Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.10 Determining formulae /equations experimentally-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.10 Determining formulae /equations experimentally- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.10 Determining formulae /equations experimentally- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.10 be able to determine a formula or confirm an equation by experiment, including evaluation of the data
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.10 Determining Formulae and Confirming Equations by Experiment
Experimental measurements can be used to determine the formula of a compound or to confirm whether a proposed chemical equation is correct. This involves measuring masses or volumes during a reaction and comparing the experimental results with theoretical predictions.
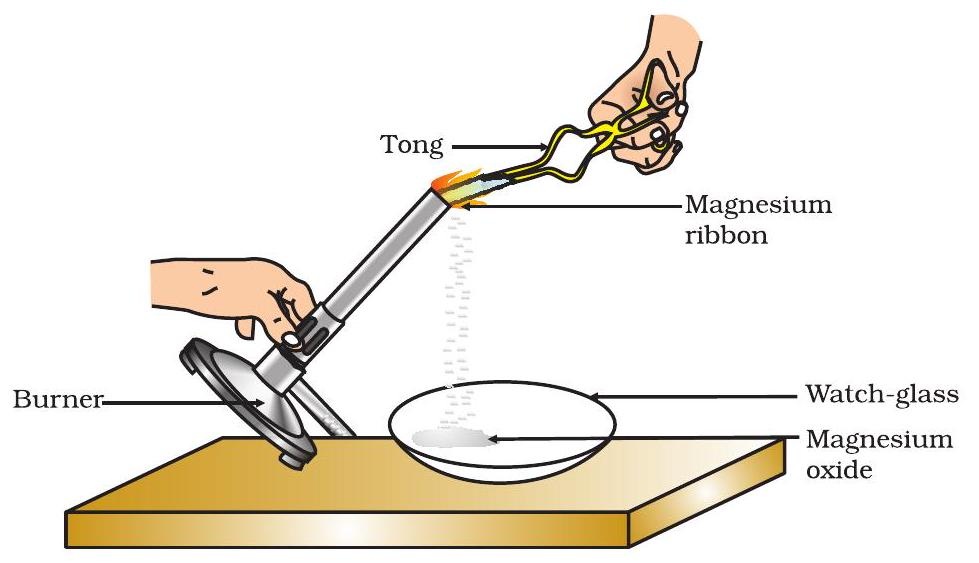
Determining a Chemical Formula Experimentally
The formula of a compound can be determined by measuring the masses of elements that react together or the masses of products formed.
From the experimental data, the number of moles of each element can be calculated and used to determine the simplest ratio of atoms in the compound.
Example: Determining the Formula of Magnesium Oxide
Magnesium reacts with oxygen when heated in air to form magnesium oxide.
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
In an experiment, the mass of magnesium is measured before heating and the mass of magnesium oxide is measured after heating. The increase in mass corresponds to the oxygen that has reacted.
Using these masses, the moles of magnesium and oxygen can be calculated and the ratio of atoms determined to confirm the formula \( \mathrm{MgO} \).
Confirming a Chemical Equation
Experimental data can also be used to confirm whether a balanced chemical equation is correct by comparing measured quantities with theoretical predictions.
For example, the volume of gas produced or the mass of product formed in an experiment can be compared with values calculated using the balanced equation.
Evaluation of Experimental Data
When analysing experimental results, it is important to evaluate the reliability of the data.
Differences between experimental and theoretical results may occur due to:
- measurement errors
- loss of product during the experiment
- incomplete reactions
- impurities in the reactants
Evaluating these factors helps determine whether the experimental results support the proposed formula or chemical equation.
Example 1
A student heats \( 0.48\ \mathrm{g} \) of magnesium in air and obtains \( 0.80\ \mathrm{g} \) of magnesium oxide. Determine whether the experimental data supports the formula \( \mathrm{MgO} \).
▶️ Answer/Explanation
Step 1: Calculate mass of oxygen
Mass of oxygen = \( 0.80 – 0.48 = 0.32\ \mathrm{g} \)
Step 2: Convert masses to moles
\( n(\mathrm{Mg}) = \dfrac{0.48}{24} = 0.020 \)
\( n(\mathrm{O}) = \dfrac{0.32}{16} = 0.020 \)
Step 3: Determine ratio
Mg : O = \( 1 : 1 \)
This ratio confirms the formula \( \mathrm{MgO} \).
Example 2
In an experiment, a reaction is predicted to produce \( 24\ \mathrm{dm^3} \) of hydrogen gas at RTP, but only \( 22\ \mathrm{dm^3} \) is measured. Suggest reasons for the difference.
▶️ Answer/Explanation
- Some hydrogen gas may have escaped during the experiment.
- The reaction may not have gone to completion.
- Experimental measurement errors may have occurred.
These factors could explain why the experimental result is lower than the theoretical prediction.
