Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.1 Chemical terms and formulae-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.1 Chemical terms and formulae- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.1 Chemical terms and formulae- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.1 know the terms ‘atom’, ‘element’, ‘ion’, ‘molecule’, ‘compound’, ’empirical formula’ and ‘molecular formula’
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.1 Key Chemical Terms
Understanding the language of chemistry is essential. These terms describe the particles that make up matter and the different ways atoms combine to form substances.
Atom
An atom is the smallest particle of an element that can exist and still retain the chemical properties of that element.
Atoms are the fundamental building blocks of matter. Each atom contains a nucleus made of protons and neutrons, with electrons arranged in shells around the nucleus.
Subatomic Particles
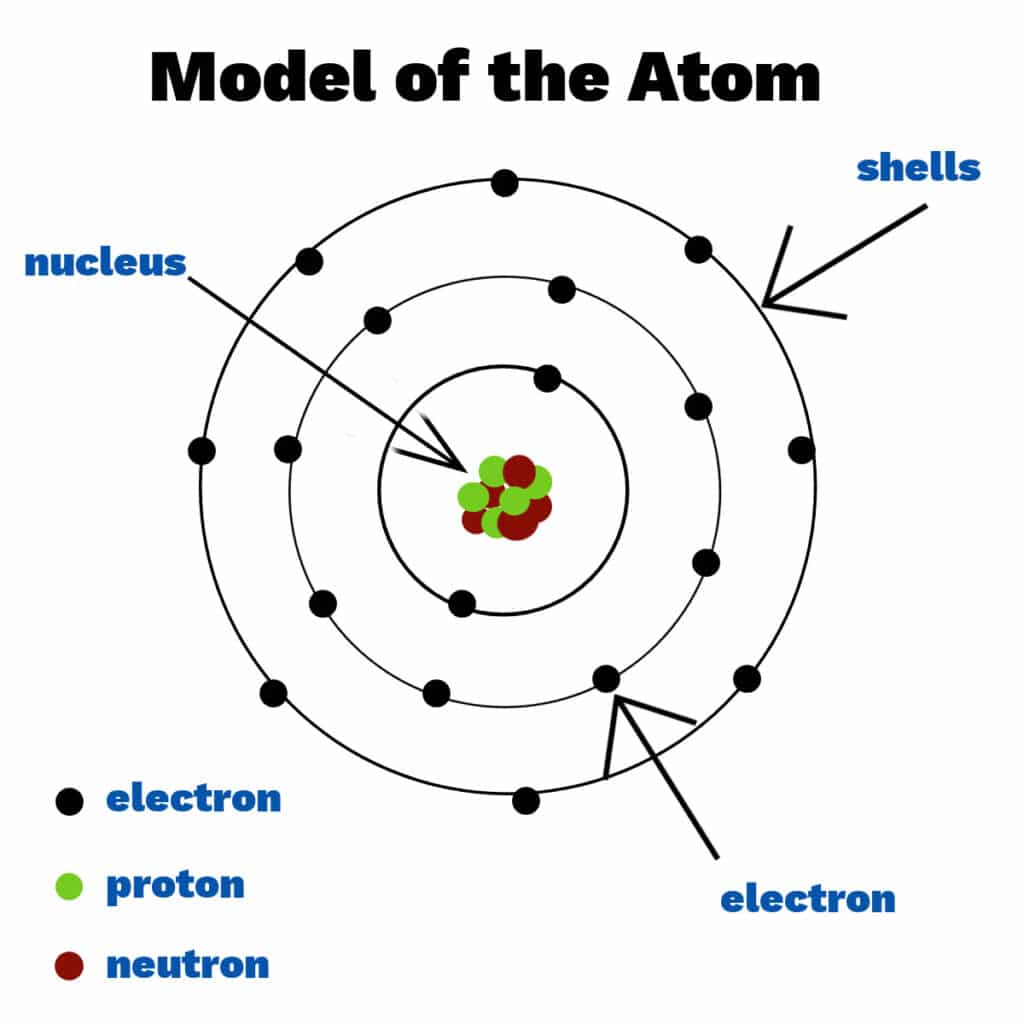
| Particle | Charge | Relative Mass | Location |
|---|---|---|---|
| Proton | +1 | 1 | Nucleus |
| Neutron | 0 | 1 | Nucleus |
| Electron | −1 | 1/1836 | Electron shells |
Examples of atoms
- Hydrogen atom \( \mathrm{H} \)
- Oxygen atom \( \mathrm{O} \)
- Sodium atom \( \mathrm{Na} \)
Element
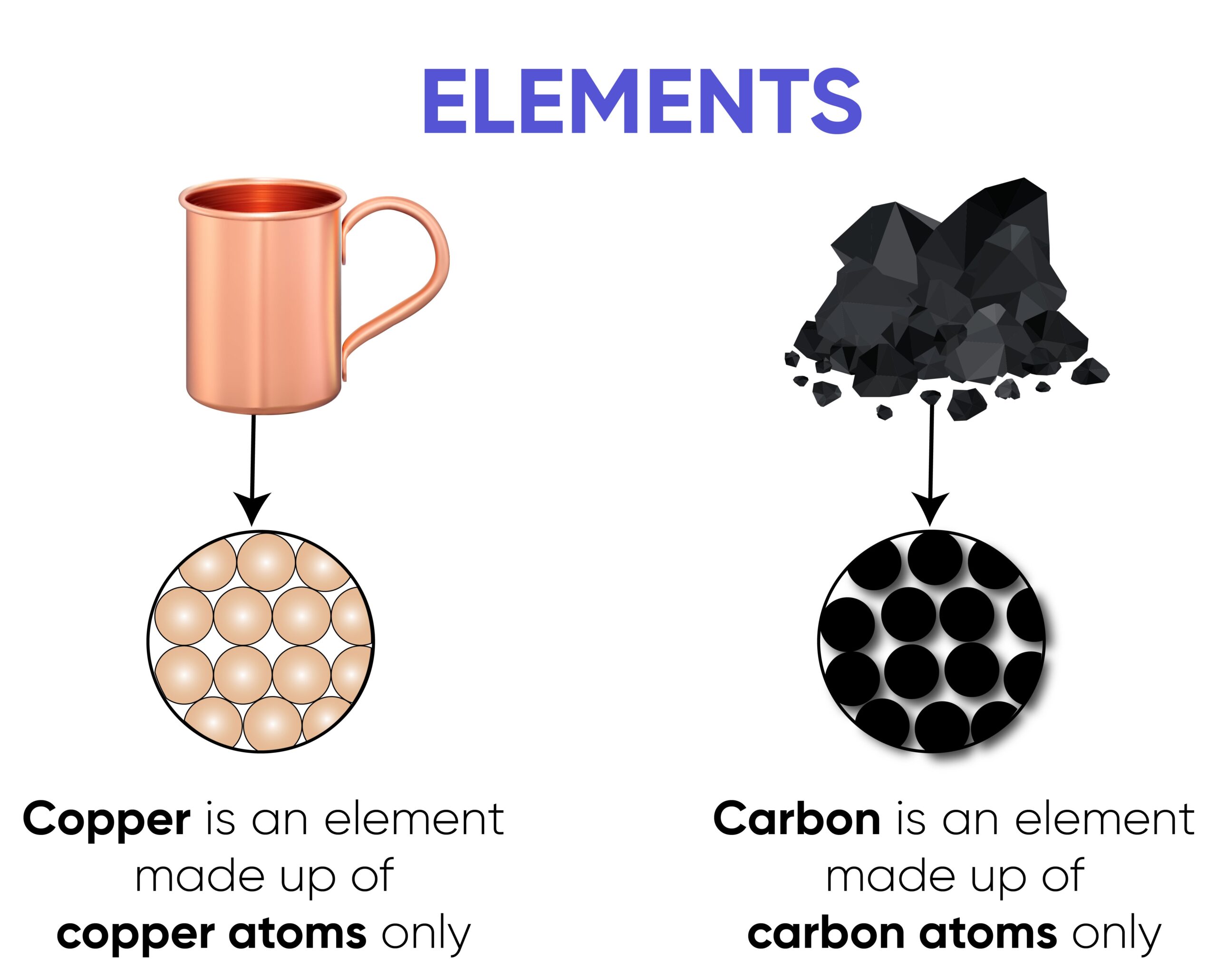
An element is a substance made of only one type of atom.
