Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.12 Ionic equations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.12 Ionic equations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.12 Ionic equations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.12 be able to relate ionic and full equations, with state symbols, to observations from simple test-tube experiments, to include:
i displacement reactions
ii typical reactions of acids
iii precipitation reactions
Further suggested practicals:
i preparation of a salt and calculating the percentage yield of product, including the preparation of a double salt, such as ammonium iron(II) sulfate from iron, ammonia and sulfuric acid
ii determine a chemical formula by experiment, such as the formula of copper(II) oxide by reduction
iii determine a chemical equation by experiment, such as the reaction between lithium and water, or the reaction between magnesium and an acid
iv carry out and interpret the results of simple test-tube reactions, as outlined in 1.12
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.12 Relating Ionic and Full Equations to Observations
In many chemical reactions, especially those carried out in aqueous solution, the full equation and the ionic equation can be used to explain the observations seen in a simple test-tube experiment.
Typical observations include colour changes, formation of precipitates, gas production, or displacement of metals.
Displacement Reactions
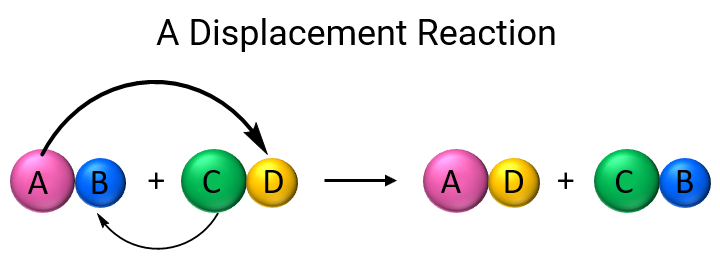
A displacement reaction occurs when a more reactive metal displaces a less reactive metal from its compound in solution.
Example
Zinc placed in copper(II) sulfate solution displaces copper.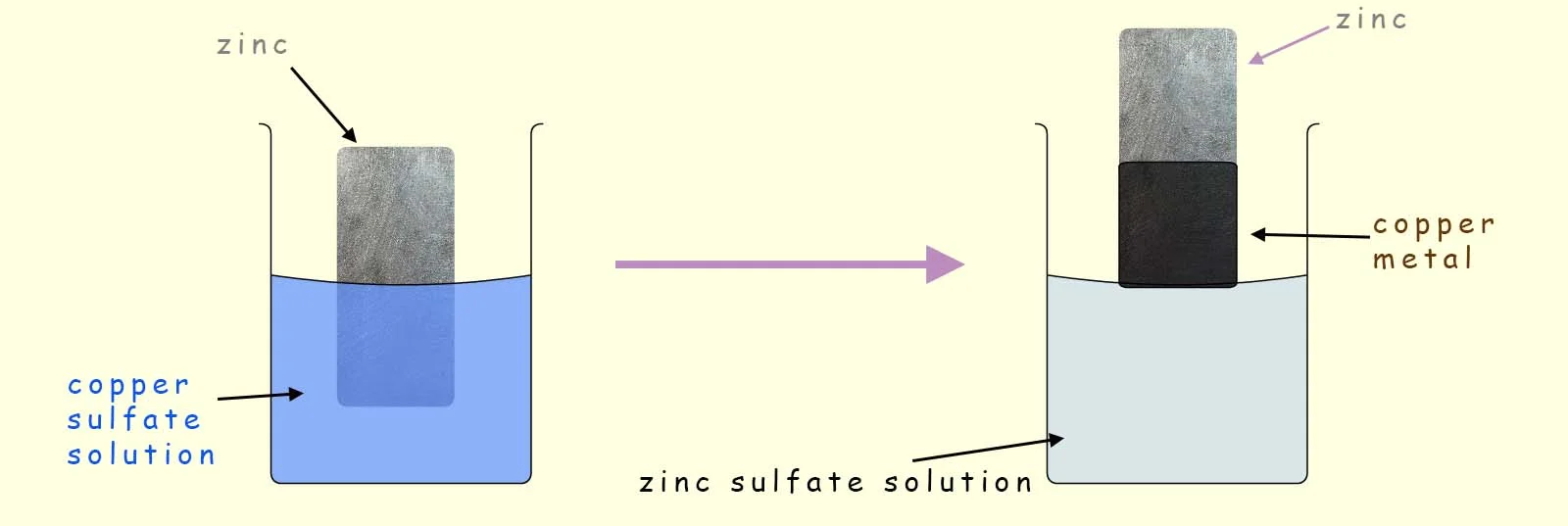
Full equation:
\( \mathrm{Zn(s) + CuSO_4(aq) \rightarrow ZnSO_4(aq) + Cu(s)} \)
Ionic equation:
\( \mathrm{Zn(s) + Cu^{2+}(aq) \rightarrow Zn^{2+}(aq) + Cu(s)} \)
Observation
- Blue copper(II) sulfate solution becomes colourless.
