Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.11 CORE PRACTICAL 1: Molar volume of a gas-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.11 CORE PRACTICAL 1: Molar volume of a gas- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.11 CORE PRACTICAL 1: Molar volume of a gas- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.11 CORE PRACTICAL 1 Measurement of the molar volume of a gas
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.11 Core Practical 1 – Measurement of the Molar Volume of a Gas
Aim
To determine the volume occupied by one mole of carbon dioxide gas by reacting calcium carbonate with ethanoic acid.
Background
The molar volume of any gas at a given temperature and pressure is the same for all gases.
By collecting the carbon dioxide gas produced in a reaction and measuring its volume, the molar volume of the gas can be determined using experimental data.
Overall Reaction
\( \mathrm{CaCO_3(s) + 2CH_3COOH(aq) \rightarrow Ca(CH_3COO)_2(aq) + H_2O(l) + CO_2(g)} \)
Safety
- Wear eye protection.
- Avoid skin contact with ethanoic acid.
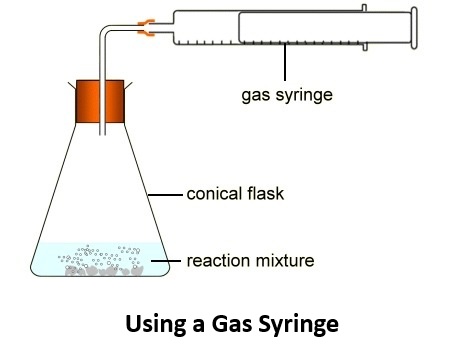
- If the delivery tube becomes blocked, remove the bung and clear the blockage before restarting the experiment.
Apparatus
- boiling tube or conical flask
- bung with delivery tube
- stand and clamp
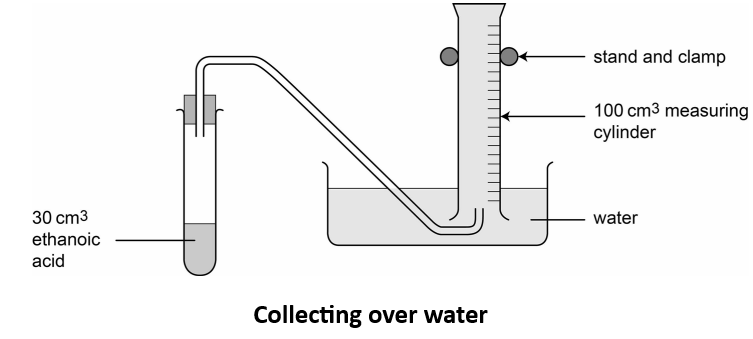
- gas syringe or water displacement apparatus
- 100 cm3 and 50 cm3 measuring cylinders
- test tube
- 2 d.p. balance
- \( 1\ \mathrm{mol\ dm^{-3}} \) ethanoic acid
- powdered calcium carbonate
Method
- Measure \( 30\ \mathrm{cm^3} \) of \( 1\ \mathrm{mol\ dm^{-3}} \) ethanoic acid and place it in a conical flask.
- Attach the flask to a gas syringe or set up the water displacement apparatus.
- Weigh a boiling tube containing approximately \( 0.5\ \mathrm{g} \) of calcium carbonate.
- Add the calcium carbonate to the acid and immediately fit the bung so no gas escapes.
- Allow the reaction to proceed and record the total volume of carbon dioxide gas produced.
- Reweigh the empty boiling tube to determine the exact mass of calcium carbonate used.
- Repeat the experiment several times using increasing masses of calcium carbonate.
Processing Results
The mass of calcium carbonate is converted into moles using:
\( n = \dfrac{m}{M} \)
From the balanced equation, one mole of calcium carbonate produces one mole of carbon dioxide:
\( \mathrm{CaCO_3 : CO_2 = 1 : 1} \)
A graph of volume of CO₂ produced against mass of CaCO₃ used is plotted. The graph should be a straight line through the origin.
From the graph, the volume of carbon dioxide produced by one mole of calcium carbonate can be determined.
Evaluation and Sources of Error
- Gas may escape before the bung is fitted.
- The bung may not be completely airtight.
- Carbon dioxide is slightly soluble in water, which can reduce the measured volume.
- Some gas may escape while adding calcium carbonate.
- Gas syringe plungers may not move freely, affecting readings.
Example Calculation
Suppose \( 0.25\ \mathrm{g} \) of calcium carbonate produces \( 61\ \mathrm{cm^3} \) of carbon dioxide.
▶️ Answer/Explanation
Step 1: Calculate moles of calcium carbonate
\( n(\mathrm{CaCO_3}) = \dfrac{0.25}{100.1} = 0.0025\ \mathrm{mol} \)
Moles of \( \mathrm{CO_2} = 0.0025\ \mathrm{mol} \)
Step 2: Calculate molar volume
Molar volume \( = \dfrac{61}{0.0025} \)
\( = 24.4\ \mathrm{dm^3\ mol^{-1}} \)
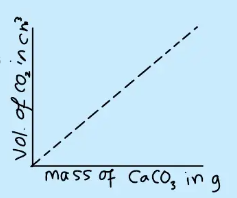
This value is close to the accepted molar volume of gases at room temperature (\( \approx 24\ \mathrm{dm^3\ mol^{-1}} \)).
