Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 1.3 Balanced full equations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.3 Balanced full equations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 1.3 Balanced full equations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
1.3 write balanced full and ionic equations, including state symbols, for chemical reactions
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
1.3 Balanced Full and Ionic Equations
Chemical reactions are represented using chemical equations. A correct equation must be balanced, meaning that the number of atoms of each element is the same on both sides of the equation.
Chemical equations may be written as full equations or ionic equations. State symbols are also included to show the physical state of each substance.
Balanced Chemical Equation
A balanced equation shows the correct ratio of reactants and products so that the number of atoms of each element is the same on both sides of the reaction.
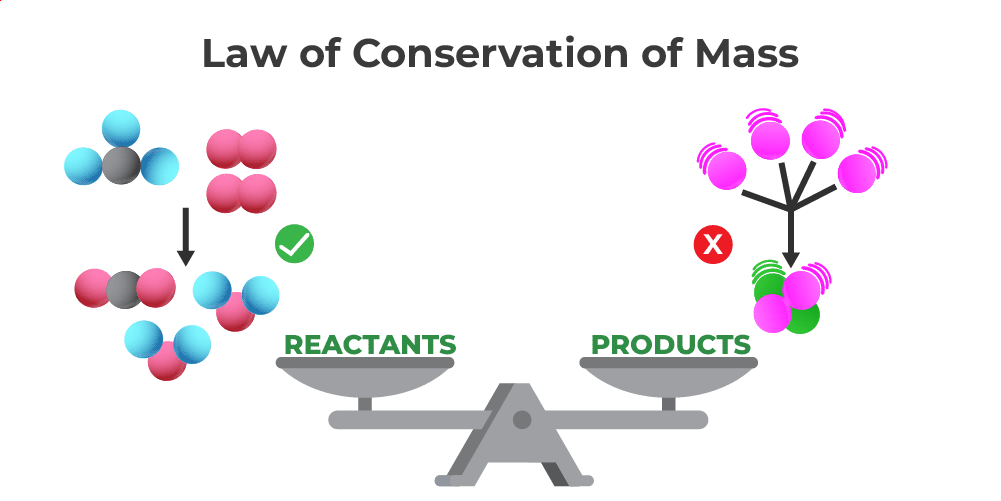
Balancing equations follows the law of conservation of mass, which states that matter is neither created nor destroyed in a chemical reaction.
Example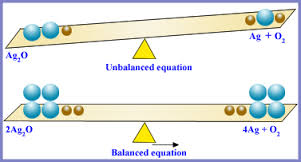
Unbalanced equation:
\( \mathrm{H_2 + O_2 \rightarrow H_2O} \)
Balanced equation:
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
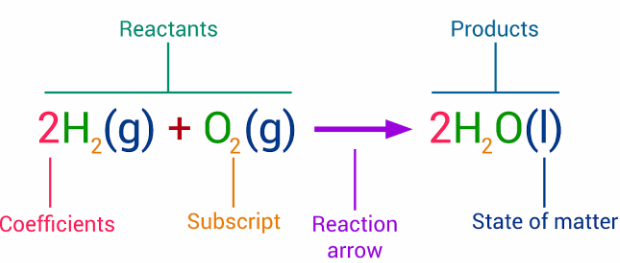
State Symbols
State symbols show the physical state of each substance involved in a reaction.
| State Symbol | Meaning |
|---|---|
| \( \mathrm{(s)} \) | Solid |
| \( \mathrm{(l)} \) | Liquid |
| \( \mathrm{(g)} \) | Gas |
| \( \mathrm{(aq)} \) | Aqueous (dissolved in water) |
Example with State Symbols
Full Ionic Equations
A full ionic equation shows all soluble ionic compounds as separate ions in aqueous solution.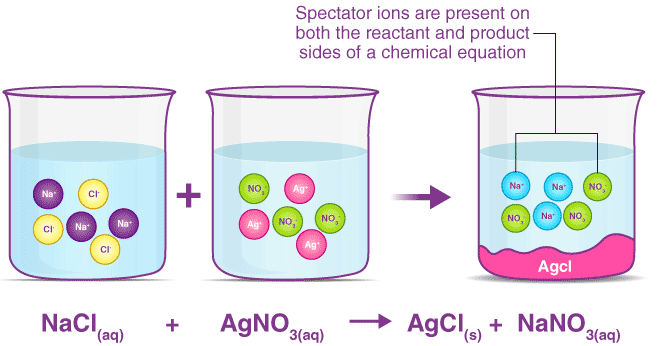
In aqueous solutions, ionic compounds dissociate into their ions.
Example
Reaction between silver nitrate and sodium chloride:
Full equation:
\( \mathrm{AgNO_3(aq) + NaCl(aq) \rightarrow AgCl(s) + NaNO_3(aq)} \)
Full ionic equation:
\( \mathrm{Ag^+(aq) + NO_3^-(aq) + Na^+(aq) + Cl^-(aq) \rightarrow AgCl(s) + Na^+(aq) + NO_3^-(aq)} \)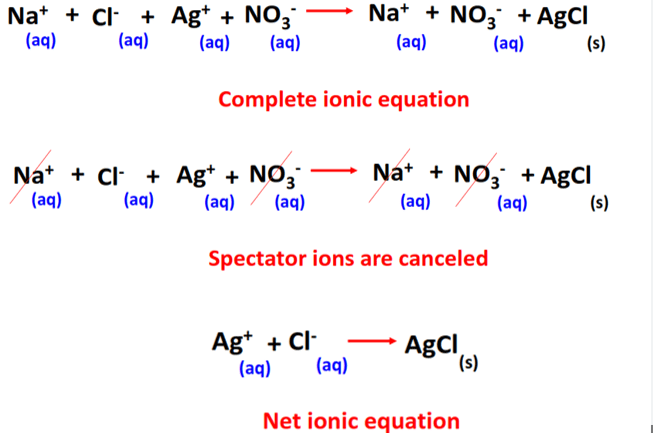
Spectator Ions
Spectator ions are ions that appear on both sides of the ionic equation and do not participate in the reaction.
In the example above, \( \mathrm{Na^+} \) and \( \mathrm{NO_3^-} \) are spectator ions.
Net Ionic Equation
The net ionic equation is obtained by removing spectator ions from the full ionic equation.
\( \mathrm{Ag^+(aq) + Cl^-(aq) \rightarrow AgCl(s)} \)
Example 1
Write the balanced equation for the reaction between zinc and hydrochloric acid including state symbols.
▶️ Answer/Explanation
Zinc reacts with hydrochloric acid to produce zinc chloride and hydrogen gas.
\( \mathrm{Zn(s) + 2HCl(aq) \rightarrow ZnCl_2(aq) + H_2(g)} \)
The equation is balanced because the number of each type of atom is the same on both sides.
Example 2
Write the net ionic equation for the reaction between barium chloride and sodium sulfate.
▶️ Answer/Explanation
First write the full equation:
\( \mathrm{BaCl_2(aq) + Na_2SO_4(aq) \rightarrow BaSO_4(s) + 2NaCl(aq)} \)
Write the full ionic equation:
\( \mathrm{Ba^{2+}(aq) + 2Cl^-(aq) + 2Na^+(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s) + 2Na^+(aq) + 2Cl^-(aq)} \)
Remove spectator ions \( \mathrm{Na^+} \) and \( \mathrm{Cl^-} \):
\( \mathrm{Ba^{2+}(aq) + SO_4^{2-}(aq) \rightarrow BaSO_4(s)} \)
