Edexcel iGCSE Physics -5.18–5.19 Temperature, Molecular Speed, and Kinetic Energy- Study Notes- New Syllabus
Edexcel iGCSE Physics -5.18–5.19 Temperature, Molecular Speed, and Kinetic Energy- Study Notes- New syllabus
Edexcel iGCSE Physics -5.18–5.19 Temperature, Molecular Speed, and Kinetic Energy- Study Notes -Edexcel iGCSE Physics – per latest Syllabus.
Key Concepts:
5.18 understand why an increase in temperature results in an increase in the average speed of gas molecules
5.19 know that the Kelvin temperature of a gas is proportional to the average kinetic energy of its molecules
Temperature and the Speed of Gas Molecules
Gas molecules are in constant random motion. The temperature of a gas is directly related to the average kinetic energy of its molecules.
Link Between Temperature and Kinetic Energy
- Temperature measures the average kinetic energy of gas molecules.
- Kinetic energy depends on the speed of the molecules.
- Higher kinetic energy means higher average speed.
Therefore, when temperature increases, the average speed of gas molecules increases.
What Happens When a Gas Is Heated
- Heating transfers energy to the gas.
- This energy increases the internal energy of the gas.
- The kinetic energy of molecules increases.
- Molecules move faster.
Effect on Molecular Motion
- Molecules collide more frequently.
- Collisions are more forceful.
- This leads to increased gas pressure if volume is constant.
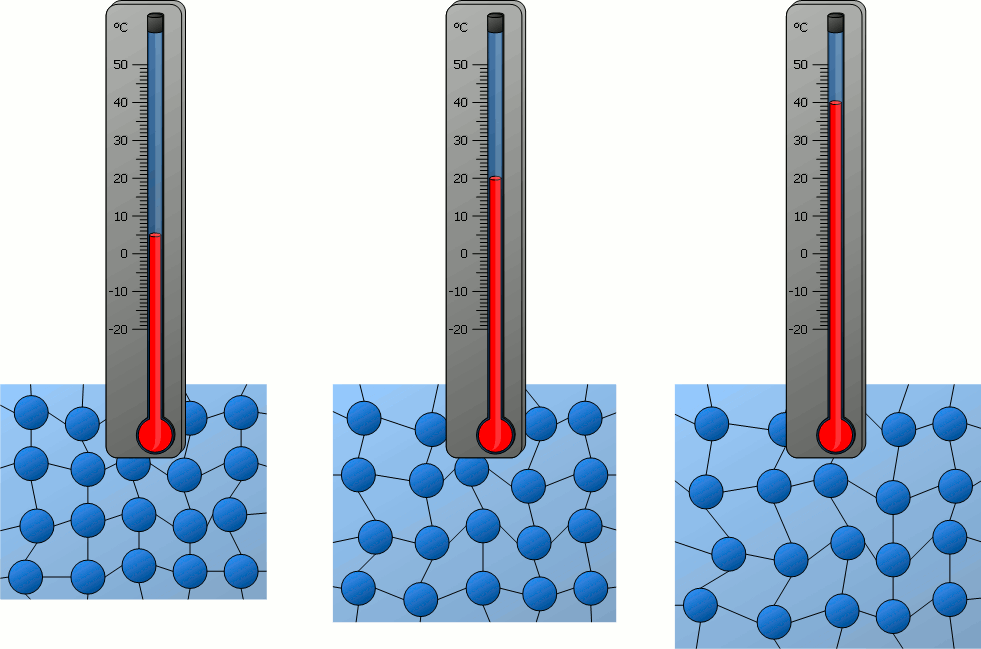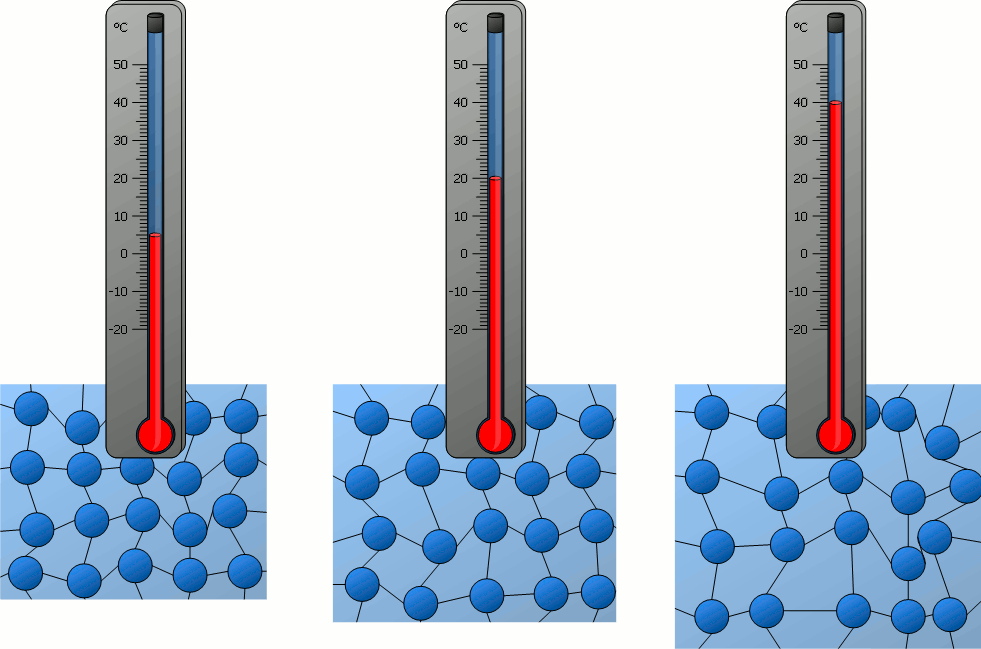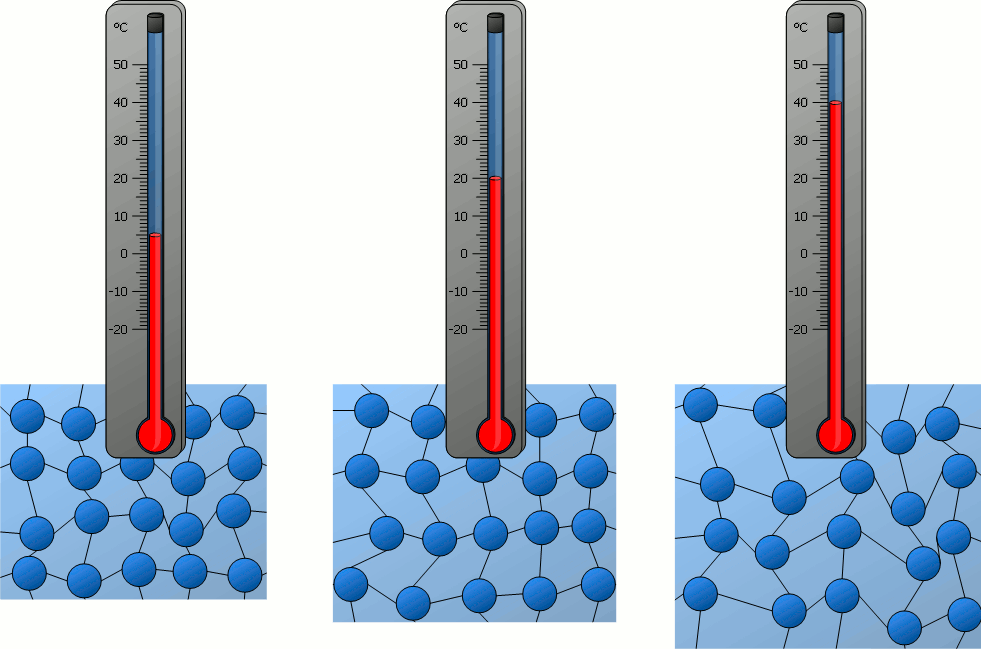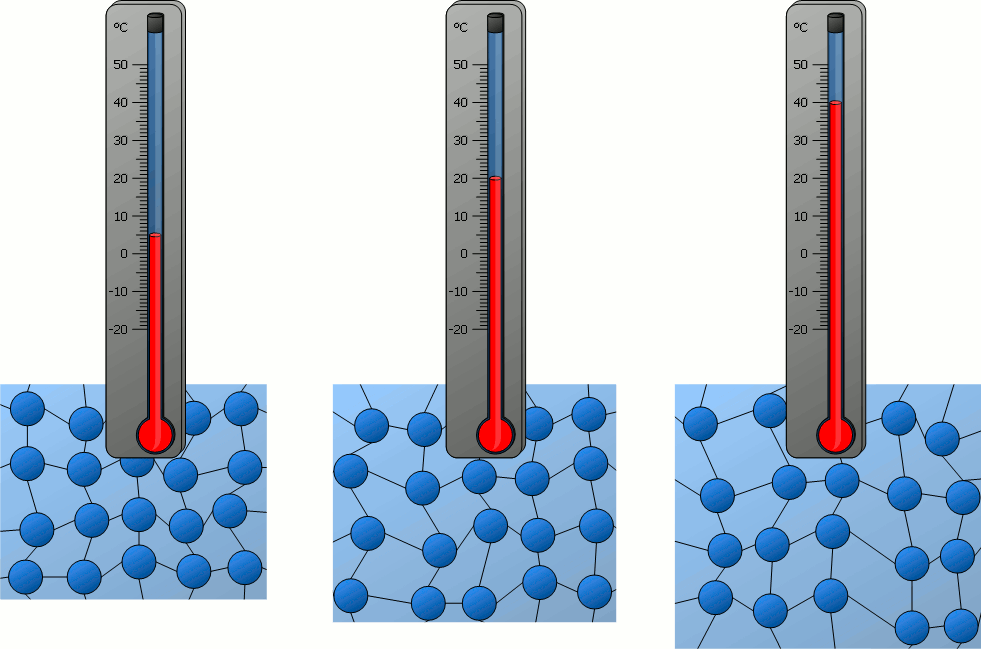
Particle Model Explanation
- Molecules gain energy when heated.
