Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.2 The mole and Avogadro constant-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.2 The mole and Avogadro constant- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.2 The mole and Avogadro constant- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.2 know that the mole (mol) is the unit for the amount of a substance and be able to perform calculations using the Avogadro constant L (6.02 x 1023 mol-1)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.2 The Mole and the Avogadro Constant
In chemistry, we often need to count extremely large numbers of atoms, molecules, or ions. Because these particles are extremely small, chemists use a special unit called the mole to measure the amount of a substance.
Mole
The mole (mol) is the SI unit used to measure the amount of substance.
One mole of any substance contains a fixed number of particles (atoms, molecules, ions, or electrons).
Avogadro Constant
The Avogadro constant \( L \) is the number of particles in one mole of a substance.
\( L = 6.02 \times 10^{23} \; \mathrm{mol^{-1}} \)
This means that one mole of any substance contains \( 6.02 \times 10^{23} \) particles.
Examples
- 1 mol of carbon atoms contains \( 6.02 \times 10^{23} \) carbon atoms.
- 1 mol of water molecules contains \( 6.02 \times 10^{23} \) molecules.
- 1 mol of sodium ions contains \( 6.02 \times 10^{23} \) ions.
Relationship Between Moles and Number of Particles
The number of particles in a substance can be calculated using the Avogadro constant.
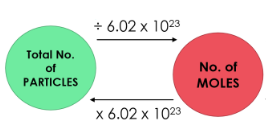
Number of particles = number of moles \( \times \) Avogadro constant
\( N = nL \)
Where:
- \( N \) = number of particles
- \( n \) = number of moles
- \( L = 6.02 \times 10^{23} \mathrm{mol^{-1}} \)
Example 1
How many atoms are present in \( 2.0 \) mol of helium?
▶️ Answer/Explanation
Use the relationship
\( N = nL \)
Substitute the values:
\( N = 2.0 \times 6.02 \times 10^{23} \)
\( N = 1.204 \times 10^{24} \)
Therefore, there are \( 1.204 \times 10^{24} \) helium atoms.
Example 2
A sample contains \( 3.01 \times 10^{23} \) molecules of oxygen. Calculate the number of moles present.
▶️ Answer/Explanation
Use the relationship
\( n = \dfrac{N}{L} \)
Substitute the values:
\( n = \dfrac{3.01 \times 10^{23}}{6.02 \times 10^{23}} \)
\( n = 0.50 \; \mathrm{mol} \)
Therefore, the sample contains \( 0.50 \) mol of oxygen molecules.