All atoms of an element contain the same number of protons, known as the atomic number.
Examples
- Hydrogen \( \mathrm{H} \)
- Oxygen \( \mathrm{O} \)
- Neon \( \mathrm{Ne} \)
Ion
An ion is a charged particle formed when an atom or group of atoms gains or loses electrons.
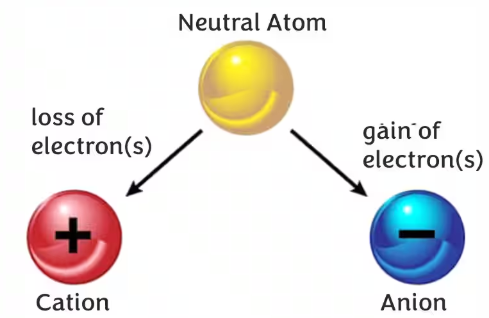
Loss of electrons forms a cation (positive ion), while gain of electrons forms an anion (negative ion).
Examples
- \( \mathrm{Na^+} \) sodium ion
- \( \mathrm{Cl^-} \) chloride ion
- \( \mathrm{Mg^{2+}} \) magnesium ion
Molecule
A molecule is a group of two or more atoms covalently bonded together.
Molecules may contain atoms of the same element or different elements.
Examples
- Oxygen molecule \( \mathrm{O_2} \)
- Nitrogen molecule \( \mathrm{N_2} \)
- Water molecule \( \mathrm{H_2O} \)
Compound
A compound is a substance formed when atoms of two or more different elements are chemically bonded together in fixed proportions.
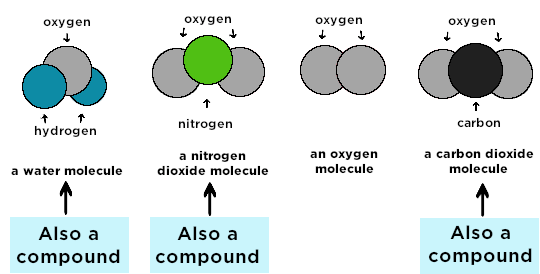
Examples
- Water \( \mathrm{H_2O} \)
- Carbon dioxide \( \mathrm{CO_2} \)
- Sodium chloride \( \mathrm{NaCl} \)
Empirical Formula
The empirical formula shows the simplest whole-number ratio of atoms of each element in a compound.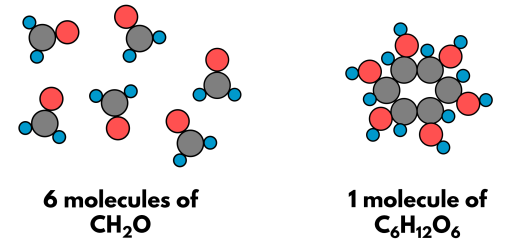
Example: \( \mathrm{C_6H_{12}O_6} \rightarrow \mathrm{CH_2O} \)
Molecular Formula
The molecular formula shows the actual number of atoms of each element present in a molecule.
Examples: \( \mathrm{C_2H_4} \), \( \mathrm{C_6H_{12}O_6} \)
Example 1:
Identify whether each of the following represents an element, molecule, ion, or compound: \( \mathrm{O_2} \), \( \mathrm{Na^+} \), \( \mathrm{CO_2} \), \( \mathrm{Ne} \).
▶️ Answer/Explanation
\( \mathrm{O_2} \) → molecule (two oxygen atoms covalently bonded)
\( \mathrm{Na^+} \) → ion (sodium atom that has lost one electron)
\( \mathrm{CO_2} \) → compound and molecule (contains atoms of different elements chemically bonded)
\( \mathrm{Ne} \) → element (a single neon atom)
Conclusion: A molecule must consist of atoms covalently bonded together, while a compound must contain atoms of two or more different elements.
Example 2:
A compound has the molecular formula \( \mathrm{C_6H_{12}} \). Determine its empirical formula.
▶️ Answer/Explanation
The ratio of atoms is
C : H = 6 : 12
Divide both numbers by the smallest value (6).
C : H = 1 : 2
Therefore the empirical formula is
\( \mathrm{CH_2} \)
Conclusion: The empirical formula shows the simplest ratio of atoms present in the compound.
