it is impossible to cool a substance below –273 °C.
▶️ Answer / Explanation
Temperature depends on the kinetic energy of particles.
At –273 °C, particles have the minimum possible kinetic energy.
They cannot have less energy than this, so the temperature cannot go any lower.
Example
A gas is cooled at constant volume.
Explain what happens to the pressure as the temperature approaches absolute zero.
▶️ Answer / Explanation
As temperature decreases, gas molecules move more slowly.
Collisions with the container walls become less frequent and less forceful.
The pressure decreases and would reach zero at absolute zero.
The Kelvin Temperature Scale
The Kelvin scale is an absolute temperature scale used in physics. It is based on the idea that there is a lowest possible temperature, known as absolute zero.
The Kelvin scale is particularly important in scientific work because it is directly linked to the motion and energy of particles.
Key Features of the Kelvin Scale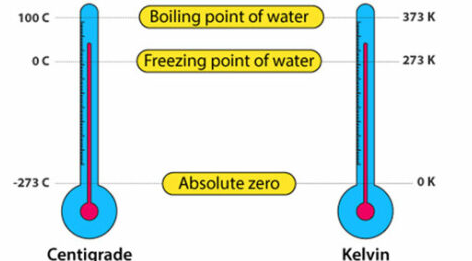
- The unit of temperature is the kelvin (K).
- The scale starts at absolute zero.
- There are no negative temperatures on the Kelvin scale.
- Temperature intervals are the same size as on the Celsius scale.
Absolute zero:
- 0 K = –273 °C
- Particles have minimum possible kinetic energy.
Comparison with the Celsius Scale
- Celsius scale is based on water freezing at 0 °C and boiling at 100 °C.
- Kelvin scale is based on particle energy.
- The size of 1 kelvin is equal to the size of 1 degree Celsius.
This means a temperature change of 1 K is the same as a temperature change of 1 °C.
Conversion Between Celsius and Kelvin
The relationship between the two scales is:
\( \mathrm{T(K) = T(^\circ C) + 273} \)
And:
\( \mathrm{T(^\circ C) = T(K) – 273} \)
Why Kelvin Is Used in Physics
- It avoids negative temperatures.
- It is proportional to the average kinetic energy of particles.
- It is required in gas laws and thermal equations.
Key Idea
- The Kelvin scale starts at absolute zero.
- Kelvin and Celsius have equal-sized units.
- Conversion is done by adding or subtracting 273.
Important Points to Remember
- Always use Kelvin in gas law calculations.
- Do not use the degree symbol (°) with kelvin.
- Temperature differences are the same in K and °C.
Example
Convert a temperature of \( \mathrm{25^\circ C} \) into kelvin.
▶️ Answer / Explanation
Use:
\( \mathrm{T(K) = T(^\circ C) + 273} \)
\( \mathrm{T = 25 + 273} \)
\( \mathrm{T = 298\ K} \)
Example
A gas has a temperature of \( \mathrm{320\ K} \).
Convert this temperature into degrees Celsius.
▶️ Answer / Explanation
Use:
\( \mathrm{T(^\circ C) = T(K) – 273} \)
\( \mathrm{T = 320 – 273} \)
\( \mathrm{T = 47^\circ C} \)