- A reddish-brown copper solid forms on the zinc.
Typical Reactions of Acids
Acids react with metals, metal oxides, metal hydroxides, and carbonates.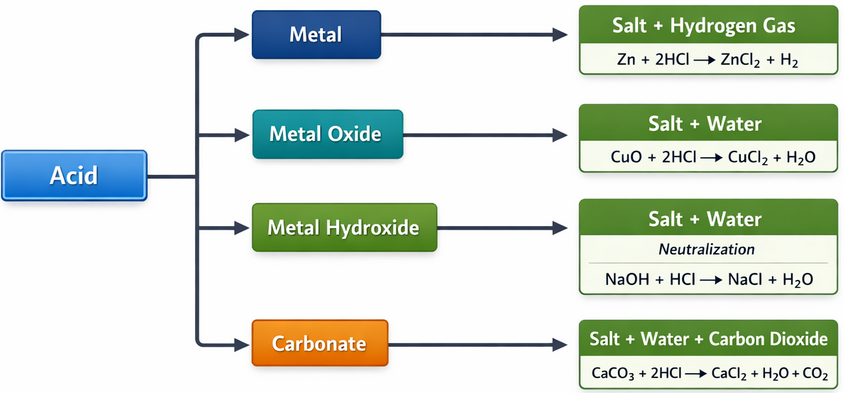
Example: Acid and Metal
Full equation:
\( \mathrm{Mg(s) + 2HCl(aq) \rightarrow MgCl_2(aq) + H_2(g)} \)
Ionic equation:
\( \mathrm{Mg(s) + 2H^+(aq) \rightarrow Mg^{2+}(aq) + H_2(g)} \)
Observation
- Effervescence occurs as hydrogen gas is produced.
- The metal gradually dissolves.
Precipitation Reactions
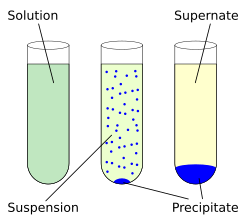
A precipitation reaction occurs when two aqueous solutions react to form an insoluble solid called a precipitate.
Example
Mixing silver nitrate solution with sodium chloride solution forms silver chloride.
Full equation:
\( \mathrm{AgNO_3(aq) + NaCl(aq) \rightarrow AgCl(s) + NaNO_3(aq)} \)
Ionic equation:
\( \mathrm{Ag^+(aq) + Cl^-(aq) \rightarrow AgCl(s)} \)
Observation
- A white precipitate of silver chloride forms.
Example 1
Copper metal is placed in silver nitrate solution. Write the ionic equation and state the observation.
▶️ Answer/Explanation
\( \mathrm{Cu(s) + 2Ag^+(aq) \rightarrow Cu^{2+}(aq) + 2Ag(s)} \)
Observation
- Grey silver metal forms.
- The solution gradually turns blue due to \( \mathrm{Cu^{2+}} \) ions.
Example 2
When barium chloride solution is mixed with sodium sulfate solution, a white precipitate forms. Write the ionic equation.
▶️ Answer/Explanation
\( \mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)} \)
The white solid formed is the precipitate \( \mathrm{BaSO_4} \).
Further Suggested Practicals
The following practical activities help develop experimental skills and reinforce key concepts from Topic 1. These experiments involve preparing salts, determining chemical formulae and equations, and observing typical test-tube reactions.
Preparation of a Salt and Calculation of Percentage Yield
A salt can be prepared by reacting an acid with a suitable base, metal, or carbonate. After isolating and drying the product, the mass obtained can be compared with the theoretical yield to calculate the percentage yield.

An example is the preparation of a double salt, such as ammonium iron(II) sulfate, from iron, ammonia and sulfuric acid.
The product is crystallised from solution, filtered, dried and weighed. The percentage yield is calculated using:
\( \text{Percentage yield} = \dfrac{\text{actual mass obtained}}{\text{theoretical mass}} \times 100\% \)
Determining a Chemical Formula by Experiment
The formula of a compound can be determined by measuring the masses of reactants or products in a reaction and calculating the ratio of moles.
For example, the formula of copper(II) oxide can be determined by reduction using hydrogen gas.
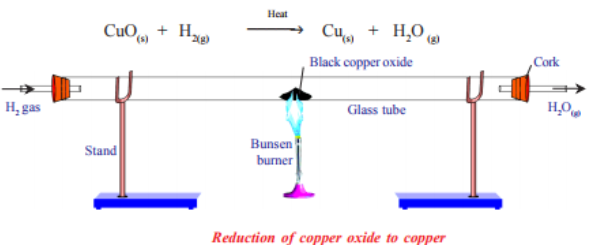
By measuring the mass of copper produced and the mass of copper oxide used, the ratio of copper to oxygen can be calculated to confirm the formula \( \mathrm{CuO} \).