- The energy increases their speed, not their size.
- All molecules move faster on average, even though individual speeds differ.
Key Idea
- Temperature is a measure of average kinetic energy.
- Higher temperature → higher average speed.
- Gas behaviour is explained by particle motion.
Important Points to Remember
- Average speed increases, not necessarily every molecule’s speed equally.
- Kelvin temperature is used in gas laws.
- Faster molecules cause greater pressure.
Example
Explain why the pressure of a gas in a sealed container increases when the gas is heated.
▶️ Answer / Explanation
Heating increases the kinetic energy of gas molecules.
The molecules move faster and collide with the container walls more often and with greater force.
This increases the pressure.
Example
A gas is heated while its volume is kept constant.
Explain what happens to the average speed of the molecules.
▶️ Answer / Explanation
The temperature increases, which increases the average kinetic energy of the molecules.
As a result, the molecules move faster on average.
Kelvin Temperature and Kinetic Energy of Gas Molecules
The Kelvin temperature of a gas is directly related to the average kinetic energy of its molecules. As the Kelvin temperature increases, the average kinetic energy of the molecules increases in the same proportion.
Key Relationship
- Kelvin temperature is a measure of average kinetic energy.
- Higher Kelvin temperature → greater average kinetic energy.
- If the Kelvin temperature doubles, the average kinetic energy doubles.
This proportionality is why the Kelvin scale is used in gas laws.
Why the Kelvin Scale Is Used (Not Celsius)
- Kelvin scale starts at absolute zero (0 K).
- At 0 K, average kinetic energy is at its minimum.
- Celsius temperatures can be negative, but kinetic energy cannot.
Important: A change of 1 K is the same size as a change of 1 °C, but proportional comparisons must use kelvin.
Particle Model Explanation
- Gas molecules are in constant random motion.
- Heating transfers energy to the gas.
- This energy increases the kinetic energy of molecules.
- Molecules move faster on average.
Evidence for Proportionality
- When a gas is heated at constant volume, pressure increases.
- This happens because faster molecules collide more forcefully.
- Pressure is proportional to Kelvin temperature.
Key Idea
- Kelvin temperature ∝ average kinetic energy.
- Use kelvin for gas behaviour.
- Absolute zero corresponds to minimum kinetic energy.
Important Points to Remember
- Always convert °C to K when comparing energies.
- Doubling °C does NOT double kinetic energy.
- Doubling K DOES double kinetic energy.
Example
The temperature of a gas increases from \( \mathrm{300\ K} \) to \( \mathrm{600\ K} \).
Describe what happens to the average kinetic energy of the gas molecules.
▶️ Answer / Explanation
The Kelvin temperature doubles.
Therefore, the average kinetic energy of the gas molecules also doubles.
Example
A gas is heated from \( \mathrm{27^\circ C} \) to \( \mathrm{127^\circ C} \).
Explain why the average kinetic energy does not increase by a factor of 4.
▶️ Answer / Explanation
The temperatures must be converted to kelvin.
\( \mathrm{27^\circ C = 300\ K} \) and \( \mathrm{127^\circ C = 400\ K} \).
The Kelvin temperature increases by a factor of \( \mathrm{\frac{4}{3}} \), not 4.
So the average kinetic energy increases by the same factor.