Determining a Chemical Equation by Experiment
Experimental observations can be used to determine or confirm the balanced chemical equation for a reaction.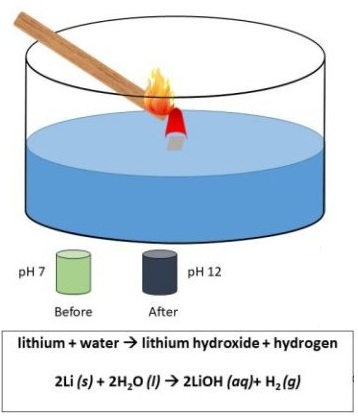
Examples include reactions such as lithium reacting with water or magnesium reacting with an acid.
Example: Lithium and Water
\( \mathrm{2Li(s) + 2H_2O(l) \rightarrow 2LiOH(aq) + H_2(g)} \)
Observation
- Lithium floats and moves on the surface of the water.
- Effervescence occurs as hydrogen gas is produced.
- The solution becomes alkaline due to lithium hydroxide.
Simple Test-Tube Reactions
Simple test-tube reactions allow students to observe chemical changes directly and relate these observations to full and ionic equations.
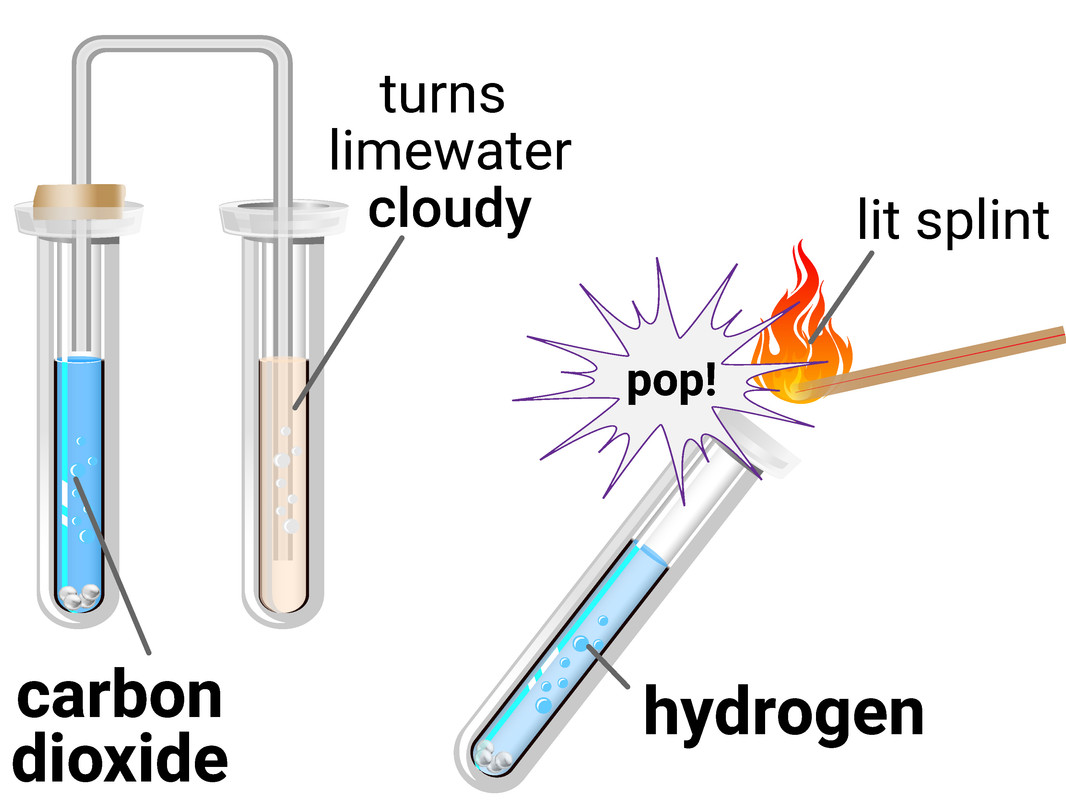
These reactions include the types discussed in section 1.12:
- displacement reactions
- typical reactions of acids
- precipitation reactions
By comparing observations with the corresponding equations, students can understand how ionic reactions explain the changes seen during experiments.
Example
A student prepares copper(II) sulfate crystals by reacting copper(II) oxide with sulfuric acid. After filtration and crystallisation, the student obtains \( 7.8\ \mathrm{g} \) of copper(II) sulfate crystals. The theoretical yield calculated from the reaction is \( 10.0\ \mathrm{g} \).
Calculate the percentage yield of the product.
▶️ Answer/Explanation
Use the percentage yield equation:
\( \text{Percentage yield} = \dfrac{\text{actual yield}}{\text{theoretical yield}} \times 100 \)
Substitute the values:
\( \dfrac{7.8}{10.0} \times 100 \)
\( = 78\% \)
Therefore the percentage yield of copper(II) sulfate crystals is 78%.
